J Vet Sci.
2015 Mar;16(1):107-112. 10.4142/jvs.2015.16.1.107.
Ultrasonographic and macroscopic comparison of the thickness of the capsule, corium, and soft tissues in bovine claws: an in vitro study
- Affiliations
-
- 1Department of Surgery, Faculty of Veterinary Medicine, Uludag University, 16059 Bursa, Turkey. hsalci@uludag.edu.tr
- 2Department of Veterinary, Ihsangazi Vocational School of Higher Education, Kastamonu University, 37200 Kastamonu, Turkey.
- KMID: 2160827
- DOI: http://doi.org/10.4142/jvs.2015.16.1.107
Abstract
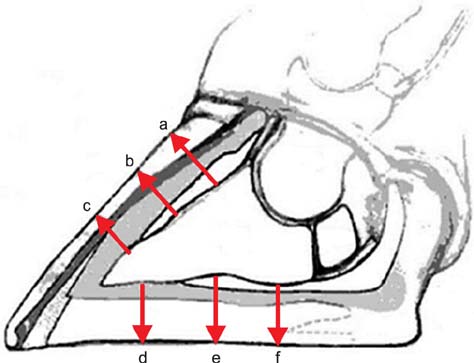
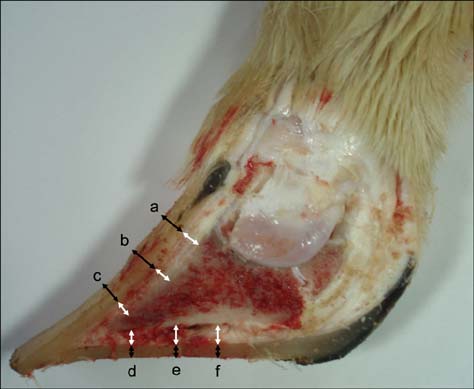
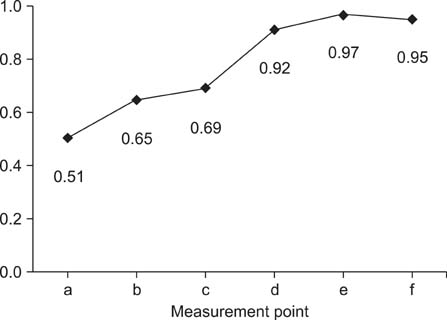
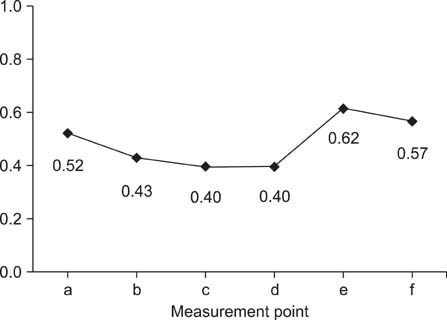
- This study aimed to compare thickness of the capsule, corium, and soft tissues measured ultrasonographically and macroscopically in selected regions of bovine claws. A hundred and twenty claws (n = 120) of 15 healthy Holstein bovines were obtained. After cleaning the claws, ultrasonographic measurement of the capsule, corium, and soft tissues was performed while submerging the claws in a water bath. Macroscopic measurements were taken after cutting of the claws axially. These values were compared statistically. According to the macroscopic measurements, the mean thickness +/- standard deviation (SD) of the capsule for dorsal wall and sole was 6.2 +/- 0.1 and 9.5 +/- 0.4 mm, respectively. The thickness of the corium and soft tissues for dorsal wall and sole was 4.5 +/- 0.1 and 5.3 +/- 0.1 mm, respectively. Ultrasonographically, the mean thickness +/- SD of the capsule for dorsal wall and sole was 4.7 +/- 0.1 and 7.8 +/- 0.3 mm, respectively. The thickness of the corium and soft tissues for dorsal wall and sole was 4.3 +/- 0.1 and 5.9 +/- 0.2 mm, respectively. Findings demonstrated that ultrasonography can be reliably to measure of the thickness of the hoof capsule, corium, and soft tissue in bovine claw.
Keyword
MeSH Terms
Figure
Reference
-
1. Browne MP, Hukins DWL, Skakle JMS, Knight CH, Hendry KAK, Wilde CJ, Galbraith H. X-ray diffraction patterns and anatomical properties of claw tissues of beef and dairy cattle. J Agric Sci. 2007; 145:623–633.
Article2. Bushberg JT, Seibert JA, Leidholdt EM Jr, Boone JM. The Essential Physics of Medical Imaging. 2nd ed. Philadelphia: Lippincott Williams & Wilkins;2002. p. 469–554.3. Dietz O, Prietz G. Quality and status of cattle hoof horn. Monatsh Veterinarmed. 1981; 36:419–422.4. Greenough PR, Vermunt JJ, McKinnon JJ, Fathy FA, Berg PA, Cohen RDH. Laminitis-like changes in the claws of feedlot cattle. Can Vet J. 1990; 31:202–208.5. Greenough PR. Bovine Laminitis and Lameness: a hands-on approach. Saunders/Elsevier: Toronto;2007. p. 176.6. Kofler J, Kübber P, Henninger W. Ultrasonographic imaging and thickness measurement of the sole horn and underlying soft tissue layer in bovine claws. Vet J. 1999; 157:322–331.
Article7. Nuss K, Paulus N. Measurements of claw dimensions in cows before and after functional trimming: a post-mortem study. Vet J. 2006; 172:284–292.
Article8. Nyland TG, Mattoon JS. Veterinary Diagnostic Ultrasound. Philadelphia: Saunders;1995. p. 3–17.9. Räber M, Lischer ChJ, Geyer H, Ossent P. The bovine digital cushion – a descriptive anatomical study. Vet J. 2004; 167:258–264.
Article10. Shakespeare AS. Inadequate thickness of the weight-bearing surface of claws in ruminants. J S Afr Vet Assoc. 2009; 80:247–253.
Article11. Shearer JK, van Amstel SR. Functional and corrective claw trimming. Vet Clin North Am Food Anim Pract. 2001; 17:53–72.
Article12. Simmins PH, Brooks PH. Supplementary biotin for sows: effect on claw integrity. Vet Rec. 1988; 122:431–435.
Article13. Singh SS, Murray RD, Ward WR. Histopathological and morphometric studies on the hooves of dairy and beef cattle in relation to overgrown sole and laminitis. J Comp Pathol. 1992; 107:319–328.
Article14. Toussaint Raven E, Haalstra RT, Peterse DJ. Cattle Footcare and Claw Trimming. Ipswich: Farming Press;1985. p. 75–106.15. van Amstel SR, Palin FL, Rorhbach BW, Shearer JK. Ultrasound measurement of sole horn thickness in trimmed claws of dairy cows. J Am Vet Med Assoc. 2003; 223:492–494.
Article16. van Amstel SR, Palin FL, Shearer JK. Measurement of the thickness of the corium and subcutaneous tissue of the hind claws of dairy cattle by ultrasound. Vet Rec. 2004; 155:630–633.
Article17. van Amstel SR, Shearer JK, Palin FL. Moisture content, thickness, and lesions of sole horn associated with thin soles in dairy cattle. J Dairy Sci. 2004; 87:757–763.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Bovine Serum Albumin on in-Vitro Growth and Development of Mouse Two-Cell Stage Embryos Fertilized in-Vivo
- Soft Tissue Masses: Ultrasonographic Findings
- Comparison of Skin Thickness in Lymphedema Using Ultrasonography and Skin Biopsy
- Ultrasonographic Measurement of the Thickness of Axillary Recess Capsule in Healthy Volunteers
- The Effect of Bovine Serum Albumin on in Vitro Growth and Development of Mouse Two-Cell Stage Embryos