Healthc Inform Res.
2013 Sep;19(3):222-228. 10.4258/hir.2013.19.3.222.
Design and Realization of Integrated Management System for Data Interoperability between Point-of-Care Testing Equipment and Hospital Information System
- Affiliations
-
- 1Software Team of Diagnosis Business, i-SENS Inc., Seoul, Korea.
- 2Department of Computer Science, Kwangwoon University, Seoul, Korea. ygchoi@kw.ac.kr
- KMID: 2166701
- DOI: http://doi.org/10.4258/hir.2013.19.3.222
Abstract
OBJECTIVES
The purpose of this study was to design an integrated data management system based on the POCT1-A2, LIS2-A, LIS2-A2, and HL7 standard to ensure data interoperability between mobile equipment, such as point-of-care testing equipment and the existing hospital data system, its efficiency was also evaluated.
METHODS
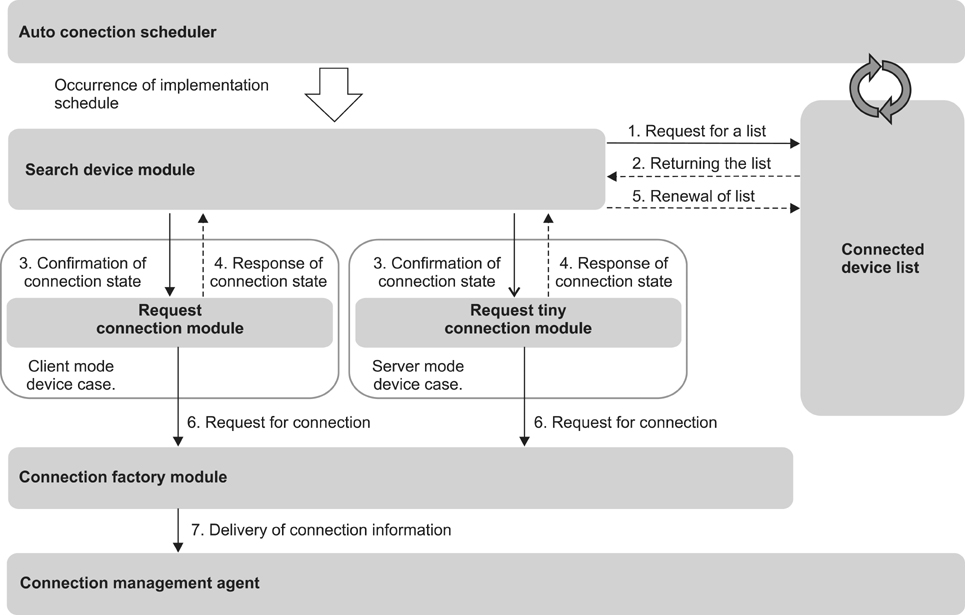
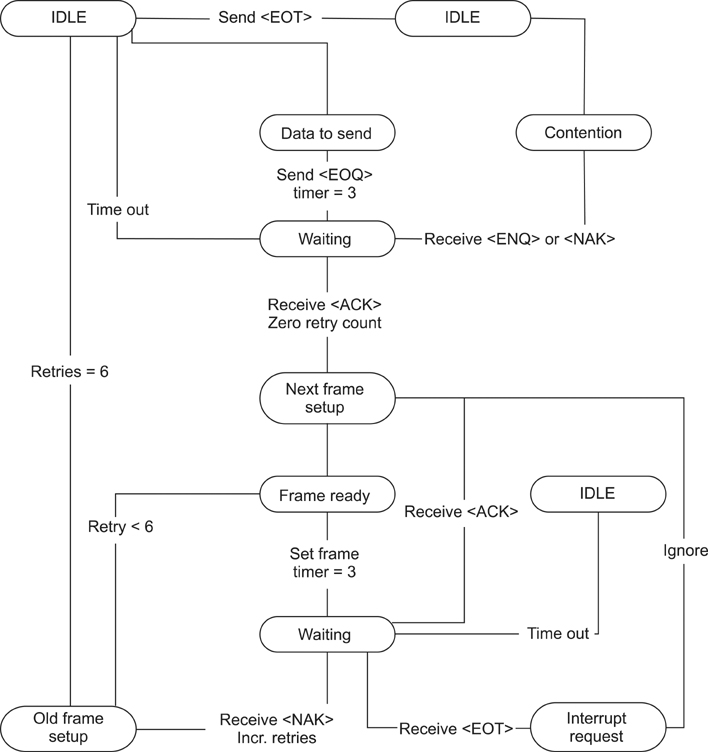
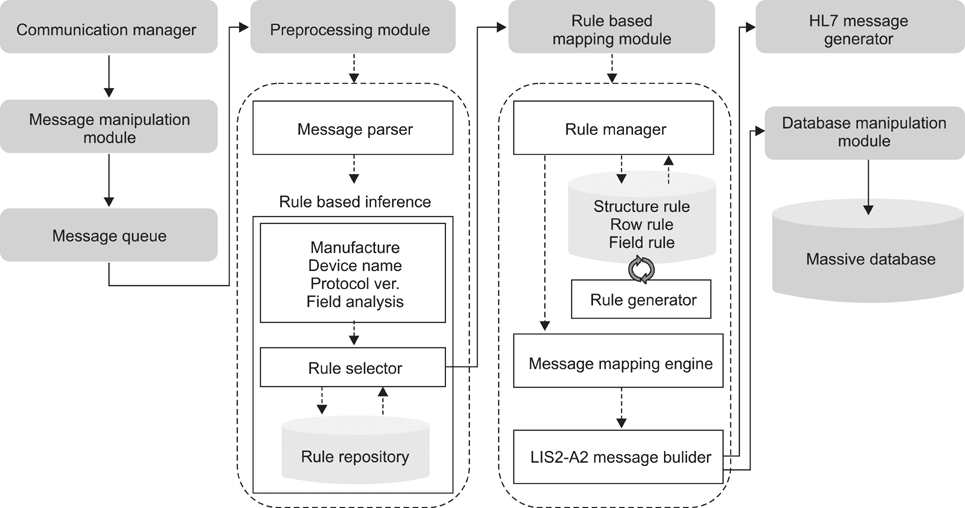
The method of this study was intended to design and realize a data management system which would provide a solution for the problems that occur when point-of-care testing equipment is introduced to existing hospital data, after classifying such problems into connectivity, integration, and interoperability. This study also checked if the data management system plays a sufficient role as a bridge between the point-of-care testing equipment and the hospital information system through connection persistence and reliability testing, as well as data integration and interoperability testing.
RESULTS
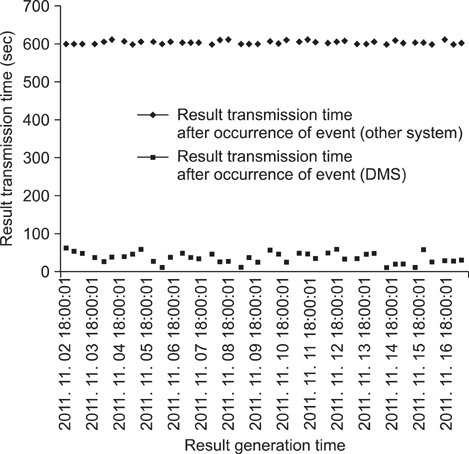
In comparison with the existing system, the data management system facilitated integration by improving the result receiving time, improving the collection rate, and by enabling the integration of disparate types of data into a single system. And it was found out that we can solve the problems related to connectivity, integration and interoperability through generating the message in standardized types.
CONCLUSIONS
It is expected that the proposed data management system, which is designed to improve the integration point-of-care testing equipment with existing systems, will establish a solid foundation on which better medical service may be provided by hospitals by improving the quality of patient service.
Keyword
MeSH Terms
Figure
Reference
-
1. Clinical and Laboratory Standards Institute. Point-of-care in vitro diagnostic (IVD) testing: approved guideline. 2nd ed. Wayne (PA): Clinical and Laboratory Standards Institute;2006.2. Nichols JH. Point of care testing. Clin Lab Med. 2007; 27(4):893–908.
Article3. Espicom Business Intelligence. Point-of-care diagnostics 2011: player, products and future market prospects. Chichester, UK: Espicom Business Intelligence;2011.4. Kost GJ, Ehrmeyer SS, Chernow B, Winkelman JW, Zaloga GP, Dellinger RP, et al. The laboratory-clinical interface: point-of-care testing. Chest. 1999; 115(4):1140–1154.5. Breil B, Fritz F, Thiemann V, Dugas M. Mapping turn-around times (TAT) to a generic timeline: a systematic review of TAT definitions in clinical domains. BMC Med Inform Decis Mak. 2011; 11:34.
Article6. Blick KE. The essential role of information management in point-of-care/critical care testing. Clin Chim Acta. 2001; 307(1-2):159–168.
Article7. Choe YJ. Point-of-care blood gas analyzers and point-of-care glucometers connectivity in tertiary care hospital [dissertation]. Seoul, Korea: Soonchunhyang University;2007.8. Kim DE. The effect of POCT on the second and third tier hospitals: a case study of successful POCT and implications for future direction [master's thesis]. Seoul, Korea: Yonsei University;2009.9. Clinical and Laboratory Standards Institute. Standard specification for low-level protocol to transfer messages between clinical laboratory instruments and computer system. Wayne (PA): Clinical and Laboratory Standards Institute;2003.10. Clinical and Laboratory Standards Institute. Specification for transferring information between clinical laboratory instruments and information systems: approved standard. 2nd ed. Wayne (PA): Clinical and Laboratory Standards Institute;2004.11. Markey B, Berry D. A quiet success story in the laboratory: survey of 30 implementations of the ASTM 1394 standard for analyser interfaces. In : Proceedings of the 15th Annual Conference of the Health Informatics Society of Ireland; 2010 Nov 17-18; Dublin, Ireland.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Study on The Integrated Interface Implementation for Medical Treatment Examination Equipment
- Factors Influencing on Operation Efficiency of Information Management System for Supply and Demand of Materials at Health Care: Case Study in General Hospitals of Daejeon City
- Construction And Evaluation Of A Point Of Care Computer System (Pccs) To Improve The Efficiency Of A Nursing Information System
- National Computerized Leprosy Information System
- Development of an Electronic Claim System Based on an Integrated Electronic Health Record Platform to Guarantee Interoperability