J Korean Soc Radiol.
2012 Dec;67(6):461-464. 10.3348/jksr.2012.67.6.461.
Apocrine Ductal Carcinoma In Situ of the Breast Presented Mass with Morphological Change on Follow-Up Ultrasound: A Report of Case
- Affiliations
-
- 1Department of Radiology, Chung-Ang University Hospital, Seoul, Korea. ksj1567@hanmail.net
- 2Department of Pathology, Chung-Ang University Hospital, Seoul, Korea.
- KMID: 2097982
- DOI: http://doi.org/10.3348/jksr.2012.67.6.461
Abstract
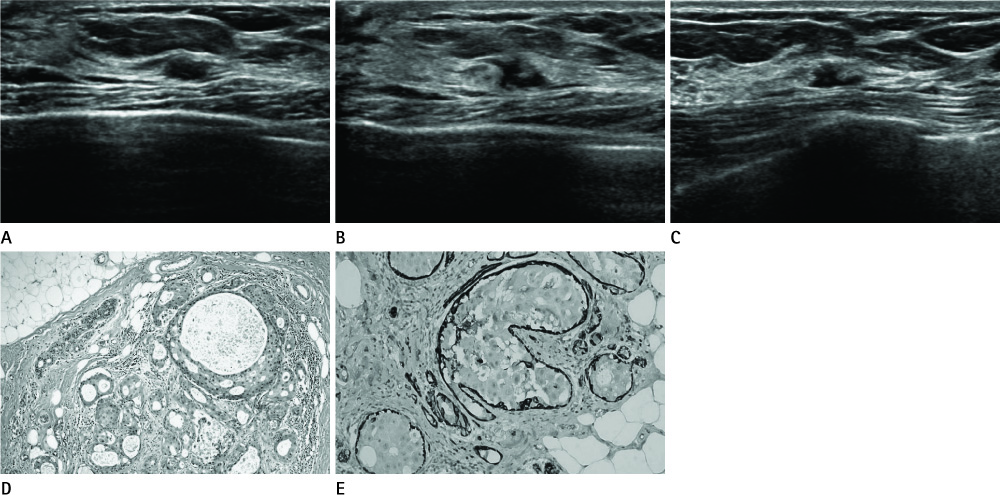
- Apocrine carcinoma in situ of breast is a rare, unique, and morphologically distinct type of breast carcinoma. Low-grade apocrine ductal carcinoma in situ (DCIS) and apocrine metaplasia with atypia are the pathologic spectrum of apocrine breast lesions. Differentiating these two lesions is difficult due to partial microscopic overlap. We describe a case of apocrine DCIS which presented an asymptomatic hypoechoic mass with morphological change on a follow up ultrasonography.
MeSH Terms
Figure
Reference
-
1. O'Malley FP, Bane A. An update on apocrine lesions of the breast. Histopathology. 2008. 52:3–10.2. Visscher DW. Apocrine ductal carcinoma in situ involving a sclerosing lesion with adenosis: report of a case. Arch Pathol Lab Med. 2009. 133:1817–1821.3. ACR BI-RADS-ultrasound. ACR breast imaging reporting and data system, breast imaging atlas. 2003. Reston, Va: American College of Radiology.4. Doshi DJ, March DE, Crisi GM, Coughlin BF. Complex cystic breast masses: diagnostic approach and imaging-pathologic correlation. Radiographics. 2007. 27:Suppl 1. S53–S64.5. Ogiya A, Horii R, Osako T, Ito Y, Iwase T, Eishi Y, et al. Apocrine metaplasia of breast cancer: clinicopathological features and predicting response. Breast Cancer. 2010. 17:290–297.6. Seidman JD, Ashton M, Lefkowitz M. Atypical apocrine adenosis of the breast: a clinicopathologic study of 37 patients with 8.7-year follow-up. Cancer. 1996. 77:2529–2537.7. Wells CA, McGregor IL, Makunura CN, Yeomans P, Davies JD. Apocrine adenosis: a precursor of aggressive breast cancer? J Clin Pathol. 1995. 48:737–742.8. Warner JK, Kumar D, Berg WA. Apocrine metaplasia: mammographic and sonographic appearances. AJR Am J Roentgenol. 1998. 170:1375–1379.9. Kim DY, Kang SS, Ji EK, Kwon TH, Park HL, Shim JY. Sonographic and mammographic features of breast apocrine metaplasia. J Korean Soc Ultrasound Med. 2008. 27:35–40.10. O'Malley FP, Bane AL. The spectrum of apocrine lesions of the breast. Adv Anat Pathol. 2004. 11:1–9.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Apocrine Carcinoma of the Breast: Report of two cases
- Apocrine Carcinoma of the Breast: The report of 2 cases
- Invasive Lobular Carcinoma of the Breast Associated with Mixed Lobular and Ductal Carcinoma In Situ: A Case Report
- An Unusual Presentation of Extensive Ductal Carcinoma in Situ Accompanying Invasive Ductal Carcinoma on MRI: A Case Report
- Clinicopathologic Characteristics of Apocrine Breast Carcinoma