Cancer Res Treat.
2012 Mar;44(1):43-49.
Weekly Gemcitabine and Docetaxel in Refractory Soft Tissue Sarcoma: A Retrospective Analysis
- Affiliations
-
- 1Department of Internal Medicine, Yonsei Cancer Center, Yonsei University College of Medicine, Seoul, Korea. rha7655@yuhs.ac
Abstract
- PURPOSE
The combination of gemcitabine and docetaxel (GD) is used to effectively treat patients with soft tissue sarcoma (STS). It is widely considered that the conventional doses used are too high for long term use and many patients must discontinue GD treatment due to its toxicity. Therefore, to determine the appropriate dose meeting acceptable efficacy results, while minimizing toxic side effects, we treated patients with a weekly infusion of GD (weekly GD).
MATERIALS AND METHODS
A total of 22 patients presenting a variety of STSs were treated at Yonsei Cancer Center. All patients had metastatic or recurrent cancer and had previously received doxorubicin and ifosfamide combination chemotherapy. In all cases, gemcitabine (1,000 mg/m2) and docetaxel (35 mg/m2) were administered intravenously on days 1 and 8 of a 21-day cycle. We retrospectively reviewed the medical records of these patients.
RESULTS
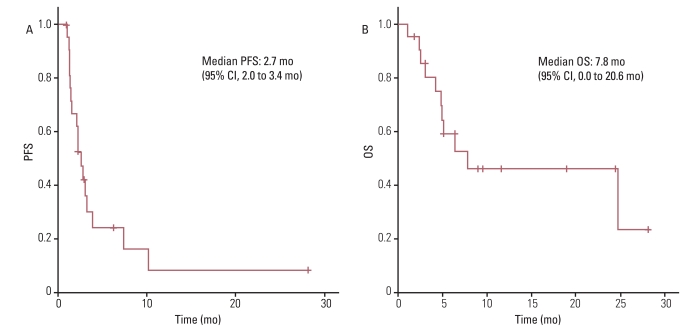
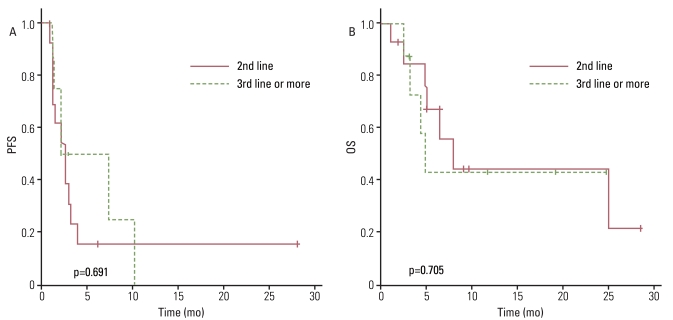
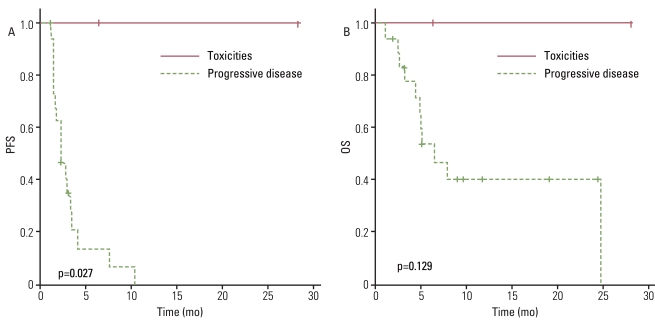
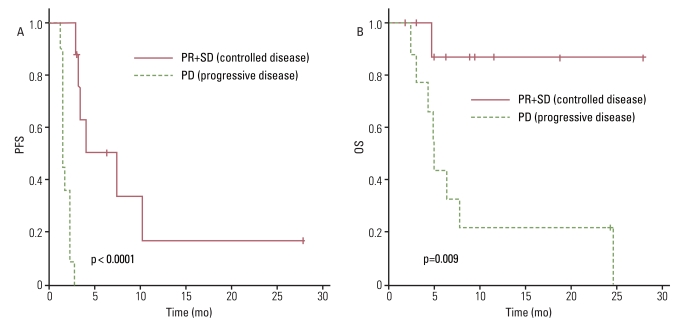
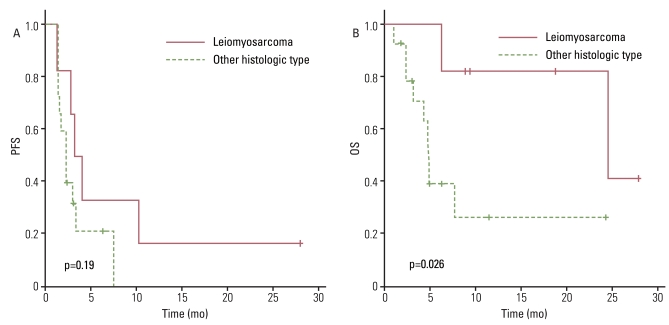
The response rate was 4.5%, with one patient diagnosed with leiomyosarcoma having a partial response, and the disease control rate was 40.9%. The median progression-free survival (PFS) duration was 2.7 months and the PFS was correlated with the treatment response to a weekly GD. The median overall survival (OS) duration was 7.8 months and the OS was correlated with histology. There was no significant difference in OS between patients who received weekly GD as a 2nd line chemotherapy and those who received 3rd line or more. Treatment was generally well tolerated.
CONCLUSION
Weekly GD was well tolerated and showed moderate efficacy, indicating that this could be a reasonable option as a salvage treatment for metastatic STS.
Keyword
MeSH Terms
Figure
Reference
-
1. Patel SR, Gandhi V, Jenkins J, Papadopolous N, Burgess MA, Plager C, et al. Phase II clinical investigation of gemcitabine in advanced soft tissue sarcomas and window evaluation of dose rate on gemcitabine triphosphate accumulation. J Clin Oncol. 2001; 19:3483–3489. PMID: 11481354.
Article2. Okuno S, Edmonson J, Mahoney M, Buckner JC, Frytak S, Galanis E. Phase II trial of gemcitabine in advanced sarcomas. Cancer. 2002; 94:3225–3229. PMID: 12115355.
Article3. van Hoesel QG, Verweij J, Catimel G, Clavel M, Kerbrat P, van Oosterom AT, et al. EORTC Soft Tissue and Bone Sarcoma Group. Phase II study with docetaxel (Taxotere) in advanced soft tissue sarcomas of the adult. Ann Oncol. 1994; 5:539–542. PMID: 7918126.
Article4. Verweij J, Lee SM, Ruka W, Buesa J, Coleman R, van Hoessel R, et al. Randomized phase II study of docetaxel versus doxorubicin in first- and second-line chemotherapy for locally advanced or metastatic soft tissue sarcomas in adults: a study of the European organization for research and treatment of cancer soft tissue and bone sarcoma group. J Clin Oncol. 2000; 18:2081–2086. PMID: 10811673.
Article5. Edmonson JH, Ebbert LP, Nascimento AG, Jung SH, McGaw H, Gerstner JB. Phase II study of docetaxel in advanced soft tissue sarcomas. Am J Clin Oncol. 1996; 19:574–576. PMID: 8931674.
Article6. Köstler WJ, Brodowicz T, Attems Y, Hejna M, Tomek S, Amann G, et al. Docetaxel as rescue medication in anthracycline- and ifosfamide-resistant locally advanced or metastatic soft tissue sarcoma: results of a phase II trial. Ann Oncol. 2001; 12:1281–1288. PMID: 11697841.
Article7. Hensley ML, Maki R, Venkatraman E, Geller G, Lovegren M, Aghajanian C, et al. Gemcitabine and docetaxel in patients with unresectable leiomyosarcoma: results of a phase II trial. J Clin Oncol. 2002; 20:2824–2831. PMID: 12065559.
Article8. Leu KM, Ostruszka LJ, Shewach D, Zalupski M, Sondak V, Biermann JS, et al. Laboratory and clinical evidence of synergistic cytotoxicity of sequential treatment with gemcitabine followed by docetaxel in the treatment of sarcoma. J Clin Oncol. 2004; 22:1706–1712. PMID: 15117993.9. Spiridonidis CH, Laufman LR, Jones J, Rhodes VA, Wallace K, Nicol S. Phase I study of docetaxel dose escalation in combination with fixed weekly gemcitabine in patients with advanced malignancies. J Clin Oncol. 1998; 16:3866–3873. PMID: 9850032.
Article10. Georgoulias V, Kouroussis C, Androulakis N, Kakolyris S, Dimopoulos MA, Papadakis E, et al. Front-line treatment of advanced non-small-cell lung cancer with docetaxel and gemcitabine: a multicenter phase II trial. J Clin Oncol. 1999; 17:914–920. PMID: 10071284.
Article11. Maki RG, Wathen JK, Patel SR, Priebat DA, Okuno SH, Samuels B, et al. Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002. J Clin Oncol. 2007; 25:2755–2763. PMID: 17602081.
Article12. Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, et al. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. New guidelines to evaluate the response to treatment in solid tumors. J Natl Cancer Inst. 2000; 92:205–216. PMID: 10655437.
Article13. Shewach DS, Hahn TM, Chang E, Hertel LW, Lawrence TS. Metabolism of 2',2'-difluoro-2'-deoxycytidine and radiation sensitization of human colon carcinoma cells. Cancer Res. 1994; 54:3218–3223. PMID: 8205542.14. Iwasaki H, Huang P, Keating MJ, Plunkett W. Differential incorporation of ara-C, gemcitabine, and fludarabine into replicating and repairing DNA in proliferating human leukemia cells. Blood. 1997; 90:270–278. PMID: 9207462.
Article15. Huang P, Chubb S, Hertel LW, Grindey GB, Plunkett W. Action of 2',2'-difluorodeoxycytidine on DNA synthesis. Cancer Res. 1991; 51:6110–6117. PMID: 1718594.16. Ross DD, Cuddy DP. Molecular effects of 2',2'-difluorodeoxycytidine (Gemcitabine) on DNA replication in intact HL-60 cells. Biochem Pharmacol. 1994; 48:1619–1630. PMID: 7980627.
Article17. Schiff PB, Fant J, Horwitz SB. Promotion of microtubule assembly in vitro by taxol. Nature. 1979; 277:665–667. PMID: 423966.
Article18. Rowinsky EK, Onetto N, Canetta RM, Arbuck SG. Taxol: the first of the taxanes, an important new class of antitumor agents. Semin Oncol. 1992; 19:646–662. PMID: 1361079.19. Smorenburg CH, Sparreboom A, Bontenbal M, Verweij J. Combination chemotherapy of the taxanes and antimetabolites: its use and limitations. Eur J Cancer. 2001; 37:2310–2323. PMID: 11720823.20. Bay JO, Ray-Coquard I, Fayette J, Leyvraz S, Cherix S, Piperno-Neumann S, et al. Docetaxel and gemcitabine combination in 133 advanced soft-tissue sarcomas: a retrospective analysis. Int J Cancer. 2006; 119:706–711. PMID: 16496406.
Article21. Aihara T, Kim Y, Takatsuka Y. Phase II study of weekly docetaxel in patients with metastatic breast cancer. Ann Oncol. 2002; 13:286–292. PMID: 11886007.
Article22. Schuette W, Nagel S, Blankenburg T, Lautenschlaeger C, Hans K, Schmidt EW, et al. Phase III study of second-line chemotherapy for advanced non-small-cell lung cancer with weekly compared with 3-weekly docetaxel. J Clin Oncol. 2005; 23:8389–8395. PMID: 16293869.
Article23. Baker SD, Zhao M, Lee CK, Verweij J, Zabelina Y, Brahmer JR, et al. Comparative pharmacokinetics of weekly and every-three-weeks docetaxel. Clin Cancer Res. 2004; 10:1976–1983. PMID: 15041715.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Gemcitabine and Docetaxel Combination for Advanced Soft Tissue Sarcoma: A Nationwide Retrospective Study
- Comparative Evaluation of Second-Line Chemotherapy Agents for Advanced Soft Tissue Sarcoma: Gemcitabine/Docetaxel, Pazopanib, and Alternatives
- Alveolar soft part Sarcoma with Metastasis to Bone: A Case Report
- Infantile Extraosseous Ewing's Sarcoma in the Left Arm: A Case Report
- Sonographic Features of Common Soft Tissue Masses in the Extremities