Korean J Radiol.
2015 Aug;16(4):783-790. 10.3348/kjr.2015.16.4.783.
Imaging Features of Primary Tumors and Metastatic Patterns of the Extraskeletal Ewing Sarcoma Family of Tumors in Adults: A 17-Year Experience at a Single Institution
- Affiliations
-
- 1Department of Radiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul 138-736, Korea. medimash@gmail.com
- 2Department of Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul 138-736, Korea.
- 3Department of Imaging, Dana-Farber Cancer Institute, Brigham and Women's Hospital, Harvard Medical School, Boston, MA 02115, USA.
- KMID: 2155551
- DOI: http://doi.org/10.3348/kjr.2015.16.4.783
Abstract
OBJECTIVE
To comprehensively analyze the spectrum of imaging features of the primary tumors and metastatic patterns of the Extraskeletal Ewing sarcoma family of tumors (EES) in adults.
MATERIALS AND METHODS
We performed a computerized search of our hospital's data-warehouse from 1996 to 2013 using codes for Ewing sarcoma and primitive neuroectodermal tumors as well as the demographic code for > or = 18 years of age. We selected subjects who were histologically confirmed to have Ewing sarcoma of extraskeletal origin. Imaging features of the primary tumor and metastatic disease were evaluated for lesion location, size, enhancement pattern, necrosis, margin, and invasion of adjacent organs.
RESULTS
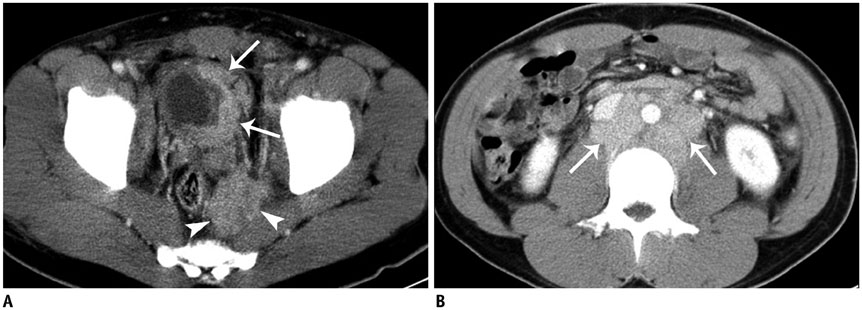
Among the 70 patients (mean age, 35.8 +/- 15.6 years; range, 18-67 years) included in our study, primary tumors of EES occurred in the soft tissue and extremities (n = 20), abdomen and pelvis (n = 18), thorax (n = 14), paravertebral space (n = 8), head and neck (n = 6), and an unknown primary site (n = 4). Most primary tumors manifested as large and bulky soft-tissue masses (mean size, 9.0 cm; range, 1.3-23.0 cm), frequently invading adjacent organs (45.6%) and showed heterogeneous enhancement (73.7%), a well-defined (66.7%) margin, and partial necrosis/cystic degeneration (81.9%). Notably, 29 patients had metastatic disease detected at their initial diagnosis. The most frequent site of metastasis was lymph nodes (75.9%), followed by bone (31.0%), lung (20.7%), abdominal solid organs (13.8%), peritoneum (13.8%), pleura (6.9%), and brain (3.4%).
CONCLUSION
Primary tumors of EES can occur anywhere and mostly manifest as large and bulky, soft-tissue masses. Lymph nodes are the most frequent metastasis sites.
MeSH Terms
-
Adolescent
Adult
Aged
Asian Continental Ancestry Group
Bone Neoplasms/*pathology/radiography
Brain Neoplasms/pathology/radiography
Female
Humans
Lymph Nodes/pathology
Magnetic Resonance Imaging
Male
Middle Aged
Neoplasm Metastasis
Neuroectodermal Tumors, Primitive/pathology/radiography
Positron-Emission Tomography
Sarcoma, Ewing/*pathology/radiography
Tomography, X-Ray Computed
Young Adult
Figure
Reference
-
1. Cote GM, Choy E. Update in treatment and targets in Ewing sarcoma. Hematol Oncol Clin North Am. 2013; 27:1007–1019.2. Jain S, Xu R, Prieto VG, Lee P. Molecular classification of soft tissue sarcomas and its clinical applications. Int J Clin Exp Pathol. 2010; 3:416–428.3. Murphey MD, Senchak LT, Mambalam PK, Logie CI, Klassen-Fischer MK, Kransdorf MJ. From the radiologic pathology archives: ewing sarcoma family of tumors: radiologic-pathologic correlation. Radiographics. 2013; 33:803–831.4. Javery O, Krajewski K, O'Regan K, Kis B, Giardino A, Jagannathan J, et al. A to Z of extraskeletal Ewing sarcoma family of tumors in adults: imaging features of primary disease, metastatic patterns, and treatment responses. AJR Am J Roentgenol. 2011; 197:W1015–W1022.5. Krasin MJ, Davidoff AM, Rodriguez-Galindo C, Billups CA, Fuller CE, Neel MD, et al. Definitive surgery and multiagent systemic therapy for patients with localized Ewing sarcoma family of tumors: local outcome and prognostic factors. Cancer. 2005; 104:367–373.6. Womer RB, West DC, Krailo MD, Dickman PS, Pawel BR, Grier HE, et al. Randomized controlled trial of interval-compressed chemotherapy for the treatment of localized Ewing sarcoma: a report from the Children's Oncology Group. J Clin Oncol. 2012; 30:4148–4154.7. von Mehren M, Randall RL, Benjamin RS, Boles S, Bui MM, Casper ES, et al. Soft tissue sarcoma, version 2.2014. J Natl Compr Canc Netw. 2014; 12:473–483.8. Paulussen M, Bielack S, Jürgens H, Casali PG. ESMO Guidelines Working Group. Ewing's sarcoma of the bone: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2009; 20:Suppl 4. 140–142.9. Somarouthu BS, Shinagare AB, Rosenthal MH, Tirumani H, Hornick JL, Ramaiya NH, et al. Multimodality imaging features, metastatic pattern and clinical outcome in adult extraskeletal Ewing sarcoma: experience in 26 patients. Br J Radiol. 2014; 87:20140123.10. Jawad MU, Cheung MC, Min ES, Schneiderbauer MM, Koniaris LG, Scully SP. Ewing sarcoma demonstrates racial disparities in incidence-related and sex-related differences in outcome: an analysis of 1631 cases from the SEER database, 1973-2005. Cancer. 2009; 115:3526–3536.11. Kumar S, Pack S, Kumar D, Walker R, Quezado M, Zhuang Z, et al. Detection of EWS-FLI-1 fusion in Ewing's sarcoma/peripheral primitive neuroectodermal tumor by fluorescence in situ hybridization using formalin-fixed paraffin-embedded tissue. Hum Pathol. 1999; 30:324–330.12. Bernstein M, Kovar H, Paulussen M, Randall RL, Schuck A, Teot LA, et al. Ewing's sarcoma family of tumors: current management. Oncologist. 2006; 11:503–519.13. El Weshi A, Allam A, Ajarim D, Al Dayel F, Pant R, Bazarbashi S, et al. Extraskeletal Ewing's sarcoma family of tumours in adults: analysis of 57 patients from a single institution. Clin Oncol (R Coll Radiol). 2010; 22:374–381.14. Applebaum MA, Goldsby R, Neuhaus J, DuBois SG. Clinical features and outcomes in patients with Ewing sarcoma and regional lymph node involvement. Pediatr Blood Cancer. 2012; 59:617–620.15. Lee JA, Kim DH, Cho J, Lim JS, Koh JS, Yoo JY, et al. Treatment outcome of Korean patients with localized Ewing sarcoma family of tumors: a single institution experience. Jpn J Clin Oncol. 2011; 41:776–782.