J Korean Med Assoc.
2003 Dec;46(12):1064-1069. 10.5124/jkma.2003.46.12.1064.
Development of Clinical Trials: 'Why It is Necessary and How It can be Achieved?'
- KMID: 2183139
- DOI: http://doi.org/10.5124/jkma.2003.46.12.1064
Abstract
- No abstract available.
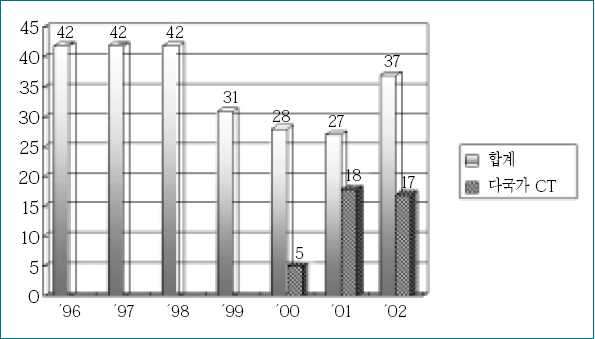
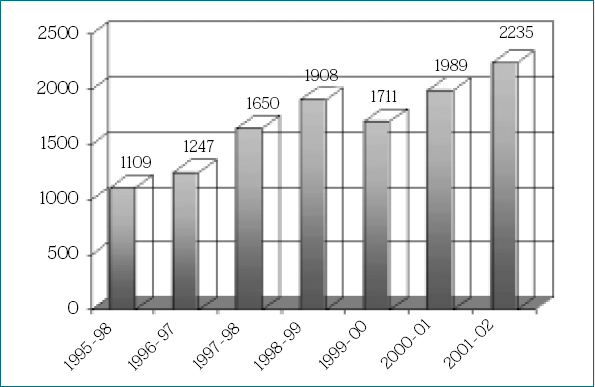
Figure
Reference
-
1. Griffin JP, O'Grady J. The Regulation of Medical Products. BMJ. 2003.2. Spilker B. Guide to Clinical trials. 1996. Lippincott-Raven.3. Fletcher AJ, Edwards LD, Fox AW, Stoner P. Principle and Practice of Pharmaceutical Medicine. 2002. Wiley.5. Woolley K, Woolley M. Clinical Trials in Australia : Findings out the FACTS(First Australian Clinical Trial Survey). 2003. GCPI;13–16.6. Baume PE. A Question of Balance : Report on the Future of Drug Evaluation in Australia. 1991. Canberra: AGPS.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Non-Inferiority Trials in Stroke Research: What Are They, and How Should We Interpret Them?
- Clinical trials for vaccine development in registry of Korea Food and Drug Administration
- Knowledge and Attitudes of Oncology Nurses towards Clinical Trials
- Chemotherapy in Rectal Cancer
- Understanding noninferiority trials