Allergy Asthma Immunol Res.
2011 Oct;3(4):226-235. 10.4168/aair.2011.3.4.226.
Chronic Rhinosinusitis: Therapeutic Efficacy of Anti-Inflammatory and Antibiotic Approaches
- Affiliations
-
- 1Department of Allergy and Medical Rhinology, Royal National Throat Nose Ear Hospital London, University College London, London, UK. g.scadding@ucl.ac.uk
- KMID: 2167073
- DOI: http://doi.org/10.4168/aair.2011.3.4.226
Abstract
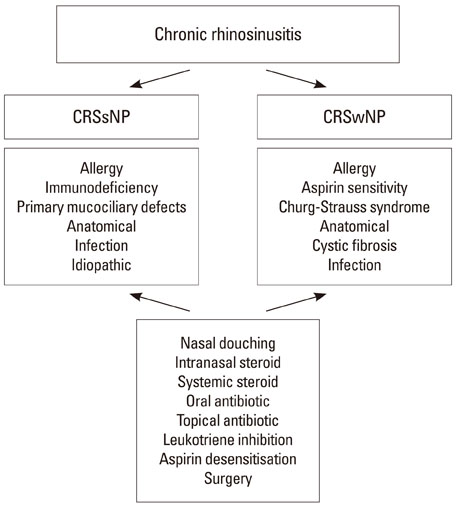
- Despite the high prevalence of chronic rhinosinusitis (CRS) worldwide, the exact pathogenesis of the disease remains unknown. Even with therapeutic intervention, treatment response is often only partial and frequently ineffective. The inability to define exact disease phenotypes in relation to specific disease mechanisms has led to a broad based approach with both anti-inflammatory and anti-microbial intervention. The clinical efficacy of such current therapeutic strategies is highlighted and the urgent need for further robust therapeutic intervention studies in CRS is discussed in this article.
MeSH Terms
Figure
Reference
-
1. Kaliner MA, Osguthorpe JD, Fireman P, Anon J, Georgitis J, Davis ML, Naclerio R, Kennedy D. Sinusitis: bench to bedside. Current findings, future directions. Otolaryngol Head Neck Surg. 1997. 116:S1–S20.2. Fokkens W, Lund V, Mullol J. European position paper on rhinosinusitis and nasal polyps 2007. Rhinol Suppl. 2007. 1–136.3. Meltzer EO, Hamilos DL, Hadley JA, Lanza DC, Marple BF, Nicklas RA, Bachert C, Baraniuk J, Baroody FM, Benninger MS, Brook I, Chowdhury BA, Druce HM, Durham S, Ferguson B, Gwaltney JM, Kaliner M, Kennedy DW, Lund V, Naclerio R, Pawankar R, Piccirillo JF, Rohane P, Simon R, Slavin RG, Togias A, Wald ER, Zinreich SJ. Rhinosinusitis: establishing definitions for clinical research and patient care. J Allergy Clin Immunol. 2004. 114:155–212.4. Min YG. The pathophysiology, diagnosis and treatment of allergic rhinitis. Allergy Asthma Immunol Res. 2010. 2:65–76.5. Van Zele T, Claeys S, Gevaert P, Van Maele G, Holtappels G, Van Cauwenberge P, Bachert C. Differentiation of chronic sinus diseases by measurement of inflammatory mediators. Allergy. 2006. 61:1280–1289.6. Van Bruaene N, Pérez-Novo CA, Basinski TM, Van Zele T, Holtappels G, De Ruyck N, Schmidt-Weber C, Akdis C, Van Cauwenberge P, Bachert C, Gevaert P. T-cell regulation in chronic paranasal sinus disease. J Allergy Clin Immunol. 2008. 121:1435–1441. 1441.e1–1441.e3.7. Van Bruaene N, Derycke L, Perez-Novo CA, Gevaert P, Holtappels G, De Ruyck N, Cuvelier C, Van Cauwenberge P, Bachert C. TGF-beta signaling and collagen deposition in chronic rhinosinusitis. J Allergy Clin Immunol. 2009. 124:253–259. 259.e1–259.e2.8. Reh DD, Wang Y, Ramanathan M Jr, Lane AP. Treatment-recalcitrant chronic rhinosinusitis with polyps is associated with altered epithelial cell expression of interleukin-33. Am J Rhinol Allergy. 2010. 24:105–109.9. Makihara S, Okano M, Fujiwara T, Kariya S, Noda Y, Higaki T, Nishizaki K. Regulation and characterization of IL-17A expression in patients with chronic rhinosinusitis and its relationship with eosinophilic inflammation. J Allergy Clin Immunol. 2010. 126:397–400. 400.e1–400.e11.10. Zhang N, Van Zele T, Perez-Novo C, Van Bruaene N, Holtappels G, DeRuyck N, Van Cauwenberge P, Bachert C. Different types of T-effector cells orchestrate mucosal inflammation in chronic sinus disease. J Allergy Clin Immunol. 2008. 122:961–968.11. Kariyawasam HH, Robinson DS. The role of eosinophils in airway tissue remodelling in asthma. Curr Opin Immunol. 2007. 19:681–686.12. Tieu DD, Peters AT, Carter RG, Suh L, Conley DB, Chandra R, Norton J, Grammer LC, Harris KE, Kato A, Kern RC, Schleimer RP. Evidence for diminished levels of epithelial psoriasin and calprotectin in chronic rhinosinusitis. J Allergy Clin Immunol. 2010. 125:667–675.13. Wang X, Moylan B, Leopold DA, Kim J, Rubenstein RC, Togias A, Proud D, Zeitlin PL, Cutting GR. Mutation in the gene responsible for cystic fibrosis and predisposition to chronic rhinosinusitis in the general population. JAMA. 2000. 284:1814–1819.14. Alqudah M, Graham SM, Ballas ZK. High prevalence of humoral immunodeficiency patients with refractory chronic rhinosinusitis. Am J Rhinol Allergy. 2010. 24:409–412.15. Vanlerberghe L, Joniau S, Jorissen M. The prevalence of humoral immunodeficiency in refractory rhinosinusitis: a retrospective analysis. B-ENT. 2006. 2:161–166.16. Lane AP, Truong-Tran QA, Schleimer RP. Altered expression of genes associated with innate immunity and inflammation in recalcitrant rhinosinusitis with polyps. Am J Rhinol. 2006. 20:138–144.17. Cormier C, Bossé Y, Mfuna L, Hudson TJ, Desrosiers M. Polymorphisms in the tumour necrosis factor alpha-induced protein 3 (TNFAIP3) gene are associated with chronic rhinosinusitis. J Otolaryngol Head Neck Surg. 2009. 38:133–141.18. Kilty SJ, Bossé Y, Cormier C, Endam LM, Desrosiers MY. Polymorphisms in the SERPINA1 (Alpha-1-Antitrypsin) gene are associated with severe chronic rhinosinusitis unresponsive to medical therapy. Am J Rhinol Allergy. 2010. 24:e4–e9.19. Cryer J, Schipor I, Perloff JR, Palmer JN. Evidence of bacterial biofilms in human chronic sinusitis. ORL J Otorhinolaryngol Relat Spec. 2004. 66:155–158.20. Bendouah Z, Barbeau J, Hamad WA, Desrosiers M. Biofilm formation by Staphylococcus aureus and Pseudomonas aeruginosa is associated with an unfavorable evolution after surgery for chronic sinusitis and nasal polyposis. Otolaryngol Head Neck Surg. 2006. 134:991–996.21. Psaltis AJ, Weitzel EK, Ha KR, Wormald PJ. The effect of bacterial biofilms on post-sinus surgical outcomes. Am J Rhinol. 2008. 22:1–6.22. Donlan RM, Costerton JW. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev. 2002. 15:167–193.23. Stewart PS, Costerton JW. Antibiotic resistance of bacteria in biofilms. Lancet. 2001. 358:135–138.24. Harvey R, Hannan SA, Badia L, Scadding G. Nasal saline irrigations for the symptoms of chronic rhinosinusitis. Cochrane Database Syst Rev. 2007. CD006394.25. Joe SA, Thambi R, Huang J. A systematic review of the use of intranasal steroids in the treatment of chronic rhinosinusitis. Otolaryngol Head Neck Surg. 2008. 139:340–347.26. Cuenant G, Stipon JP, Plante-Longchamp G, Baudoin C, Guerrier Y. Efficacy of endonasal neomycin-tixocortol pivalate irrigation in the treatment of chronic allergic and bacterial sinusitis. ORL J Otorhinolaryngol Relat Spec. 1986. 48:226–232.27. Sykes DA, Wilson R, Chan KL, Mackay IS, Cole PJ. Relative importance of antibiotic and improved clearance in topical treatment of chronic mucopurulent rhinosinusitis. A controlled study. Lancet. 1986. 2:359–360.28. Parikh A, Scadding GK, Darby Y, Baker RC. Topical corticosteroids in chronic rhinosinusitis: a randomized, double-blind, placebo-controlled trial using fluticasone propionate aqueous nasal spray. Rhinology. 2001. 39:75–79.29. Lavigne F, Cameron L, Renzi PM, Planet JF, Christodoulopoulos P, Lamkioued B, Hamid Q. Intrasinus administration of topical budesonide to allergic patients with chronic rhinosinusitis following surgery. Laryngoscope. 2002. 112:858–864.30. Lund VJ, Black JH, Szabó LZ, Schrewelius C, Akerlund A. Efficacy and tolerability of budesonide aqueous nasal spray in chronic rhinosinusitis patients. Rhinology. 2004. 42:57–62.31. Hansen FS, Djupesland PG, Fokkens WJ. Preliminary efficacy of fluticasone delivered by a novel device in recalcitrant chronic rhinosinusitis. Rhinology. 2010. 48:292–299.32. Mygind N, Pedersen CB, Prytz S, Sørensen H. Treatment of nasal polyps with intranasal beclomethasone dipropionate aerosol. Clin Allergy. 1975. 5:159–164.33. Tos M, Svendstrup F, Arndal H, Orntoft S, Jakobsen J, Borum P, Schrewelius C, Larsen PL, Clement F, Barfoed C, Rømeling F, Tvermosegaard T. Efficacy of an aqueous and a powder formulation of nasal budesonide compared in patients with nasal polyps. Am J Rhinol. 1998. 12:183–189.34. Lildholdt T, Rundcrantz H, Lindqvist N. Efficacy of topical corticosteroid powder for nasal polyps: a double-blind, placebo-controlled study of budesonide. Clin Otolaryngol Allied Sci. 1995. 20:26–30.35. Filiaci F, Passali D, Puxeddu R, Schrewelius C. A randomized controlled trial showing efficacy of once daily intranasal budesonide in nasal polyposis. Rhinology. 2000. 38:185–190.36. Jankowski R, Schrewelius C, Bonfils P, Saban Y, Gilain L, Prades JM, Strunski V. Efficacy and tolerability of budesonide aqueous nasal spray treatment in patients with nasal polyps. Arch Otolaryngol Head Neck Surg. 2001. 127:447–452.37. Small CB, Hernandez J, Reyes A, Schenkel E, Damiano A, Stryszak P, Staudinger H, Danzig M. Efficacy and safety of mometasone furoate nasal spray in nasal polyposis. J Allergy Clin Immunol. 2005. 116:1275–1281.38. Stjärne P, Blomgren K, Cayé-Thomasen P, Salo S, Søderstrøm T. The efficacy and safety of once-daily mometasone furoate nasal spray in nasal polyposis: a randomized, double-blind, placebo-controlled study. Acta Otolaryngol. 2006. 126:606–612.39. Ruhno J, Andersson B, Denburg J, Anderson M, Hitch D, Lapp P, Vanzieleghem M, Dolovich J. A double-blind comparison of intranasal budesonide with placebo for nasal polyposis. J Allergy Clin Immunol. 1990. 86:946–953.40. Vendelo Johansen L, Illum P, Kristensen S, Winther L, Vang Petersen S, Synnerstad B. The effect of budesonide (Rhinocort) in the treatment of small and medium-sized nasal polyps. Clin Otolaryngol Allied Sci. 1993. 18:524–527.41. Holmberg K, Juliusson S, Balder B, Smith DL, Richards DH, Karlsson G. Fluticasone propionate aqueous nasal spray in the treatment of nasal polyposis. Ann Allergy Asthma Immunol. 1997. 78:270–276.42. Penttilä M, Poulsen P, Hollingworth K, Holmström M. Dose-related efficacy and tolerability of fluticasone propionate nasal drops 400 microg once daily and twice daily in the treatment of bilateral nasal polyposis: a placebo-controlled randomized study in adult patients. Clin Exp Allergy. 2000. 30:94–102.43. Keith P, Nieminen J, Hollingworth K, Dolovich J. Efficacy and tolerability of fluticasone propionate nasal drops 400 microgram once daily compared with placebo for the treatment of bilateral polyposis in adults. Clin Exp Allergy. 2000. 30:1460–1468.44. Lund VJ, Flood J, Sykes AP, Richards DH. Effect of fluticasone in severe polyposis. Arch Otolaryngol Head Neck Surg. 1998. 124:513–518.45. Hardy JG, Lee SW, Wilson CG. Intranasal drug delivery by spray and drops. J Pharm Pharmacol. 1985. 37:294–297.46. Aukema AA, Mulder PG, Fokkens WJ. Treatment of nasal polyposis and chronic rhinosinusitis with fluticasone propionate nasal drops reduces need for sinus surgery. J Allergy Clin Immunol. 2005. 115:1017–1023.47. Holopainen E, Grahne B, Malmberg H, Mäkinien J, Lindqvist N. Budesonide in the treatment of nasal polyposis. Eur J Respir Dis Suppl. 1982. 122:221–228.48. Patiar S, Reece P. Oral steroids for nasal polyps. Cochrane Database Syst Rev. 2007. CD005232.49. Benítez P, Alobid I, de Haro J, Berenguer J, Bernal-Sprekelsen M, Pujols L, Picado C, Mullol J. A short course of oral prednisone followed by intranasal budesonide is an effective treatment of severe nasal polyps. Laryngoscope. 2006. 116:770–775.50. Hissaria P, Smith W, Wormald PJ, Taylor J, Vadas M, Gillis D, Kette F. Short course of systemic corticosteroids in sinonasal polyposis: a double-blind, randomized, placebo-controlled trial with evaluation of outcome measures. J Allergy Clin Immunol. 2006. 118:128–133.51. Ragab S, Parikh A, Darby YC, Scadding GK. An open audit of montelukast, a leukotriene receptor antagonist, in nasal polyposis associated with asthma. Clin Exp Allergy. 2001. 31:1385–1391.52. Kieff DA, Busaba NY. Efficacy of montelukast in the treatment of nasal polyposis. Ann Otol Rhinol Laryngol. 2005. 114:941–945.53. Dahlén B, Nizankowska E, Szczeklik A, Zetterström O, Bochenek G, Kumlin M, Mastalerz L, Pinis G, Swanson LJ, Boodhoo TI, Wright S, Dubé LM, Dahlén SE. Benefits from adding the 5-lipoxygenase inhibitor zileuton to conventional therapy in aspirin-intolerant asthmatics. Am J Respir Crit Care Med. 1998. 157:1187–1194.54. Parnes SM, Chuma AV. Acute effects of antileukotrienes on sinonasal polyposis and sinusitis. Ear Nose Throat J. 2000. 79:18–20. 24–25.55. Ulualp SO, Sterman BM, Toohill RJ. Antileukotriene therapy for the relief of sinus symptoms in aspirin triad disease. Ear Nose Throat J. 1999. 78:604–606. 608613passim.56. Mostafa BE, Abdel Hay H, Mohammed HE, Yamani M. Role of leukotriene inhibitors in the postoperative management of nasal polyps. ORL J Otorhinolaryngol Relat Spec. 2005. 67:148–153.57. Scadding GW, Scadding GK. Recent advances in antileukotriene therapy. Curr Opin Allergy Clin Immunol. 2010. 10:370–376.58. Parikh AA, Scadding GK. Intranasal lysine-aspirin in aspirin-sensitive nasal polyposis: a controlled trial. Laryngoscope. 2005. 115:1385–1390.59. Stevenson DD, Hankammer MA, Mathison DA, Christiansen SC, Simon RA. Aspirin desensitization treatment of aspirin-sensitive patients with rhinosinusitis-asthma: long-term outcomes. J Allergy Clin Immunol. 1996. 98:751–758.60. Stevenson DD. Aspirin desensitization in patients with AERD. Clin Rev Allergy Immunol. 2003. 24:159–168.61. Mardiney M, Borish L. Aspirin desensitization for chronic hyperplastic sinusitis, nasal polyposis, and asthma triad. Arch Otolaryngol Head Neck Surg. 2001. 127:1287.62. Nucera E, Schiavino D, Milani A, Del Ninno M, Misuraca C, Buonomo A, D'Ambrosio C, Paludetti G, Patriarca G. Effects of lysine-acetylsalicylate (LAS) treatment in nasal polyposis: two controlled long term prospective follow up studies. Thorax. 2000. 55:Suppl 2. S75–S78.63. Scadding GK, Hassab M, Darby YC, Lund VJ, Freedman A. Intranasal lysine aspirin in recurrent nasal polyposis. Clin Otolaryngol Allied Sci. 1995. 20:561–563.64. Ogata N, Darby Y, Scadding G. Intranasal lysine-aspirin administration decreases polyp volume in patients with aspirin-intolerant asthma. J Laryngol Otol. 2007. 121:1156–1160.65. Sousa AR, Parikh A, Scadding G, Corrigan CJ, Lee TH. Leukotriene-receptor expression on nasal mucosal inflammatory cells in aspirin-sensitive rhinosinusitis. N Engl J Med. 2002. 347:1493–1499.66. Sydnor TA Jr, Scheld WM, Gwaltney J Jr, Nielsen RW, Huck W, Therasse DG. Loracarbef (LY 163892) vs amoxicillin/clavulanate in bacterial maxillary sinusitis. Ear Nose Throat J. 1992. 71:225–232.67. Legent F, Bordure P, Beauvillain C, Berche P. A double-blind comparison of ciprofloxacin and amoxycillin/clavulanic acid in the treatment of chronic sinusitis. Chemotherapy. 1994. 40:Suppl 1. 8–15.68. Namyslowski G, Misiolek M, Czecior E, Malafiej E, Orecka B, Namyslowski P, Misiolek H. Comparison of the efficacy and tolerability of amoxycillin/clavulanic acid 875 mg b.i.d. with cefuroxime 500 mg b.i.d. in the treatment of chronic and acute exacerbation of chronic sinusitis in adults. J Chemother. 2002. 14:508–517.69. McNally PA, White MV, Kaliner MA. Sinusitis in an allergist's office: analysis of 200 consecutive cases. Allergy Asthma Proc. 1997. 18:169–175.70. Subramanian HN, Schechtman KB, Hamilos DL. A retrospective analysis of treatment outcomes and time to relapse after intensive medical treatment for chronic sinusitis. Am J Rhinol. 2002. 16:303–312.71. Hashiba M, Baba S. Efficacy of long-term administration of clarithromycin in the treatment of intractable chronic sinusitis. Acta Otolaryngol Suppl. 1996. 525:73–78.72. Ragab SM, Lund VJ, Scadding G. Evaluation of the medical and surgical treatment of chronic rhinosinusitis: a prospective, randomised, controlled trial. Laryngoscope. 2004. 114:923–930.73. Wallwork B, Coman W, Mackay-Sim A, Greiff L, Cervin A. A double-blind, randomized, placebo-controlled trial of macrolide in the treatment of chronic rhinosinusitis. Laryngoscope. 2006. 116:189–193.74. Wales D, Woodhead M. The anti-inflammatory effects of macrolides. Thorax. 1999. 54:Suppl 2. S58–S62.75. Tamaoki J. The effects of macrolides on inflammatory cells. Chest. 2004. 125:41S–50S.76. Cai Y, Chai D, Wang R, Bai N, Liang BB, Liu Y. Effectiveness and safety of macrolides in cystic fibrosis patients: a meta-analysis and systematic review. J Antimicrob Chemother. 2011. 66:968–978.77. Tamaoki J, Takeyama K, Yamawaki I, Kondo M, Konno K. Lipopolysaccharide-induced goblet cell hypersecretion in the guinea pig trachea: inhibition by macrolides. Am J Physiol. 1997. 272:L15–L19.78. Abdelghaffar H, Vazifeh D, Labro MT. Erythromycin A-derived macrolides modify the functional activities of human neutrophils by altering the phospholipase D-phosphatidate phosphohydrolase transduction pathway: L-cladinose is involved both in alterations of neutrophil functions and modulation of this transductional pathway. J Immunol. 1997. 159:3995–4005.79. Kikuchi T, Hagiwara K, Honda Y, Gomi K, Kobayashi T, Takahashi H, Tokue Y, Watanabe A, Nukiwa T. Clarithromycin suppresses lipopolysaccharide-induced interleukin-8 production by human monocytes through AP-1 and NF-kappa B transcription factors. J Antimicrob Chemother. 2002. 49:745–755.80. Desaki M, Takizawa H, Ohtoshi T, Kasama T, Kobayashi K, Sunazuka T, Omura S, Yamamoto K, Ito K. Erythromycin suppresses nuclear factor-kappaB and activator protein-1 activation in human bronchial epithelial cells. Biochem Biophys Res Commun. 2000. 267:124–128.81. Cervin A, Wallwork B, Mackay-Sim A, Coman WB, Greiff L. Effects of long-term clarithromycin treatment on lavage-fluid markers of inflammation in chronic rhinosinusitis. Clin Physiol Funct Imaging. 2009. 29:136–142.82. Oda H, Kadota J, Kohno S, Hara K. Leukotriene B4 in bronchoalveolar lavage fluid of patients with diffuse panbronchiolitis. Chest. 1995. 108:116–122.83. Kondoh K, Hashiba M, Baba S. Inhibitory activity of clarithromycin on biofilm synthesis with Pseudomonas aeruginosa. Acta Otolaryngol Suppl. 1996. 525:56–60.84. Chung AW, Yang HH, Radomski MW, van Breemen C. Long-term doxycycline is more effective than atenolol to prevent thoracic aortic aneurysm in marfan syndrome through the inhibition of matrix metalloproteinase-2 and -9. Circ Res. 2008. 102:e73–e85.85. Van Zele T, Gevaert P, Holtappels G, Beule A, Wormald PJ, Mayr S, Hens G, Hellings P, Ebbens FA, Fokkens W, Van Cauwenberge P, Bachert C. Oral steroids and doxycycline: two different approaches to treat nasal polyps. J Allergy Clin Immunol. 2010. 125:1069–1076.e4.86. Desrosiers M, Bendouah Z, Barbeau J. Effectiveness of topical antibiotics on Staphylococcus aureus biofilm in vitro. Am J Rhinol. 2007. 21:149–153.87. Ha KR, Psaltis AJ, Butcher AR, Wormald PJ, Tan LW. In vitro activity of mupirocin on clinical isolates of Staphylococcus aureus and its potential implications in chronic rhinosinusitis. Laryngoscope. 2008. 118:535–540.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Antibiotic Nebulizer Therapy for Treatment of Pediatric Chronic Rhinosinusitis
- Medical treatment according to phenotypes of chronic rhinosinusitis
- Endotypes of Asian chronic rhinosinusitis with nasal polyps: A narrative review
- Tissue Remodeling in Rhinosinusitis
- Medical Management of Chronic Rhinosinusitis