Clin Exp Otorhinolaryngol.
2012 Sep;5(3):150-155. 10.3342/ceo.2012.5.3.150.
Relationships between Lymph Node Metastasis and Expression of CD31, D2-40, and Vascular Endothelial Growth Factors A and C in Papillary Thyroid Cancer
- Affiliations
-
- 1Department of Otorhinolaryngology-Head and Neck Surgery, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea. lnhkk@hanmail.net
- 2Department of Pathology, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 3Department of Dermatology, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea.
- KMID: 2134598
- DOI: http://doi.org/10.3342/ceo.2012.5.3.150
Abstract
OBJECTIVES
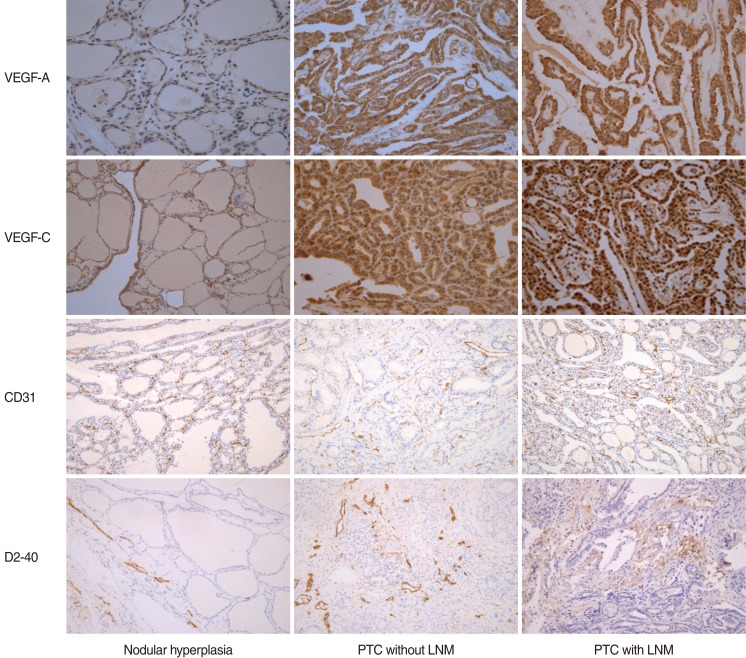
To investigate the relationships between lymph node metastasis (LNM) and expression of CD31, D2-40 and vascular endothelial growth factors (VEGF)-A and -C in patients with papillary thyroid cancer (PTC).
METHODS
Paraffin-embedded thyroid tissues of 72 patients were evaluated, which included 25 patients with thyroid nodular hyperplasia (TNH), 24 PTC patients without LNM, and 23 PTC patients with LNM. Three pathologists, who were blinded to the patient's clinical information, assessed the immunohistochemical staining results. The amount of expression was scored as high (>25% of cells stained) or low (0-25%).
RESULTS
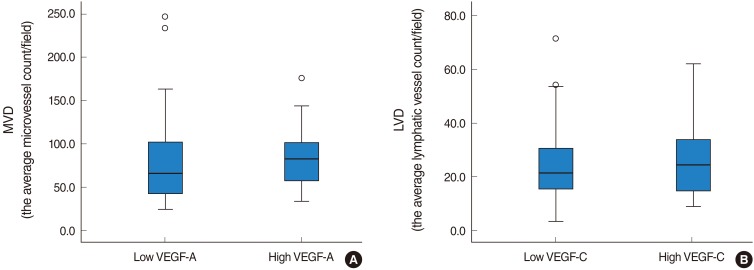
A higher level of VEGF-A expression was observed in the PTC groups regardless of LNM when compared to the group with TNH (91.3%, 79.2%, 4.0%, respectively). VEGF-C expression in the PTC with LNM group was significantly higher than the other two groups (P<0.05). No difference in microvessel density (MVD) scores was observed using CD31 among the three groups. The lymphatic vessel density (LVD) score using D2-40 was significantly higher in patients having PTC with LNM than the other groups (P<0.05).
CONCLUSION
VEGF-C and D2-40 were more highly expressed in patients having PTC with LNM than in patients having PTC without LNM or in those having TNH. Analysis of VEGF-C level and LVD using D2-40 may be helpful in the diagnosis of PTC and the evaluation of LNM potential in patients with PTC.
Keyword
MeSH Terms
-
Carcinoma, Papillary
Factor IX
Glycosaminoglycans
Humans
Hyperplasia
Lymph Nodes
Lymphatic Vessels
Microvessels
Neoplasm Metastasis
Thyroid Gland
Thyroid Neoplasms
Vascular Endothelial Growth Factor A
Vascular Endothelial Growth Factor C
Vascular Endothelial Growth Factors
Factor IX
Glycosaminoglycans
Vascular Endothelial Growth Factor A
Vascular Endothelial Growth Factor C
Vascular Endothelial Growth Factors
Figure
Cited by 2 articles
-
Effectiveness of an i-PTH Measurement in Predicting Post Thyroidectomy Hypocalcemia: Prospective Controlled Study
Jin Pyeong Kim, Jung Je Park, Hee Young Son, Rock Bum Kim, Ho Youp Kim, Seung Hoon Woo
Yonsei Med J. 2013;54(3):637-642. doi: 10.3349/ymj.2013.54.3.637.The Cancer Genome Atlas Validation of Ancillary Tests for Classifying Papillary Thyroid Carcinoma
Yong Joon Suh, Hyoun Jong Moon, Ji-Young Choe, Hyo Jin Park
Int J Thyroidol. 2017;10(1):24-35. doi: 10.11106/ijt.2017.10.1.24.
Reference
-
1. Tian X, Cong M, Zhou W, Zhu J, Liu Q. Relationship between protein expression of VEGF-C, MMP-2 and lymph node metastasis in papillary thyroid cancer. J Int Med Res. 2008; Jul-Aug. 36(4):699–703. PMID: 18652765.
Article2. Noguchi S, Murakami N, Yamashita H, Toda M, Kawamoto H. Papillary thyroid carcinoma: modified radical neck dissection improves prognosis. Arch Surg. 1998; 3. 133(3):276–280. PMID: 9517740.3. Jebreel A, England J, Bedford K, Murphy J, Karsai L, Atkin S. Vascular endothelial growth factor (VEGF), VEGF receptors expression and microvascular density in benign and malignant thyroid diseases. Int J Exp Pathol. 2007; 8. 88(4):271–277. PMID: 17696908.
Article4. Ferrara N, Houck K, Jakeman L, Leung DW. Molecular and biological properties of the vascular endothelial growth factor family of proteins. Endocr Rev. 1992; 2. 13(1):18–32. PMID: 1372863.
Article5. Lohela M, Helotera H, Haiko P, Dumont DJ, Alitalo K. Transgenic induction of vascular endothelial growth factor-C is strongly angiogenic in mouse embryos but leads to persistent lymphatic hyperplasia in adult tissues. Am J Pathol. 2008; 12. 173(6):1891–1901. PMID: 18988807.
Article6. Wang SL, Li SH, Chen WT, Chai CY. Expression of D2-40 in adjunct diagnosis of papillary thyroid carcinoma. APMIS. 2007; 8. 115(8):906–910. PMID: 17696946.
Article7. DeLisser HM, Christofidou-Solomidou M, Strieter RM, Burdick MD, Robinson CS, Wexler RS, et al. Involvement of endothelial PECAM-1/CD31 in angiogenesis. Am J Pathol. 1997; 9. 151(3):671–677. PMID: 9284815.8. Lennard CM, Patel A, Wilson J, Reinhardt B, Tuman C, Fenton C, et al. Intensity of vascular endothelial growth factor expression is associated with increased risk of recurrence and decreased disease-free survival in papillary thyroid cancer. Surgery. 2001; 5. 129(5):552–558. PMID: 11331447.
Article9. de la Torre NG, Buley I, Wass JA, Turner HE. Angiogenesis and lymphangiogenesis in thyroid proliferative lesions: relationship to type and tumour behaviour. Endocr Relat Cancer. 2006; 9. 13(3):931–944. PMID: 16954441.
Article10. Chung MK, Kim JH, Ko YH, Son YI. Correlation of lymphatic vessel density and vascular endothelial growth factor with nodal metastasis in papillary thyroid microcarcinoma. Head Neck. 2011; 8. 18. [Epub]. http://dx.doi.org/10.1002/hed.21822.
Article11. Bono P, Wasenius VM, Heikkila P, Lundin J, Jackson DG, Joensuu H. High LYVE-1-positive lymphatic vessel numbers are associated with poor outcome in breast cancer. Clin Cancer Res. 2004; 11. 10(21):7144–7149. PMID: 15534085.
Article12. Fox SB. Tumour angiogenesis and prognosis. Histopathology. 1997; 3. 30(3):294–301. PMID: 9088964.
Article13. Pepper MS. Lymphangiogenesis and tumor metastasis: myth or reality? Clin Cancer Res. 2001; 3. 7(3):462–468. PMID: 11297234.14. Mohammed RA, Green A, El-Shikh S, Paish EC, Ellis IO, Martin SG. Prognostic significance of vascular endothelial cell growth factors -A, -C and -D in breast cancer and their relationship with angio- and lymphangiogenesis. Br J Cancer. 2007; 4. 96(7):1092–1100. PMID: 17353919.
Article15. Sahni D, Robson A, Orchard G, Szydlo R, Evans AV, Russell-Jones R. The use of LYVE-1 antibody for detecting lymphatic involvement in patients with malignant melanoma of known sentinel node status. J Clin Pathol. 2005; 7. 58(7):715–721. PMID: 15976338.
Article16. Ramsden JD. Angiogenesis in the thyroid gland. J Endocrinol. 2000; 9. 166(3):475–480. PMID: 11029748.
Article17. Turner HE, Harris AL, Melmed S, Wass JA. Angiogenesis in endocrine tumors. Endocr Rev. 2003; 10. 24(5):600–632. PMID: 14570746.
Article18. Bunone G, Vigneri P, Mariani L, Buto S, Collini P, Pilotti S, et al. Expression of angiogenesis stimulators and inhibitors in human thyroid tumors and correlation with clinical pathological features. Am J Pathol. 1999; 12. 155(6):1967–1976. PMID: 10595926.
Article19. Kilicarslan AB, Ogus M, Arici C, Pestereli HE, Cakir M, Karpuzoglu G. Clinical importance of vascular endothelial growth factor (VEGF) for papillary thyroid carcinomas. APMIS. 2003; 3. 111(3):439–443. PMID: 12752224.
Article20. Herrmann G, Schumm-Draeger PM, Muller C, Atai E, Wenzel B, Fabian T, et al. T lymphocytes, CD68-positive cells and vascularisation in thyroid carcinomas. J Cancer Res Clin Oncol. 1994; 11. 120(11):651–656. PMID: 7525593.
Article21. Akslen LA, Livolsi VA. Increased angiogenesis in papillary thyroid carcinoma but lack of prognostic importance. Hum Pathol. 2000; 4. 31(4):439–442. PMID: 10821490.22. Tanaka K, Sonoo H, Kurebayashi J, Nomura T, Ohkubo S, Yamamoto Y, et al. Inhibition of infiltration and angiogenesis by thrombospondin-1 in papillary thyroid carcinoma. Clin Cancer Res. 2002; 5. 8(5):1125–1131. PMID: 12006528.23. Fontanini G, Vignati S, Pacini F, Pollina L, Basolo F. Microvessel count: an indicator of poor outcome in medullary thyroid carcinoma but not in other types of thyroid carcinoma. Mod Pathol. 1996; 6. 9(6):636–641. PMID: 8782200.24. Dadras SS, Paul T, Bertoncini J, Brown LF, Muzikansky A, Jackson DG, et al. Tumor lymphangiogenesis: a novel prognostic indicator for cutaneous melanoma metastasis and survival. Am J Pathol. 2003; 6. 162(6):1951–1960. PMID: 12759251.25. Franchi A, Gallo O, Massi D, Baroni G, Santucci M. Tumor lymphangiogenesis in head and neck squamous cell carcinoma: a morphometric study with clinical correlations. Cancer. 2004; 9. 101(5):973–978. PMID: 15329906.26. Birner P, Obermair A, Schindl M, Kowalski H, Breitenecker G, Oberhuber G. Selective immunohistochemical staining of blood and lymphatic vessels reveals independent prognostic influence of blood and lymphatic vessel invasion in early-stage cervical cancer. Clin Cancer Res. 2001; 1. 7(1):93–97. PMID: 11205924.27. Dhar DK, Kubota H, Kotoh T, Tabara H, Watanabe R, Tachibana M, et al. Tumor vascularity predicts recurrence in differentiated thyroid carcinoma. Am J Surg. 1998; 11. 176(5):442–447. PMID: 9874430.
Article28. He Y, Karpanen T, Alitalo K. Role of lymphangiogenic factors in tumor metastasis. Biochim Biophys Acta. 2004; 3. 1654(1):3–12. PMID: 14984763.
Article29. Mandriota SJ, Jussila L, Jeltsch M, Compagni A, Baetens D, Prevo R, et al. Vascular endothelial growth factor-C-mediated lymphangiogenesis promotes tumour metastasis. EMBO J. 2001; 2. 20(4):672–682. PMID: 11179212.
Article30. Beasley NJ, Prevo R, Banerji S, Leek RD, Moore J, van Trappen P, et al. Intratumoral lymphangiogenesis and lymph node metastasis in head and neck cancer. Cancer Res. 2002; 3. 62(5):1315–1320. PMID: 11888898.31. Siironen P, Ristimaki A, Narko K, Nordling S, Louhimo J, Andersson S, et al. VEGF-C and COX-2 expression in papillary thyroid cancer. Endocr Relat Cancer. 2006; 6. 13(2):465–473. PMID: 16728574.
Article32. Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z. Vascular endothelial growth factor (VEGF) and its receptors. FASEB J. 1999; 1. 13(1):9–22. PMID: 9872925.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of Vascular Endothelial Growth Factor (VEGF) in Papillary Thyroid Carcinoma
- Angiogenic and Lymphangiogenic Microvessel Density in Terms of the Expression of CD31 and D2-40 in Colon Cancer
- D2-40, Podoplanin, and CD31 as a Prognostic Predictor in Invasive Ductal Carcinomas of the Breast
- Correlation between Tumor Markers and Clinicopathologic Factors in Peripheral Blood of Papillary Thyroid Cancer Patients
- Correlations of the Aggressiveness of Thyroid Cancer with Microvessel Density, Expression of Vascular Endothelial Growth Factor and Its Receptors