Cancer Res Treat.
2014 Jul;46(3):223-233.
Cost-Effectiveness of Liver Cancer Screening in Adults at High Risk for Liver Cancer in the Republic of Korea
- Affiliations
-
- 1National Cancer Control Institute, National Cancer Center, Goyang, Korea. kschoi@ncc.re.kr
- 2Department of Social Medicine, College of Medicine, Dankook University, Cheonan, Korea.
- 3Department of Biostatistics, Yonsei University College of Medicine, Seoul, Korea.
- 4Department of Preventive Medicine and Institute of Health Services Research, Yonsei University College of Medicine, Seoul, Korea.
Abstract
- PURPOSE
This study was conducted in order to determine the most cost-effective strategy, in terms of interval and age range, for liver cancer screening in the high-risk population of Korea.
MATERIALS AND METHODS
A stochastic model was used to simulate the cost-effectiveness of liver cancer screening by combined ultrasonography and alpha-fetoprotein testing when varying both screening intervals and age ranges. The effectiveness of these screening strategies in the high-risk population was defined as the probability of detecting preclinical liver cancer, and cost was based on the direct cost of the screening and confirmative tests. Optimal cost-effectiveness was determined using the incremental cost-effectiveness ratio.
RESULTS
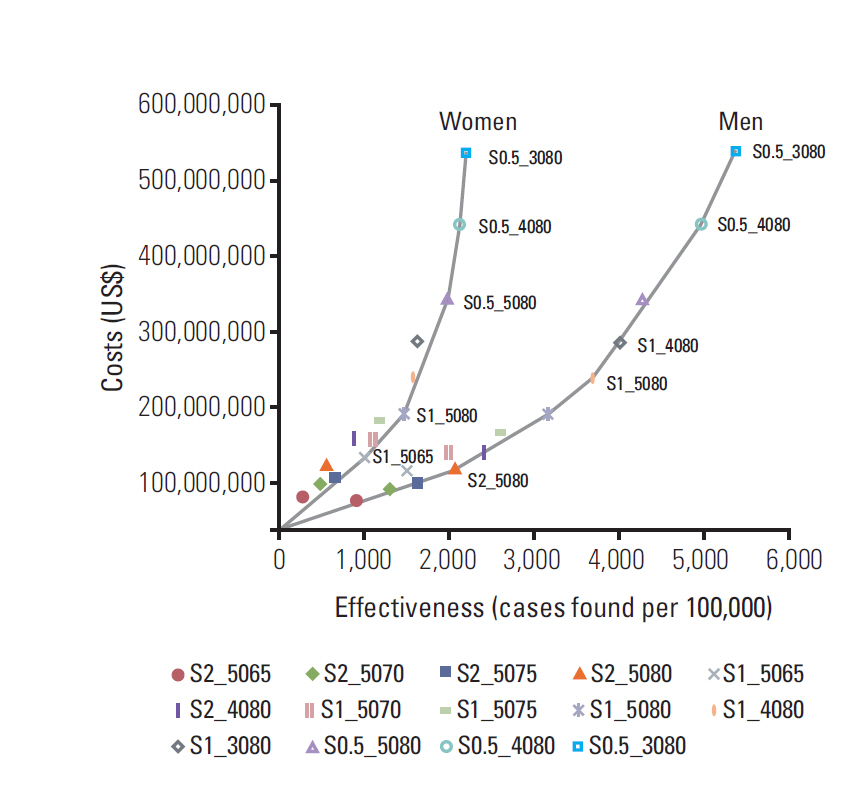
Among the 36 alternative strategies, one-year or two-year interval screening for men aged between 50 and 80 years, six-month or one-year interval screening for men aged between 40 and 80 years, and six-month interval screening for men aged between 30 and 80 years were identified as non-dominated strategies. For women, identified non-dominated strategies were: one-year interval screening between age 50 and 65 years, one-year or six-month interval screening between age 50 and 80 years, six-month interval screening between age 40 and 80 years, and six-month interval screening between age 30 and 80 years.
CONCLUSION
In Korea, a one-year screening interval for men aged 50 to 80 years would be marginally cost-effective. Further studies should be conducted in order to evaluate effectiveness of liver cancer screening, and compare the cost effectiveness of different liver cancer screening programs with a final outcome indicator such as quality-adjusted life-years or disability-adjusted life-years.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010; 127:2893–917.
Article2. Jung KW, Park S, Kong HJ, Won YJ, Lee JY, Seo HG, et al. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2009. Cancer Res Treat. 2012; 44:11–24.
Article3. Chen TH, Chen CJ, Yen MF, Lu SN, Sun CA, Huang GT, et al. Ultrasound screening and risk factors for death from hepatocellular carcinoma in a high risk group in Taiwan. Int J Cancer. 2002; 98:257–61.
Article4. Yang B, Zhang B, Xu Y, Wang W, Shen Y, Zhang A, et al. Prospective study of early detection for primary liver cancer. J Cancer Res Clin Oncol. 1997; 123:357–60.
Article5. Yuen MF, Cheng CC, Lauder IJ, Lam SK, Ooi CG, Lai CL. Early detection of hepatocellular carcinoma increases the chance of treatment: Hong Kong experience. Hepatology. 2000; 31:330–5.
Article6. Bolondi L, Sofia S, Siringo S, Gaiani S, Casali A, Zironi G, et al. Surveillance programme of cirrhotic patients for early diagnosis and treatment of hepatocellular carcinoma: a cost effectiveness analysis. Gut. 2001; 48:251–9.
Article7. Zhang BH, Yang BH, Tang ZY. Randomized controlled trial of screening for hepatocellular carcinoma. J Cancer Res Clin Oncol. 2004; 130:417–22.
Article8. National Cancer Institute. PDQ® Liver (Hepatocellular) Cancer Screening [Internet].Bethesda: National Cancer Institute;2012. [cited 2013 Feb 28]. Available from: http://cancer.gov/cancertopics/pdq/screening/hepatocellular/HealthProfessional.9. Daniele B. Alfa fetoprotein and ultrasonography screening. Hepatocellular carcinoma screening, diagnosis, and management. Bethesda: Natcher Conference Center, National Institutes of Health;2004. p. 69–73.10. Bruix J, Sherman M, Llovet JM, Beaugrand M, Lencioni R, Burroughs AK, et al. Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the Study of the Liver. J Hepatol. 2001; 35:421–30.11. Asia-Pacific Working Party on Prevention of Hepatocellular Carcinoma. Prevention of hepatocellular carcinoma in the Asia-Pacific region: consensus statements. J Gastroenterol Hepatol. 2010; 25:657–63.12. Han KH, Park JW. The usefulness and current status of the screening program for early diagnosis of hepatocellular carcinoma. J Korean Med Assoc. 2002; 45:972–80.
Article13. Lee SJ, Zelen M. Scheduling periodic examinations for the early detection of disease: applications to breast cancer. J Am Stat Assoc. 1998; 93:1271–81.
Article14. Sheu JC, Sung JL, Chen DS, Yang PM, Lai MY, Lee CS, et al. Growth rate of asymptomatic hepatocellular carcinoma and its clinical implications. Gastroenterology. 1985; 89:259–66.
Article15. Zhang B, Yang B. Combined alpha fetoprotein testing and ultrasonography as a screening test for primary liver cancer. J Med Screen. 1999; 6:108–10.16. National Cancer Center. Evaluation of orgnized cancer screening program in Korea. Seoul: Ministry of Health & Welfare;2010.17. Health Insurance Review and Assessment Services. The medical insurance cost in 2009. Seoul: Health Insurance Review and Assessment Service;2009.18. Taouli B, Goh JS, Lu Y, Qayyum A, Yeh BM, Merriman RB, et al. Growth rate of hepatocellular carcinoma: evaluation with serial computed tomography or magnetic resonance imaging. J Comput Assist Tomogr. 2005; 29:425–9.19. Trevisani F, De Notariis S, Rapaccini G, Farinati F, Benvegnu L, Zoli M, et al. Semiannual and annual surveillance of cirrhotic patients for hepatocellular carcinoma: effects on cancer stage and patient survival (Italian experience). Am J Gastroenterol. 2002; 97:734–44.
Article20. Kang JY, Lee TP, Yap I, Lun KC. Analysis of cost-effectiveness of different strategies for hepatocellular carcinoma screening in hepatitis B virus carriers. J Gastroenterol Hepatol. 1992; 7:463–8.
Article21. Thompson Coon J, Rogers G, Hewson P, Wright D, Anderson R, Jackson S, et al. Surveillance of cirrhosis for hepatocellular carcinoma: a cost-utility analysis. Br J Cancer. 2008; 98:1166–75.
Article22. Santi V, Trevisani F, Gramenzi A, Grignaschi A, Mirici-Cappa F, Del Poggio P, et al. Semiannual surveillance is superior to annual surveillance for the detection of early hepatocellular carcinoma and patient survival. J Hepatol. 2010; 53:291–7.
Article23. Juon HS, Choi KS, Park EC, Kwak MS, Lee S. Hepatitis B vaccinations among Koreans: Results from 2005 Korea National Cancer Screening Survey. BMC Infect Dis. 2009; 9:185.
Article24. Drummond M, O'Brien B, Stoddart G, Torrance GW. Methods for the economic evaluation of health care programmes. 2nd ed. New York: Oxford University Press;1997.25. Grosse SD. Assessing cost-effectiveness in healthcare: history of the $50,000 per QALY threshold. Expert Rev Pharmacoecon Outcomes Res. 2008; 8:165–78.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Methodological Review of Cost Effectiveness Analysis of Cancer Screening
- Relationships between Knowledge about Early Detection, Cancer Risk Perception and Cancer Screening Tests in the General Public Aged 40 and Over
- Prevention of liver cancer: basic and clinical aspects
- Liver Magnetic Resonance Imaging for Hepatocellular Carcinoma Surveillance
- Cost-benefit issues about human papillomavirus (HPV) testing