J Clin Neurol.
2015 Oct;11(4):305-310. 10.3988/jcn.2015.11.4.305.
Postthrombolytic Antiplatelet Use for Patients with Intercerebral Hemorrhage without Extensive Parenchymal Involvement Does Not Worsen Outcome
- Affiliations
-
- 1Department of Neurology, Beijing Tian Tan Hospital, Capital Medical University, Beijing, China. yongjunwang1962@gmail.com
- 2Department of Neurology, Beijing Chao-Yang Hospital, Capital Medical University, Beijing, China.
- KMID: 2179749
- DOI: http://doi.org/10.3988/jcn.2015.11.4.305
Abstract
- BACKGROUND AND PURPOSE
It is unclear whether postthrombolytic antiplatelet (AP) therapy after thrombolytic-related hemorrhage without extensive parenchymal involvement (THEPI) affects the clinical outcome. This study explored whether AP administration in patients with THEPI affects short- and long-term outcomes.
METHODS
All of the data for this study were collected from the Thrombolysis Implementation and Monitor of Acute Ischemic Stroke in China (TIMS-China) registry. Patients with THEPI were assigned to either the AP (AP therapy should be commenced 24 h after intravenous thrombolysis) or AP-naive groups. THEPI was defined according to European-Australasian Acute Stroke Study II criteria. The 90-day functional outcome, 7-day National Institutes of Health Stroke Scale (NIHSS) score, and 7-day and 90-day mortalities were compared between the AP and AP-naive groups. Logistic regression analysis was used to evaluate the effects of AP therapy on the short- and long-term clinical outcomes.
RESULTS
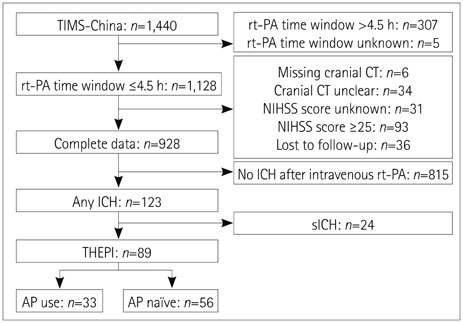
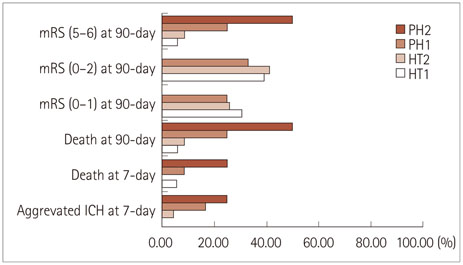
Of the 928 patients enrolled from those in the TIMS-China registry (n=1,440), 89 (9.6%) had nonsymptomatic intracerebral hemorrhage (ICH) within 24-36 h after thrombolysis; 33 (37%) of these patients were given AP therapy (AP group) and 56 (63%) were not (AP-naive group). No significant differences were found for the risk of 7-day aggravated ICH (p=0.998), 7-day NIHSS score (p=0.5491), 7-day mortality [odds ratio (OR)=3.427; 95% confidence interval (95% CI)=0.344-34.160; p=0.294], 90-day mortality (OR=0.788, 95% CI=0.154-4.040, p=0.775), or modified Rankin score 5 or 6 at 90-days (OR=1.108, 95% CI=0.249-4.928, p=0.893) between the AP and AP-naive groups after THEPI.
CONCLUSIONS
Early administration of postthrombolytic AP therapy after THEPI does not worsen either the short- or long-term outcome. AP therapy may be a reasonable treatment option for patients with THEPI to reduce the risk of ischemic stroke recurrence.
MeSH Terms
Figure
Reference
-
1. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med. 1995; 333:1581–1587.2. Hacke W, Kaste M, Fieschi C, von Kummer R, Davalos A, Meier D, et al. Second European-Australasian Acute Stroke Study Investigators. Randomised double-blind placebo-controlled trial of thrombolytic therapy with intravenous alteplase in acute ischaemic stroke (ECASS II). Lancet. 1998; 352:1245–1251.
Article3. Sussman ES, Connolly ES Jr. Hemorrhagic transformation: a review of the rate of hemorrhage in the major clinical trials of acute ischemic stroke. Front Neurol. 2013; 4:69.
Article4. Trouillas P, von Kummer R. Classification and pathogenesis of cerebral hemorrhages after thrombolysis in ischemic stroke. Stroke. 2006; 37:556–561.
Article5. Molina CA, Alvarez-Sabín J, Montaner J, Abilleira S, Arenillas JF, Coscojuela P, et al. Thrombolysis-related hemorrhagic infarction: a marker of early reperfusion, reduced infarct size, and improved outcome in patients with proximal middle cerebral artery occlusion. Stroke. 2002; 33:1551–1556.6. Kerenyi L, Kardos L, Szász J, Szatmári S, Bereczki D, Hegedüs K, et al. Factors influencing hemorrhagic transformation in ischemic stroke: a clinicopathological comparison. Eur J Neurol. 2006; 13:1251–1255.
Article7. Kernan WN, Ovbiagele B, Black HR, Bravata DM, Chimowitz MI, Ezekowitz MD, et al. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014; 45:2160–2236.
Article8. Park JH, Ko Y, Kim WJ, Jang MS, Yang MH, Han MK, et al. Is asymptomatic hemorrhagic transformation really innocuous? Neurology. 2012; 78:421–426.
Article9. Kent DM, Hinchey J, Price LL, Levine SR, Selker HP. In acute ischemic stroke, are asymptomatic intracranial hemorrhages clinically innocuous? Stroke. 2004; 35:1141–1146.
Article10. Libman R, Kwiakowski T, Lyden P, Grotta JC, Tilley BC, Fagen SC, et al. Asymptomatic hemorrhagic transformation of cerebral infarction does not worsen long-term outcome. J Stroke Cerebrovasc Dis. 2005; 14:50–54.
Article11. Kim JT, Heo SH, Park MS, Chang J, Choi KH, Cho KH. Use of antithrombotics after hemorrhagic transformation in acute ischemic stroke. PLoS One. 2014; 9:e89798.
Article12. Liao XL, Wang CX, Wang YL, Wang CJ, Zhao XQ, Zhang LQ, et al. Implementation and outcome of thrombolysis with alteplase 3 to 4.5 h after acute stroke in Chinese patients. CNS Neurosci Ther. 2013; 19:43–47.
Article13. Wahlgren N, Ahmed N, Dávalos A, Ford GA, Grond M, Hacke W, et al. Thrombolysis with alteplase for acute ischaemic stroke in the Safe Implementation of Thrombolysis in Stroke-Monitoring Study (SITS-MOST): an observational study. Lancet. 2007; 369:275–282.
Article14. Kimura K, Iguchi Y, Shibazaki K, Aoki J, Terasawa Y. Hemorrhagic transformation of ischemic brain tissue after t-PA thrombolysis as detected by MRI may be asymptomatic, but impair neurological recovery. J Neurol Sci. 2008; 272:136–142.
Article15. Derex L, Nighoghossian N. Intracerebral haemorrhage after thrombolysis for acute ischaemic stroke: an update. J Neurol Neurosurg Psychiatry. 2008; 79:1093–1099.
Article16. Jickling GC, Liu D, Stamova B, Ander BP, Zhan X, Lu A, et al. Hemorrhagic transformation after ischemic stroke in animals and humans. J Cereb Blood Flow Metab. 2014; 34:185–199.
Article17. Jia W, Zhou L. Use of antiplatelets. A survey of secondary prevention of ischemic stroke with intracranial hemorrhage history in Chinese patients. Neurosciences (Riyadh). 2011; 16:335–339.18. Kablau M, Kreisel SH, Sauer T, Binder J, Szabo K, Hennerici MG, et al. Predictors and early outcome of hemorrhagic transformation after acute ischemic stroke. Cerebrovasc Dis. 2011; 32:334–341.
Article19. Dzialowski I, Pexman JH, Barber PA, Demchuk AM, Buchan AM, Hill MD, et al. Asymptomatic hemorrhage after thrombolysis may not be benign: prognosis by hemorrhage type in the Canadian alteplase for stroke effectiveness study registry. Stroke. 2007; 38:75–79.
Article20. Wang Y, Liao X, Zhao X, Wang DZ, Wang C, Nguyen-Huynh MN, et al. Using recombinant tissue plasminogen activator to treat acute ischemic stroke in China: analysis of the results from the Chinese National Stroke Registry (CNSR). Stroke. 2011; 42:1658–1664.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of blood amount following single antiplatelet therapy and dual antiplatelet therapy in patients with traumatic acute subdural hemorrhage
- Dual Antiplatelet Therapy after Noncardioembolic Ischemic Stroke or Transient Ischemic Attack: Pros and Cons
- The Effect of Preoperative Antiplatelet Therapy on Hemorrhagic Complications after Decompressive Craniectomy in Patients with Traumatic Brain Injury
- Discontinuation of antiplatelet therapy after stent-assisted coil embolization for cerebral aneurysms
- Evaluation of Bleeding After Denal Extraction in Patients Taking Single Antiplatelet Treatment