Clin Exp Otorhinolaryngol.
2012 Sep;5(3):139-144. 10.3342/ceo.2012.5.3.139.
In Vitro Streptococcus pneumoniae Biofilm Formation and In Vivo Middle Ear Mucosal Biofilm in a Rat Model of Acute Otitis Induced by S. pneumoniae
- Affiliations
-
- 1Department of Otorhinolaryngology-Head and Neck Surgery, Dongguk University Ilsan Hospital, Goyang, Korea. jjsong23@gmail.com
- 2Department of Otorhinolaryngology-Head and Neck Surgery, Korea University College of Medicine, Seoul, Korea.
- KMID: 2134596
- DOI: http://doi.org/10.3342/ceo.2012.5.3.139
Abstract
OBJECTIVES
Streptococcus pneumoniae is one of the most common pathogens of otitis media (OM) that exists in biofilm, which enhances the resistance of bacteria against antibiotic killing and diagnosis, compared to the free-floating (planktonic) form. This study evaluated biofilm formation by S. pneumoniae on an abiotic surface and in the middle ear cavity in a rat model of OM.
METHODS
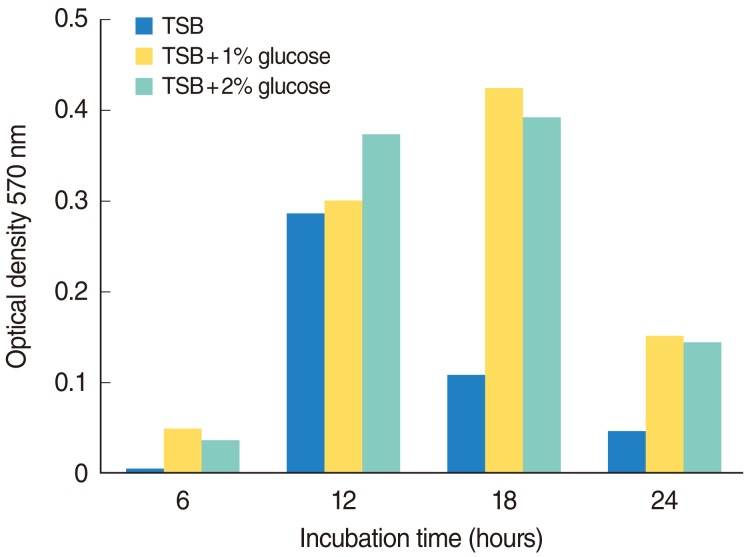
In vitro biofilm formation was evaluated by inoculation of a 1:100 diluted S. pneumoniae cell suspension in a 96-well microplate. Adherent cells were quantified spectrophotometrically following staining with crystal violet by measurement of optical density at 570 nm. The ultrastructure of pneumococcal biofilm was assessed by scanning electron microscopy (SEM). For in vitro biofilm study, S. pneumoniae cell suspensions containing 1x10(7) colony forming units were injected through transtympanic membrane into the middle ear cavity of Sprague Dawley rats. The ultrastructure of middle ear mucus was observed by SEM 1 and 2 weeks post-inoculation.
RESULTS
The in vitro study revealed robust biofilm formation by S. pneumoniae after 12-18 hours of incubation in high glucose medium, independent of exogenously supplied competence stimulating peptide and medium replacement. Adherent cells formed three-dimensional structures approximately 20-30 microm thick. The in vivo study revealed that ciliated epithelium was relatively resistant to biofilm formation and that biofilm formation occurred mainly on non-ciliated epithelium of the middle ear cavity. One week after inoculation, biofilm formation was high in 50% of the treated rats and low in 25% of the rats. After 2 weeks, biofilm formation was high and low in 25% and 37.5% of rats, respectively.
CONCLUSION
The results imply that glucose level is important for the S. pneumoniae biofilm formation and S. pneumoniae biofilm formation may play important role in the pathophysiology of OM.
Keyword
MeSH Terms
Figure
Reference
-
1. Klein JO. The burden of otitis media. Vaccine. 2000; 12. 19(Suppl 1):S2–S8. PMID: 11163456.
Article2. Hendley JO. Clinical practice: otitis media. N Engl J Med. 2002; 10. 347(15):1169–1174. PMID: 12374878.3. Revai K, Mamidi D, Chonmaitree T. Association of nasopharyngeal bacterial colonization during upper respiratory tract infection and the development of acute otitis media. Clin Infect Dis. 2008; 2. 46(4):e34–e37. PMID: 18205533.
Article4. Bogaert D, De Groot R, Hermans PW. Streptococcus pneumoniae colonisation: the key to pneumococcal disease. Lancet Infect Dis. 2004; 3. 4(3):144–154. PMID: 14998500.
Article5. Syrjanen RK, Kilpi TM, Kaijalainen TH, Herva EE, Takala AK. Nasopharyngeal carriage of Streptococcus pneumoniae in Finnish children younger than 2 years old. J Infect Dis. 2001; 8. 184(4):451–459. PMID: 11471103.6. Allegrucci M, Hu FZ, Shen K, Hayes J, Ehrlich GD, Post JC, et al. Phenotypic characterization of Streptococcus pneumoniae biofilm development. J Bacteriol. 2006; 4. 188(7):2325–2335. PMID: 16547018.7. Donlan RM, Piede JA, Heyes CD, Sanii L, Murga R, Edmonds P, et al. Model system for growing and quantifying Streptococcus pneumonia biofilms in situ and in real time. Appl Environ Microbiol. 2004; 8. 70(8):4980–4988. PMID: 15294838.8. Reid SD, Hong W, Dew KE, Winn DR, Pang B, Watt J, et al. Streptococcus pneumoniae forms surface-attached communities in the middle ear of experimentally infected chinchillas. J Infect Dis. 2009; 3. 199(6):786–794. PMID: 19434911.9. Mah TF, O'Toole GA. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 2001; 1. 9(1):34–39. PMID: 11166241.
Article10. Fitzgerald G, Williams LS. Modified penicillin enrichment procedure for the selection of bacterial mutants. J Bacteriol. 1975; 4. 122(1):345–346. PMID: 1091629.
Article11. Post JC. Direct evidence of bacterial biofilms in otitis media. Laryngoscope. 2001; 12. 111(12):2083–2094. PMID: 11802002.
Article12. Hall-Stoodley L, Hu FZ, Gieseke A, Nistico L, Nguyen D, Hayes J, et al. Direct detection of bacterial biofilms on the middle-ear mucosa of children with chronic otitis media. JAMA. 2006; 7. 296(2):202–211. PMID: 16835426.
Article13. Ehrlich GD, Veeh R, Wang X, Costerton JW, Hayes JD, Hu FZ, et al. Mucosal biofilm formation on middle-ear mucosa in the chinchilla model of otitis media. JAMA. 2002; 4. 287(13):1710–1715. PMID: 11926896.
Article14. Dohar JE, Hebda PA, Veeh R, Awad M, Costerton JW, Hayes J, et al. Mucosal biofilm formation on middle-ear mucosa in a nonhuman primate model of chronic suppurative otitis media. Laryngoscope. 2005; 8. 115(8):1469–1472. PMID: 16094127.
Article15. Southey-Pillig CJ, Davies DG, Sauer K. Characterization of temporal protein production in Pseudomonas aeruginosa biofilms. J Bacteriol. 2005; 12. 187(23):8114–8126. PMID: 16291684.16. Oggioni MR, Trappetti C, Kadioglu A, Cassone M, Iannelli F, Ricci S, et al. Switch from planktonic to sessile life: a major event in pneumococcal pathogenesis. Mol Microbiol. 2006; 9. 61(5):1196–1210. PMID: 16925554.
Article17. Baldassarri L, Creti R, Recchia S, Imperi M, Facinelli B, Giovanetti E, et al. Therapeutic failures of antibiotics used to treat macrolide-susceptible Streptococcus pyogenes infections may be due to biofilm formation. J Clin Microbiol. 2006; 8. 44(8):2721–2727. PMID: 16891483.18. Tonnaer EL, Graamans K, Sanders EA, Curfs JH. Advances in understanding the pathogenesis of pneumococcal otitis media. Pediatr Infect Dis J. 2006; 6. 25(6):546–552. PMID: 16732155.
Article19. Block SL, Hedrick J, Harrison CJ, Tyler R, Smith A, Findlay R, et al. Community-wide vaccination with the heptavalent pneumococcal conjugate significantly alters the microbiology of acute otitis media. Pediatr Infect Dis J. 2004; 9. 23(9):829–833. PMID: 15361721.
Article20. McEllistrem MC, Adams J, Mason EO, Wald ER. Epidemiology of acute otitis media caused by Streptococcus pneumoniae before and after licensure of the 7-valent pneumococcal protein conjugate vaccine. J Infect Dis. 2003; 12. 188(11):1679–1684. PMID: 14639539.21. Diamond C, Sisson PR, Kearns AM, Ingham HR. Bacteriology of chronic otitis media with effusion. J Laryngol Otol. 1989; 4. 103(4):369–371. PMID: 2497219.
Article22. Giebink GS, Juhn SK, Weber ML, Le CT. The bacteriology and cytology of chronic otitis media with effusion. Pediatr Infect Dis. 1982; Mar-Apr. 1(2):98–103. PMID: 6983682.
Article23. Rayner MG, Zhang Y, Gorry MC, Chen Y, Post JC, Ehrlich GD. Evidence of bacterial metabolic activity in culture-negative otitis media with effusion. JAMA. 1998; 1. 279(4):296–299. PMID: 9450714.
Article24. Moscoso M, Garcia E, Lopez R. Biofilm formation by Streptococcus pneumoniae: role of choline, extracellular DNA, and capsular polysaccharide in microbial accretion. J Bacteriol. 2006; 11. 188(22):7785–7795. PMID: 16936041.25. Smith A, Buchinsky FJ, Post JC. Eradicating chronic ear, nose, and throat infections: a systematically conducted literature review of advances in biofilm treatment. Otolaryngol Head Neck Surg. 2011; 3. 144(3):338–347. PMID: 21493193.26. Zuliani G, Carlisle M, Duberstein A, Haupert M, Syamal M, Berk R, et al. Biofilm density in the pediatric nasopharynx: recurrent acute otitis media versus obstructive sleep apnea. Ann Otol Rhinol Laryngol. 2009; 7. 118(7):519–524. PMID: 19708492.
Article27. Song JJ, Kown SK, Kim EJ, Lee YS, Kim BY, Chae SW. Mucosal expression of ENaC and AQP in experimental otitis media induced by Eustachian tube obstruction. Int J Pediatr Otorhinolaryngol. 2009; 11. 73(11):1589–1593. PMID: 19732969.
Article28. van Heerbeek N, Straetemans M, Wiertsema SP, Ingels KJ, Rijkers GT, Schilder AG, et al. Effect of combined pneumococcal conjugate and polysaccharide vaccination on recurrent otitis media with effusion. Pediatrics. 2006; 3. 117(3):603–608. PMID: 16510637.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effects of Antibiotics and Steroid on the Middle Ear Mucosa in the Rats with Experimental Acute Otitis Media
- Streptococcus pneumoniae Isolates with Decreased Susceptibility to Optochin
- Formation of Biofilm in Patients with Chronic Otitis Media and Cholesteatoma
- Influence of Cell Surface Hydrophobicity on Adhesion and Biofilm Formation in Candida albicans and Several Bacterial Species
- Detection of Haemophilus Influenzae and Streptococcus Pneumoniae by Polymerase Chain Reaction (PCR) in Chronic Otitis Media with Effusion (OME)