Korean J Ophthalmol.
2016 Apr;30(2):101-107. 10.3341/kjo.2016.30.2.101.
Autologous Platelet-rich Plasma Eye Drops in the Treatment of Recurrent Corneal Erosions
- Affiliations
-
- 1Department of Ophthalmology, Kyungpook National University School of Medicine, Daegu, Korea. okeye@knu.ac.kr
- 2Cheil Eye Hospital, Daegu, Korea.
- KMID: 2160459
- DOI: http://doi.org/10.3341/kjo.2016.30.2.101
Abstract
- PURPOSE
To evaluate the effect of platelet-rich plasma (PRP) eye drops in the treatment of recurrent corneal erosions (RCE).
METHODS
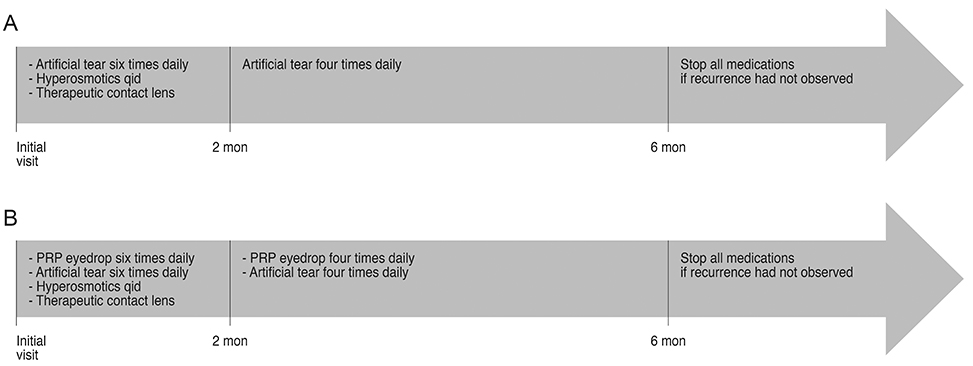
A total of 47 eyes were included in this retrospective study. Clinical records of 20 consecutive patients with RCE who had been treated with conventional lubricant eye drops (conventional treatment group) from June 2006 to December 2008 and 27 consecutive patients treated with autologous PRP eye drops in addition to lubricant eye drops (PRP eye drops treated group) from January 2009 to September 2014 were reviewed. Major and minor recurrences were recorded and compared between two groups.
RESULTS
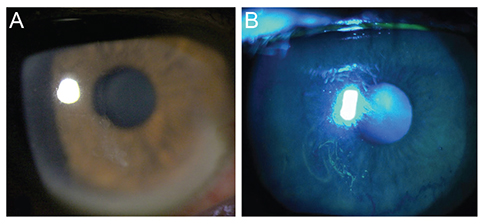
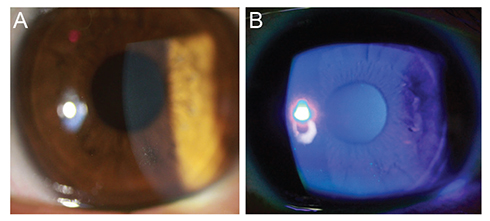
This study included 31 men and 16 women. The mean age was 44.5 ± 14.5 years (range, 19 to 86 years), and the mean follow-up duration was 14.9 ± 14.4 months (range, 6 to 64 months). Of the 27 cases in the PRP eye drops treated group, there were seven major recurrences in six eyes (22.2%) and ten minor recurrences in seven eyes (25.9%). In contrast, 16 eyes (80.0%) from the 20 patients in the conventional lubricant eye drops treated group had major recurrences, and all patients in this group reported minor recurrences. The mean frequency of recurrence was 0.06 ± 0.08 per month in the PRP eye drops treated group and 0.39 ± 0.24 per month in the conventional treatment group (p = 0.003). No side effects were noted in any of the patients over the follow-up period.
CONCLUSIONS
The use of PRP eye drops for the treatment of RCE was shown to be effective in reducing the recurrence rate without any significant complications.
Keyword
MeSH Terms
Figure
Reference
-
1. Dursun D, Kim MC, Solomon A, Pflugfelder SC. Treatment of recalcitrant recurrent corneal erosions with inhibitors of matrix metalloproteinase-9, doxycycline and corticosteroids. Am J Ophthalmol. 2001; 132:8–13.2. Hope-Ross MW, Chell PB, Kervick GN, et al. Oral tetracycline in the treatment of recurrent corneal erosions. Eye (Lond). 1994; 8(Pt 4):384–388.3. Buxton JN, Fox ML. Superficial epithelial keratectomy in the treatment of epithelial basement membrane dystrophy: a preliminary report. Arch Ophthalmol. 1983; 101:392–395.4. Soong HK, Farjo Q, Meyer RF, Sugar A. Diamond burr superficial keratectomy for recurrent corneal erosions. Br J Ophthalmol. 2002; 86:296–298.5. McLean EN, MacRae SM, Rich LF. Recurrent erosion: treatment by anterior stromal puncture. Ophthalmology. 1986; 93:784–788.6. Kim SY, Ko BY. Evaluation of anterior stromal puncture using Nd:YAG laser for refractory recurrent corneal erosion. J Korean Ophthalmol Soc. 2015; 56:331–338.7. Choi M, Jung JW, Seo KY, et al. Comparison of Nd:YAG laser versus conservative management in the treatment of recurrent corneal erosion. J Korean Ophthalmol Soc. 2015; 56:687–693.8. Forster W, Grewe S, Atzler U, et al. Phototherapeutic keratectomy in corneal diseases. Refract Corneal Surg. 1993; 9:2 Suppl. S85–S90.9. Nishida T, Nakamura M, Ofuji K, et al. Synergistic effects of substance P with insulin-like growth factor-1 on epithelial migration of the cornea. J Cell Physiol. 1996; 169:159–166.10. van Setten GB, Viinikka L, Tervo T, et al. Epidermal growth factor is a constant component of normal human tear fluid. Graefes Arch Clin Exp Ophthalmol. 1989; 227:184–187.11. Gupta A, Monroy D, Ji Z, et al. Transforming growth factor beta-1 and beta-2 in human tear fluid. Curr Eye Res. 1996; 15:605–614.12. Liu L, Hartwig D, Harloff S, et al. An optimised protocol for the production of autologous serum eyedrops. Graefes Arch Clin Exp Ophthalmol. 2005; 243:706–714.13. Tsubota K, Goto E, Fujita H, et al. Treatment of dry eye by autologous serum application in Sjogren's syndrome. Br J Ophthalmol. 1999; 83:390–395.14. Ogawa Y, Okamoto S, Mori T, et al. Autologous serum eye drops for the treatment of severe dry eye in patients with chronic graft-versus-host disease. Bone Marrow Transplant. 2003; 31:579–583.15. Noble BA, Loh RS, MacLennan S, et al. Comparison of autologous serum eye drops with conventional therapy in a randomised controlled crossover trial for ocular surface disease. Br J Ophthalmol. 2004; 88:647–652.16. Kojima T, Ishida R, Dogru M, et al. The effect of autologous serum eyedrops in the treatment of severe dry eye disease: a prospective randomized case-control study. Am J Ophthalmol. 2005; 139:242–246.17. Tananuvat N, Daniell M, Sullivan LJ, et al. Controlled study of the use of autologous serum in dry eye patients. Cornea. 2001; 20:802–806.18. Tsubota K, Goto E, Shimmura S, Shimazaki J. Treatment of persistent corneal epithelial defect by autologous serum application. Ophthalmology. 1999; 106:1984–1989.19. Poon AC, Geerling G, Dart JK, et al. Autologous serum eyedrops for dry eyes and epithelial defects: clinical and in vitro toxicity studies. Br J Ophthalmol. 2001; 85:1188–1197.20. Young AL, Cheng AC, Ng HK, et al. The use of autologous serum tears in persistent corneal epithelial defects. Eye (Lond). 2004; 18:609–614.21. Jeng BH, Dupps WJ Jr. Autologous serum 50% eyedrops in the treatment of persistent corneal epithelial defects. Cornea. 2009; 28:1104–1108.22. Matsumoto Y, Dogru M, Goto E, et al. Autologous serum application in the treatment of neurotrophic keratopathy. Ophthalmology. 2004; 111:1115–1120.23. del Castillo JM, de la Casa JM, Sardina RC, et al. Treatment of recurrent corneal erosions using autologous serum. Cornea. 2002; 21:781–783.24. Knighton DR, Ciresi K, Fiegel VD, et al. Stimulation of repair in chronic, nonhealing, cutaneous ulcers using platelet-derived wound healing formula. Surg Gynecol Obstet. 1990; 170:56–60.25. Lynch SE, Colvin RB, Antoniades HN. Growth factors in wound healing: single and synergistic effects on partial thickness porcine skin wounds. J Clin Invest. 1989; 84:640–646.26. Alio JL, Abad M, Artola A, et al. Use of autologous platelet-rich plasma in the treatment of dormant corneal ulcers. Ophthalmology. 2007; 114:1286–1293.e1.27. Alio JL, Pastor S, Ruiz-Colecha J, et al. Treatment of ocular surface syndrome after LASIK with autologous platelet-rich plasma. J Refract Surg. 2007; 23:617–619.28. Kim KM, Shin YT, Kim HK. Effect of autologous platelet-rich plasma on persistent corneal epithelial defect after infectious keratitis. Jpn J Ophthalmol. 2012; 56:544–550.29. Marx RE, Carlson ER, Eichstaedt RM, et al. Platelet-rich plasma: growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998; 85:638–646.30. Peng Y, Huang S, Wu Y, et al. Platelet rich plasma clot releasate preconditioning induced PI3K/AKT/NFκB signaling enhances survival and regenerative function of rat bone marrow mesenchymal stem cells in hostile microenvironments. Stem Cells Dev. 2013; 22:3236–3251.31. Holzer MP, Auffarth GU, Specht H, Kruse FE. Combination of transepithelial phototherapeutic keratectomy and autologous serum eyedrops for treatment of recurrent corneal erosions. J Cataract Refract Surg. 2005; 31:1603–1606.32. Hartwig D, Harloff S, Liu L, et al. Epitheliotrophic capacity of a growth factor preparation produced from platelet concentrates on corneal epithelial cells: a potential agent for the treatment of ocular surface defects? Transfusion. 2004; 44:1724–1731.33. Sakimoto T, Shoji J, Yamada A, Sawa M. Upregulation of matrix metalloproteinase in tear fluid of patients with recurrent corneal erosion. Jpn J Ophthalmol. 2007; 51:343–346.34. Nurden AT. Platelets, inflammation and tissue regeneration. Thromb Haemost. 2011; 105:Suppl 1. S13–S33.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Therapeutic Effect of Topical Autologous Serum in Recurrent Punctate Corneal Erosion
- Effect of Platelet-rich Plasma on Burn Wounds according to Time of Application: An Experimental Study on Rats
- Platelet-rich plasma treatment in patients with refractory thin endometrium and recurrent implantation failure: A comprehensive review
- Effects of Autologous Platelet-Rich Plasma on Postoperative Blood Loss and Transfusion Requirements in Cardiac Surgery
- Recalcitrant Cutaneous Ulcer of Comorbid Patient Treated with Platelet Rich Plasma: A Case Report