J Cardiovasc Ultrasound.
2014 Jun;22(2):72-79. 10.4250/jcu.2014.22.2.72.
Association of Myocardial Angiogenesis with Structural and Functional Ventricular Remodeling in Aortic Stenosis Patients with Normal Ejection Fraction
- Affiliations
-
- 1Department of Internal Medicine and Cardiovascular Center, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea. splee0624@gmail.com
- KMID: 2177462
- DOI: http://doi.org/10.4250/jcu.2014.22.2.72
Abstract
- BACKGROUND
Although rarefaction of myocardial angiogenesis has been shown to be associated with left ventricular (LV) systolic dysfunction in animal models of ventricular hypertrophy, this relationship has not been investigated in depth nor validated in humans. We aimed to analyze the relationship of myocardial angiogenesis with various functional and structural ventricular remodeling parameters in moderate to severe aortic stenosis (AS) patients with normal LV ejection fraction (LVEF).
METHODS
A total of 38 moderate or severe AS patients with LVEF > 50% were enrolled for the current study and all patients underwent LV endomyocardial biopsy at the septum during aortic valve replacement. The biopsy specimens were stained for platelet endothelial cell adhesion molecule-1 (CD31) to analyze the density of blood vessels in the myocardium.
RESULTS
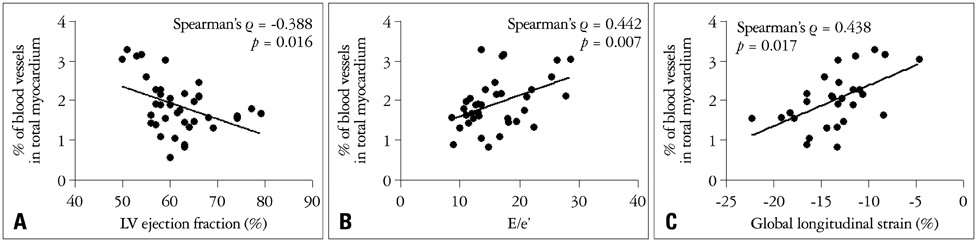
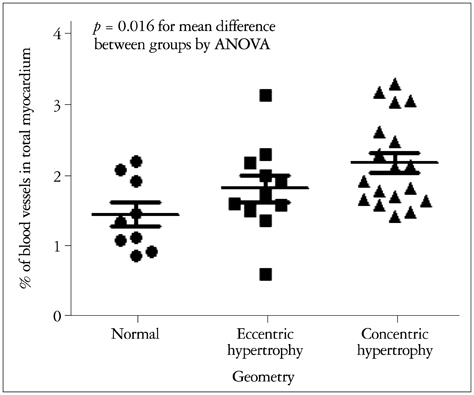
The degree of myocardial angiogenesis tended to increase with worse myocardial systolic function, LV filling pressure and progressed ventricular hypertrophy (Spearman's rho = -0.388, p = 0.016 for LVEF; Spearman's rho = 0.442, p = 0.007 for E/e'; Spearman's rho = 0.424, p = 0.008 for LV mass index). The degree of myocardial angiogenesis was also significantly associated with the degree of aortic valve stenosis (Spearman's rho = -0.368, p = 0.023). There was significant difference in the degree of myocardial angiogenesis according to the LV geometry (p = 0.016 for mean difference between different LV geometry groups by analysis of variance). Significant predictors of myocardial blood vessel density were LV mass index (beta = 0.398, p = 0.010) and LVEF (beta = -0.313, p = 0.028).
CONCLUSION
There is a close relationship between myocardial angiogenesis and LV remodeling in moderate to severe AS patients with normal LVEF, with angiogenesis increasing with LV hypertrophy. Further studies to demonstrate the mechanism underlying this phenomenon is warranted.
MeSH Terms
Figure
Reference
-
1. Freeman RV, Otto CM. Spectrum of calcific aortic valve disease: pathogenesis, disease progression, and treatment strategies. Circulation. 2005; 111:3316–3326.2. Otto CM. Valvular aortic stenosis: disease severity and timing of intervention. J Am Coll Cardiol. 2006; 47:2141–2151.3. Pibarot P, Dumesnil JG. Improving assessment of aortic stenosis. J Am Coll Cardiol. 2012; 60:169–180.
Article4. Krayenbuehl HP, Hess OM, Monrad ES, Schneider J, Mall G, Turina M. Left ventricular myocardial structure in aortic valve disease before, intermediate, and late after aortic valve replacement. Circulation. 1989; 79:744–755.
Article5. Hein S, Arnon E, Kostin S, Schönburg M, Elsässer A, Polyakova V, Bauer EP, Klövekorn WP, Schaper J. Progression from compensated hypertrophy to failure in the pressure-overloaded human heart: structural deterioration and compensatory mechanisms. Circulation. 2003; 107:984–991.
Article6. Shiojima I, Sato K, Izumiya Y, Schiekofer S, Ito M, Liao R, Colucci WS, Walsh K. Disruption of coordinated cardiac hypertrophy and angiogenesis contributes to the transition to heart failure. J Clin Invest. 2005; 115:2108–2118.
Article7. Orsinelli DA, Aurigemma GP, Battista S, Krendel S, Gaasch WH. Left ventricular hypertrophy and mortality after aortic valve replacement for aortic stenosis. A high risk subgroup identified by preoperative relative wall thickness. J Am Coll Cardiol. 1993; 22:1679–1683.8. Cioffi G, Faggiano P, Vizzardi E, Tarantini L, Cramariuc D, Gerdts E, de Simone G. Prognostic effect of inappropriately high left ventricular mass in asymptomatic severe aortic stenosis. Heart. 2011; 97:301–307.
Article9. Lee SP, Kim YJ, Kim JH, Park K, Kim KH, Kim HK, Cho GY, Sohn DW, Oh BH, Park YB. Deterioration of myocardial function in paradoxical low-flow severe aortic stenosis: two-dimensional strain analysis. J Am Soc Echocardiogr. 2011; 24:976–983.
Article10. Tomanek RJ. Response of the coronary vasculature to myocardial hypertrophy. J Am Coll Cardiol. 1990; 15:528–533.
Article11. Flanagan MF, Aoyagi T, Arnold LW, Maute C, Fujii AM, Currier J, Bergau D, Warren HB, Rakusan K. Effects of chronic heparin administration on coronary vascular adaptation to hypertension and ventricular hypertrophy in sheep. Circulation. 1999; 100:981–987.
Article12. Friehs I, Margossian RE, Moran AM, Cao-Danh H, Moses MA, del Nido PJ. Vascular endothelial growth factor delays onset of failure in pressure-overload hypertrophy through matrix metalloproteinase activation and angiogenesis. Basic Res Cardiol. 2006; 101:204–213.
Article13. Ozkan A, Kapadia S, Tuzcu M, Marwick TH. Assessment of left ventricular function in aortic stenosis. Nat Rev Cardiol. 2011; 8:494–501.
Article14. American College of Cardiology. American Heart Association Task Force on Practice Guidelines (Writing Committee to revise the 1998 guidelines for the management of patients with valvular heart disease). Society of Cardiovascular Anesthesiologists. Bonow RO, Carabello BA, Chatterjee K, de Leon AC Jr, Faxon DP, Freed MD, Gaasch WH, Lytle BW, Nishimura RA, O'Gara PT, O'Rourke RA, Otto CM, Shah PM, Shanewise JS, Smith SC Jr, Jacobs AK, Adams CD, Anderson JL, Antman EM, Fuster V, Halperin JL, Hiratzka LF, Hunt SA, Lytle BW, Nishimura R, Page RL, Riegel B. ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing Committee to Revise the 1998 guidelines for the management of patients with valvular heart disease) developed in collaboration with the Society of Cardiovascular Anesthesiologists endorsed by the Society for Cardiovascular Angiography and Interventions and the Society of Thoracic Surgeons. J Am Coll Cardiol. 2006; 48:e1–e148.15. Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, Picard MH, Roman MJ, Seward J, Shanewise J, Solomon S, Spencer KT, St John Sutton M, Stewart W. American Society of Echocardiography's Nomenclature and Standards Committee. Task Force on Chamber Quantification. American College of Cardiology Echocardiography Committee. American Heart Association. European Association of Echocardiography, European Society of Cardiology. Recommendations for chamber quantification. Eur J Echocardiogr. 2006; 7:79–108.
Article16. Hachicha Z, Dumesnil JG, Bogaty P, Pibarot P. Paradoxical low-flow, low-gradient severe aortic stenosis despite preserved ejection fraction is associated with higher afterload and reduced survival. Circulation. 2007; 115:2856–2864.
Article17. Devereux RB, Reichek N. Echocardiographic determination of left ventricular mass in man. Anatomic validation of the method. Circulation. 1977; 55:613–618.
Article18. Chambers JB. Aortic stenosis. Eur J Echocardiogr. 2009; 10:i11–i19.
Article19. Reisner SA, Lysyansky P, Agmon Y, Mutlak D, Lessick J, Friedman Z. Global longitudinal strain: a novel index of left ventricular systolic function. J Am Soc Echocardiogr. 2004; 17:630–633.
Article20. Delgado V, Tops LF, van Bommel RJ, van der Kley F, Marsan NA, Klautz RJ, Versteegh MI, Holman ER, Schalij MJ, Bax JJ. Strain analysis in patients with severe aortic stenosis and preserved left ventricular ejection fraction undergoing surgical valve replacement. Eur Heart J. 2009; 30:3037–3047.
Article21. Lancellotti P, Donal E, Magne J, O'Connor K, Moonen ML, Cosyns B, Pierard LA. Impact of global left ventricular afterload on left ventricular function in asymptomatic severe aortic stenosis: a two-dimensional speckle-tracking study. Eur J Echocardiogr. 2010; 11:537–543.
Article22. De Boer RA, Pinto YM, Van Veldhuisen DJ. The imbalance between oxygen demand and supply as a potential mechanism in the pathophysiology of heart failure: the role of microvascular growth and abnormalities. Microcirculation. 2003; 10:113–126.
Article23. Frey N, Olson EN. Cardiac hypertrophy: the good, the bad, and the ugly. Annu Rev Physiol. 2003; 65:45–79.
Article24. Selvetella G, Hirsch E, Notte A, Tarone G, Lembo G. Adaptive and maladaptive hypertrophic pathways: points of convergence and divergence. Cardiovasc Res. 2004; 63:373–380.
Article25. Adams RH, Alitalo K. Molecular regulation of angiogenesis and lymphangiogenesis. Nat Rev Mol Cell Biol. 2007; 8:464–478.
Article26. Giordano FJ, Gerber HP, Williams SP, VanBruggen N, Bunting S, Ruiz-Lozano P, Gu Y, Nath AK, Huang Y, Hickey R, Dalton N, Peterson KL, Ross J Jr, Chien KR, Ferrara N. A cardiac myocyte vascular endothelial growth factor paracrine pathway is required to maintain cardiac function. Proc Natl Acad Sci U S A. 2001; 98:5780–5785.
Article27. Yoon YS, Uchida S, Masuo O, Cejna M, Park JS, Gwon HC, Kirchmair R, Bahlman F, Walter D, Curry C, Hanley A, Isner JM, Losordo DW. Progressive attenuation of myocardial vascular endothelial growth factor expression is a seminal event in diabetic cardiomyopathy: restoration of microvascular homeostasis and recovery of cardiac function in diabetic cardiomyopathy after replenishment of local vascular endothelial growth factor. Circulation. 2005; 111:2073–2085.
Article28. Tomanek RJ, Searls JC, Lachenbruch PA. Quantitative changes in the capillary bed during developing, peak, and stabilized cardiac hypertrophy in the spontaneously hypertensive rat. Circ Res. 1982; 51:295–304.
Article29. Breisch EA, White FC, Nimmo LE, Bloor CM. Cardiac vasculature and flow during pressure-overload hypertrophy. Am J Physiol. 1986; 251(5 Pt 2):H1031–H1037.
Article30. Steadman CD, Jerosch-Herold M, Grundy B, Rafelt S, Ng LL, Squire IB, Samani NJ, McCann GP. Determinants and functional significance of myocardial perfusion reserve in severe aortic stenosis. JACC Cardiovasc Imaging. 2012; 5:182–189.
Article31. Cramariuc D, Gerdts E, Davidsen ES, Segadal L, Matre K. Myocardial deformation in aortic valve stenosis: relation to left ventricular geometry. Heart. 2010; 96:106–112.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Assessment of Left and Right Ventricular Performance by Gated Blood Pool Scan in Acute Transmural Myocardial Infarction
- Evaluation of Ejection Fraction Obtained by Echocardiography and Radionuclide Ventriculography
- Myocardial Reperfusion and Long-Term Change of Left Ventricular Volume after Acute Anterior Wall Myocardial Infarction
- Exercise Echocardiography in Asymptomatic Patients with Severe Aortic Stenosis and Preserved Left Ventricular Ejection Fraction
- The Effects of Abciximab on Left Ventricular Remodeling in Patient with Acute Myocardial Infarction Treated with Primary Percutaneous Coronary Intervention