Ann Dermatol.
2010 Feb;22(1):81-84. 10.5021/ad.2010.22.1.81.
A Case of Complication after Matridex(R) Injection
- Affiliations
-
- 1Department of Dermatology, Seoul National University College of Medicine, Seoul, Korea.
- 2Department of Dermatology, Seoul National University Boramae Hospital, Seoul, Korea.
- 3Department of Dermatology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. chwon98@chol.com
- KMID: 2172046
- DOI: http://doi.org/10.5021/ad.2010.22.1.81
Abstract
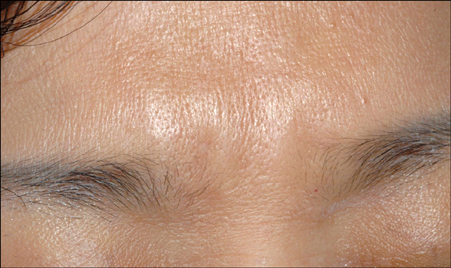
- Matridex(R) is an injectable skin filler that's composed of a mixture of cross linked hyaluronic acid and dextranomer particles, and it was recently developed for soft tissue augmentation. To the best of our knowledge, there have been few previous reports on complications associated with Matridex. We report here on a delayed inflammatory reaction to an injection of Matridex in the glabellar fold, and this developed five weeks after the injection and it lasted more than a year. The patient was treated with oral doxycycline and intralesional injection of triamcinolone acetonide; this resulted in almost complete resolution of the lesion. The patient should be informed of the potential complications with the use of injectable fillers before treatment, for it could lead to undesirable aesthetic consequences.
Keyword
MeSH Terms
Figure
Reference
-
1. Eppley BL, Summerlin DJ, Prevel CD, Sadove AM. Effects of a positively charged biomaterial for dermal and subcutaneous augmentation. Aesthetic Plast Surg. 1994. 18:413–416.
Article2. Massone C, Horn M, Kerl H, Ambros-Rudolph CM, Giovanna Brunasso AM, Cerroni L. Foreign body granuloma due to Matridex injection for cosmetic purposes. Am J Dermatopathol. 2009. 31:197–199.
Article3. Lemperle G, Morhenn V, Charrier U. Human histology and persistence of various injectable filler substances for soft tissue augmentation. Aesthetic Plast Surg. 2003. 27:354–367.
Article4. Lowe NJ, Maxwell CA, Lowe P, Duick MG, Shah K. Hyaluronic acid skin fillers: adverse reactions and skin testing. J Am Acad Dermatol. 2001. 45:930–933.
Article5. Friedman PM, Mafong EA, Kauvar AN, Geronemus RG. Safety data of injectable nonanimal stabilized hyaluronic acid gel for soft tissue augmentation. Dermatol Surg. 2002. 28:491–494.
Article6. Micheels P. Human anti-hyaluronic acid antibodies: is it possible? Dermatol Surg. 2001. 27:185–191.
Article7. Duranti F, Salti G, Bovani B, Calandra M, Rosati ML. Injectable hyaluronic acid gel for soft tissue augmentation. A clinical and histological study. Dermatol Surg. 1998. 24:1317–1325.8. Manna F, Dentini M, Desideri P, De Pita O, Mortilla E, Maras B. Comparative chemical evaluation of two commercially available derivatives of hyaluronic acid (hylaform from rooster combs and restylane from streptococcus) used for soft tissue augmentation. J Eur Acad Dermatol Venereol. 1999. 13:183–192.
Article9. Brody HJ. Use of hyaluronidase in the treatment of granulomatous hyaluronic acid reactions or unwanted hyaluronic acid misplacement. Dermatol Surg. 2005. 31:893–897.
Article10. Lloret P, Espana A, Leache A, Bauza A, Fernandez-Galar M, Idoate MA, et al. Successful treatment of granulomatous reactions secondary to injection of esthetic implants. Dermatol Surg. 2005. 31:486–490.
Article11. Bachelez H, Senet P, Cadranel J, Kaoukhov A, Dubertret L. The use of tetracyclines for the treatment of sarcoidosis. Arch Dermatol. 2001. 137:69–73.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Two Cases of Injection Complication Following an Illegal Practice
- Intracorporeal Needle Breakage during Intracavernous Self-injection of Vasoactive Agent: A Case Report
- Descemet's Membrane Detachment Associated with Inadvertent Viscoelastic Injection in Viscocanalostomy
- Pneumocephalus after Epidural Steroid Injection: A case report
- The Complications Developed after Repeated Botulinum Toxin Injection