J Clin Neurol.
2006 Dec;2(4):213-224. 10.3988/jcn.2006.2.4.213.
Therapeutic Strategies in Huntington's Disease
- Affiliations
-
- 1National Center of Neurology and Psychiatry, Kodaira, Tokyo, Japan. ichiro@ncnp.go.jp
- KMID: 1980460
- DOI: http://doi.org/10.3988/jcn.2006.2.4.213
Abstract
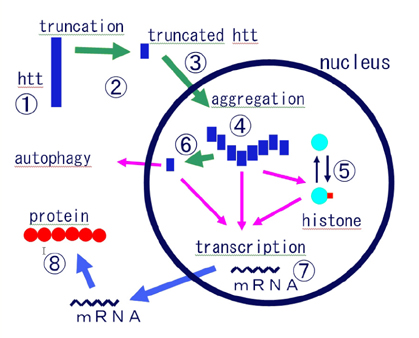
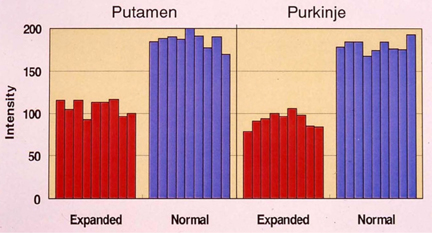
- This article provides an overview of the therapeutic strategies, from ordinary classical drugs to the modern molecular strategy at experimental level, for Huntington's disease. The disease is characterized by choreic movements, psychiatric disorders, striatal atrophy with selective small neuronal loss, and autosomal dominant inheritance. The genetic abnormality is CAG expansion in huntingtin gene. Mutant huntingtin with abnormally long glutamine stretch aggregates and forms intranuclear inclusions. In this review, I summarize the results of previous trials from the following aspects; 1. symptomatic/palliative therapies including drugs, stereotaxic surgery and repetitive transcranial magnetic stimulation, 2. anti-degenerative therapies including anti-excitotoxicity, reversal of mitochondrial dysfunction and anti-apoptosis, 3. restorative/reparative therapies including neural trophic factors and tissue or stem cell transplantation, and 4. molecular targets in specific and radical therapies including inhibition of truncation of huntingtin, inhibition of aggregate formation, normalization of transcriptional dysregulation, enhancement of autophagic clearance of mutant huntingtin, and specific inhibition of huntingtin expression by sRNAi. Although the strategies mentioned in the latter two categories are mostly at laboratory level at present, we are pleased that one can discuss such "therapeutic strategies", a matter absolutely impossible before the causal gene of Huntington's disease was identified more than 10 years ago. It is also true, however, that some of the "therapeutic strategies" mentioned here would be found difficult to implement and abandoned in the future.
Keyword
MeSH Terms
Figure
Reference
-
1. Perry TL, Hansen S, Kloster M. Huntington's chorea. Deficiency of gamma-aminobutyric acid in brain. N Engl J Med. 1973. 288:337–342.2. Spokes EG. Neurochemical alterations in Huntington's chorea: A study of post-mortem brain tissue. Brain. 1980. 103:179–210.3. Gusella JF, Wexler NS, Conneally PM, Naylor SL, Anderson MA, Tanzi RE, et al. A polymorphic DNA marker genetically linked to Huntington's disease. Nature. 1983. 306:234–238.
Article4. The Huntington's disease collaborative research group. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. Cell. 1993. 72:971–983.5. Davies SW, Turmaine M, Cozens BA, DiFiglia M, Sharp AH, Ross CA, et al. Formation of neuronal intranuclear inclusions underlies the neurological dysfunction in mice transgenic for the hd mutation. Cell. 1997. 90:537–548.
Article6. Trushina E, Heldebrant MP, Perez-Terzic CM, Bortolon R, Kovtun IV, Badger JD, et al. Microtubule destabilization and nuclear entry are sequential steps leading to toxicity in Huntington's disease. Proc Natl Acad Sci USA. 2003. 100:12171–12176.
Article7. Shoulson I, Chase TN, Roberts E, Van Balgooy JN. Letter: Huntington's disease: Treatment with imidazole-4-acetic acid. N Engl J Med. 1975. 293:504–505.8. Shoulson I, Kartzinel R, Chase TN. Huntington's disease: Treatment with dipropylacetic acid and gamma-amino-butyric acid. Neurology. 1976. 26:61–63.
Article9. Shoulson I, Goldblatt D, Charlton M, Joynt RJ. Huntington's disease: Treatment with muscimol, a gaba-mimetic drug. Ann Neurol. 1978. 4:279–284.
Article10. Perry TL, Wright JM, Hansen S, Thomas SM, Allan BM, Baird PA, et al. A double-blind clinical trial of isoniazid in Huntington disease. Neurology. 1982. 32:354–358.
Article11. Shoulson I, Odoroff C, Oakes D, Behr J, Goldblatt D, Caine E, et al. A controlled clinical trial of baclofen as protective therapy in early Huntington's disease. Ann Neurol. 1989. 25:252–259.
Article12. O'Suilleabhain P, Dewey RB Jr. A randomized trial of amantadine in Huntington disease. Arch Neurol. 2003. 60:996–998.13. Bonelli RM, Wenning GK. Pharmacological management of Huntington's disease: An evidence-based review. Curr Pharm Des. 2006. 12:2701–2720.
Article14. Huntington Study Group. Tetrabenazine as antichorea therapy in Huntington disease: A randomized controlled trial. Neurology. 2006. 66:366–372.15. Puri BK, Leavitt BR, Hayden MR, Ross CA, Rosenblatt A, Greenamyre JT, et al. Ethyl-epa in Huntington disease: A double-blind, randomized, placebo-controlled trial. Neurology. 2005. 65:286–292.
Article16. NarabayashiH SM, Ohye C. Experience with stereotaxic surgery on choreic movements with some physiological interpretations. Adv Neurol. 1973. 1:789–793.17. Priori A, Berardelli A, Inghilleri M, Polidori L, Manfredi M. Electromyographic silent period after transcranial brain stimulation in Huntington's disease. Mov Disord. 1994. 9:178–182.
Article18. Brusa L, Versace V, Koch G, Bernardi G, Iani C, Stanzione P, et al. Improvement of choreic movements by 1 hz repetitive transcranial magnetic stimulation in Huntington's disease patients. Ann Neurol. 2005. 58:655–656.
Article19. Huntington Study Group. Dosage effects of riluzole in Huntington's disease: A multicenter placebo-controlled study. Neurology. 2003. 61:1551–1556.20. Browne SE, Ferrante RJ, Beal MF. Oxidative stress in Huntington's disease. Brain Pathol. 1999. 9:147–163.
Article21. Huntington Study Group. A randomized, placebo-controlled trial of coenzyme Q10 and remacemide in Huntington's disease. Neurology. 2001. 57:397–404.22. Ryu H, Rosas HD, Hersch SM, Ferrante RJ. The therapeutic role of creatine in Huntington's disease. Pharmacol Ther. 2005. 108:193–207.
Article23. Hersch SM, Gevorkian S, Marder K, Moskowitz C, Feigin A, Cox M, et al. Creatine in Huntington disease is safe, tolerable, bioavailable in brain and reduces serum 8oh2'dg. Neurology. 2006. 66:250–252.
Article24. Chen M, Ona VO, Li M, Ferrante RJ, Fink KB, Zhu S, et al. Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nat Med. 2000. 6:797–801.
Article25. Bonelli RM, Hodl AK, Hofmann P, Kapfhammer HP. Neuroprotection in Huntington's disease: A 2-year study on minocycline. Int Clin Psychopharmacol. 2004. 19:337–342.
Article26. Keene CD, Rodrigues CM, Eich T, Chhabra MS, Steer CJ, Low WC. Tauroursodeoxycholic acid, a bile acid, is neuroprotective in a transgenic animal model of Huntington's disease. Proc Natl Acad Sci USA. 2002. 99:10671–10676.
Article27. Ravikumar B, Berger Z, Vacher C, O'Kane CJ, Rubinsztein DC. Rapamycin pre-treatment protects against apoptosis. Hum Mol Genet. 2006. 15:1209–1216.
Article28. Zuccato C, Ciammola A, Rigamonti D, Leavitt BR, Goffredo D, Conti L, et al. Loss of huntingtin-mediated bdnf gene transcription in Huntington's disease. Science. 2001. 293:493–498.
Article29. Popovic N, Maingay M, Kirik D, Brundin P. Lentiviral gene delivery of gdnf into the striatum of r6/2 Huntington mice fails to attenuate behavioral and neuropathological changes. Exp Neurol. 2005. 193:65–74.
Article30. Jin K, LaFevre-Bernt M, Sun Y, Chen S, Gafni J, Crippen D, et al. FGF-2 promotes neurogenesis and neuroprotection and prolongs survival in a transgenic mouse model of Huntington's disease. Proc Natl Acad Sci USA. 2005. 102:18189–18194.
Article31. Emerich DF, Thanos CG. Intracompartmental delivery of cntf as therapy for Huntington's disease and retinitis pigmentosa. Curr Gene Ther. 2006. 6:147–159.
Article32. Isacson O, Brundin P, Gage FH, Bjorklund A. Neural grafting in a rat model of Huntington's disease: Progressive neurochemical changes after neostriatal ibotenate lesions and striatal tissue grafting. Neuroscience. 1985. 16:799–817.
Article33. Bachoud-Levi AC, Gaura V, Brugieres P, Lefaucheur JP, Boisse MF, Maison P, et al. Effect of fetal neural transplants in patients with Huntington's disease 6 years after surgery: A long-term follow-up study. Lancet Neurol. 2006. 5:303–309.
Article34. Dunnett SB, Rosser AE. Cell therapy in Huntington's disease. NeuroRx. 2004. 1:394–405.
Article35. Lee ST, Chu K, Park JE, Lee K, Kang L, Kim SU, Kim M. Intravenous administration of human neural stem cells induces functional recovery in Huntington's disease rat model. Neurosci Res. 2005. 52:243–249.
Article36. DiFiglia M, Sapp E, Chase KO, Davies SW, Bates GP, Vonsattel JP, et al. Aggregation of huntingtin in neuronal intranuclear inclusions and dystrophic neurites in brain. Science. 1997. 277:1990–1993.
Article37. Perutz MF, Johnson T, Suzuki M, Finch JT. Glutamine repeats as polar zippers: Their possible role in inherited neurodegenerative diseases. Proc Natl Acad Sci USA. 1994. 91:5355–5358.
Article38. Hazeki N, Tsukamoto T, Yazawa I, Koyama M, Hattori S, Someki I, et al. Ultrastructure of nuclear aggregates formed by expressing an expanded polyglutamine. Biochem Biophys Res Commun. 2002. 294:429–440.
Article39. Gines S, Seong IS, Fossale E, Ivanova E, Trettel F, Gusella JF, et al. Specific progressive camp reduction implicates energy deficit in presymptomatic Huntington's disease knock-in mice. Hum Mol Genet. 2003. 12:497–508.
Article40. Cattaneo E, Rigamonti D, Goffredo D, Zuccato C, Squitieri F, Sipione S. Loss of normal huntingtin function: New developments in Huntington's disease research. Trends Neurosci. 2001. 24:182–188.
Article41. Kim M, Lee HS, LaForet G, McIntyre C, Martin EJ, Chang P. Mutant huntingtin expression in clonal striatal cells: Dissociation of inclusion formation and neuronal survival by caspase inhibition. J Neurosci. 1999. 19:964.
Article42. Graham RK, Deng Y, Slow EJ, Haigh B, Bissada N, Lu G, et al. Cleavage at the caspase-6 site is required for neuronal dysfunction and degeneration due to mutant huntingtin. Cell. 2006. 125:1179–1191.
Article43. Cariello L, de Cristofaro T, Zanetti L, Cuomo T, Di Maio L, Campanella G, et al. Transglutaminase activity is related to cag repeat length in patients with Huntington's disease. Hum Genet. 1996. 98:633–635.
Article44. Chun W, Lesort M, Tucholski J, Faber PW, MacDonald ME, Ross CA, et al. Tissue transglutaminase selectively modifies proteins associated with truncated mutant huntingtin in intact cells. Neurobiol Dis. 2001. 8:391–404.
Article45. Karpuj MV, Becher MW, Springer JE, Chabas D, Youssef S, Pedotti R, et al. Prolonged survival and decreased abnormal movements in transgenic model of Huntington disease, with administration of the transglutaminase inhibitor cystamine. Nat Med. 2002. 8:143–149.
Article46. Wang X, Sarkar A, Cicchetti F, Yu M, Zhu A, Jokivarsi K, et al. Cerebral pet imaging and histological evidence of transglutaminase inhibitor cystamine induced neuroprotection in transgenic r6/2 mouse model of Huntington's disease. J Neurol Sci. 2005. 231:57–66.
Article47. Zhang X, Smith DL, Meriin AB, Engemann S, Russel DE, Roark M, et al. A potent small molecule inhibits polyglutamine aggregation in Huntington's disease neurons and suppresses neurodegeneration in vivo. Proc Natl Acad Sci USA. 2005. 102:892–897.
Article48. Bodner RA, Outeiro TF, Altmann S, Maxwell MM, Cho SH, Hyman BT, et al. Pharmacological promotion of inclusion formation: A therapeutic approach for Huntington's and parkinson's diseases. Proc Natl Acad Sci USA. 2006. 103:4246–4251.
Article49. Heiser V, Engemann S, Brocker W, Dunkel I, Boeddrich A, Waelter S, et al. Identification of benzothiazoles as potential polyglutamine aggregation inhibitors of Huntington's disease by using an automated filter retardation assay. Proc Natl Acad Sci USA. 2002. 99:Suppl 4. 16400–16406.
Article50. Pollitt SK, Pallos J, Shao J, Desai UA, Ma AA, Thompson LM, et al. A rapid cellular fret assay of polyglutamine aggregation identifies a novel inhibitor. Neuron. 2003. 40:685–694.
Article51. Tanaka M, Machida Y, Niu S, Ikeda T, Jana NR, Doi H, et al. Trehalose alleviates polyglutamine-mediated pathology in a mouse model of Huntington disease. Nat Med. 2004. 10:148–154.
Article52. Sanchez I, Mahlke C, Yuan J. Pivotal role of oligomerization in expanded polyglutamine neurodegenerative disorders. Nature. 2003. 421:373–379.
Article53. Miller VM, Nelson RF, Gouvion CM, Williams A, Rodriguez-Lebron E, Harper SQ, et al. Chip suppresses polyglutamine aggregation and toxicity in vitro and in vivo. J Neurosci. 2005. 25:9152–9161.
Article54. Hockly E, Tse J, Barker AL, Moolman DL, Beunard JL, Revington AP, et al. Evaluation of the benzothiazole aggregation inhibitors riluzole and pgl-135 as therapeutics for Huntington's disease. Neurobiol Dis. 2006. 21:228–236.
Article55. Wang J, Gines S, MacDonald ME, Gusella JF. Reversal of a full-length mutant huntingtin neuronal cell phenotype by chemical inhibitors of polyglutamine-mediated aggregation. BMC Neurosci. 2005. 6:1–12.56. Jana NR, Tanaka M, Wang G, Nukina N. Polyglutamine length-dependent interaction of hsp40 and hsp70 family chaperones with truncated n-terminal huntingtin: Their role in suppression of aggregation and cellular toxicity. Hum Mol Genet. 2000. 9:2009–2018.
Article57. Smith DF, Whitesell L, Nair SC, Chen S, Prapapanich V, Rimerman RA. Progesterone receptor structure and function altered by geldanamycin, an hsp90-binding agent. Mol Cell Biol. 1995. 15:6804–6812.
Article58. Sittler A, Lurz R, Lueder G, Priller J, Lehrach H, Hayer-Hartl MK, et al. Geldanamycin activates a heat shock response and inhibits huntingtin aggregation in a cell culture model of Huntington's disease. Hum Mol Genet. 2001. 10:1307–1315.
Article59. Hay DG, Sathasivam K, Tobaben S, Stahl B, Marber M, Mestril R, et al. Progressive decrease in chaperone protein levels in a mouse model of Huntington's disease and induction of stress proteins as a therapeutic approach. Hum Mol Genet. 2004. 13:1389–1405.
Article60. Gerber HP, Seipel K, Georgiev O, Hofferer M, Hug M, Rusconi S, et al. Transcriptional activation modulated by homopolymeric glutamine and proline stretches. Science. 1994. 263:808–811.
Article61. Okazawa H. Polyglutamine diseases: A transcription disorder? Cell Mol Life Sci. 2003. 60:1427–1439.
Article62. Cha JH. Transcriptional dysregulation in Huntington's disease. Trends Neurosci. 2000. 23:387–392.
Article63. Zhai W, Jeong H, Cui L, Krainc D, Tjian R. In vitro analysis of huntingtin-mediated transcriptional repression reveals multiple transcription factor targets. Cell. 2005. 123:1241–1253.
Article64. Steffan JS, Bodai L, Pallos J, Poelman M, McCampbell A, Apostol BL, et al. Histone deacetylase inhibitors arrest polyglutamine-dependent neurodegeneration in drosophila. Nature. 2001. 413:739–743.
Article65. Ferrante RJ, Kubilus JK, Lee J, Ryu H, Beesen A, Zucker B, et al. Histone deacetylase inhibition by sodium butyrate chemotherapy ameliorates the neurodegenerative phenotype in Huntington's disease mice. J Neurosci. 2003. 23:9418–9427.
Article66. Noda T, Ohsumi Y. Tor, a phosphatidylinositol kinase homologue, controls autophagy in yeast. J Biol Chem. 1998. 273:3963–3966.
Article67. Ravikumar B, Stewart A, Kita H, Kato K, Duden R, Rubinsztein DC. Raised intracellular glucose concentrations reduce aggregation and cell death caused by mutant huntingtin exon 1 by decreasing mtor phosphorylation and inducing autophagy. Hum Mol Genet. 2003. 12:985–994.
Article68. Ravikumar B, Vacher C, Berger Z, Davies JE, Luo S, Oroz LG, et al. Inhibition of mtor induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nat Genet. 2004. 36:585–595.
Article69. Yamamoto A, Cremona ML, Rothman JE. Autophagy-mediated clearance of huntingtin aggregates triggered by the insulin-signaling pathway. J Cell Biol. 2006. 172:719–731.
Article70. Haque NS, Borghesani P, Isacson O. Therapeutic strategies for Huntington's disease based on a molecular understanding of the disorder. Mol Med Today. 1997. 3:175–183.
Article71. Yamamoto A, Lucas JJ, Hen R. Reversal of neuropathology and motor dysfunction in a conditional model of Huntington's disease. Cell. 2000. 101:57–66.
Article72. Boado RJ, Kazantsev A, Apostol BL, Thompson LM, Pardridge WM. Antisense-mediated down-regulation of the human huntingtin gene. J Pharmacol Exp Ther. 2000. 295:239–243.73. Nellemann C, Abell K, Norremolle A, Lokkegaard T, Naver B, Ropke C, et al. Inhibition of Huntington synthesis by antisense oligodeoxynucleotides. Mol Cell Neurosci. 2000. 16:313–323.74. Yen L, Strittmatter SM, Kalb RG. Sequence-specific cleavage of huntingtin mRNA by catalytic DNA. Ann Neurol. 1999. 46:366–373.
Article75. Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC. Potent and specific genetic interference by double-stranded RNA in caenorhabditis elegans. Nature. 1998. 391:806–811.
Article76. Hannon GJ. RNA interference. Nature. 2002. 418:244–251.
Article77. Elbashir SM, Lendeckel W, Tuschl T. RNA interference is mediated by 21- and 22-nucleotide RNAs. Genes Dev. 2001. 15:188–200.
Article78. Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001. 411:494–498.
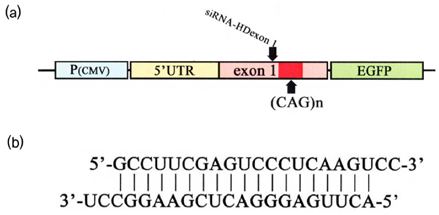
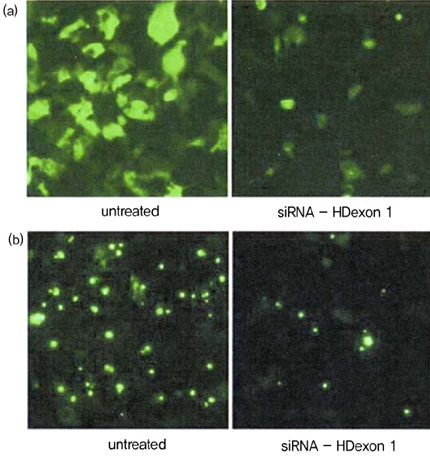
Article79. Liu WZ, Wang YL, Murata M, Wada K, Kanazawa I. Specific inhibition of Huntington's disease gene expression by siRNAs in cultured cells. Proc Japan Acad. 2003. 79:239–298.
Article80. Wang YL, Liu W, Wada E, Murata M, Wada K, Kanazawa I. Clinico-pathological rescue of a model mouse of Huntington's disease by siRNA. Neurosci Res. 2005. 53:241–249.
Article81. Harper SQ, Staber PD, He X, Eliason SL, Martins IH, Mao Q, et al. RNA interference improves motor and neuropathological abnormalities in a Huntington's disease mouse model. Proc Natl Acad Sci USA. 2005. 102:5820–5825.
Article82. Rodriguez-Lebron E, Denovan-Wright EM, Nash K, Lewin AS, Mandel RJ. Intrastriatal raav-mediated delivery of anti-huntingtin shRNAs induces partial reversal of disease progression in r6/1 Huntington's disease transgenic mice. Mol Ther. 2005. 12:618–633.
Article83. Machida Y, Okada T, Kurosawa M, Oyama F, Ozawa K, Nukina N. Raav-mediated shRNA ameliorated neuropathology in Huntington disease model mouse. Biochem Biophys Res Commun. 2006. 343:190–197.
Article84. Grimm D, Streetz KL, Jopling CL, Storm TA, Pandey K, Davis CR, et al. Fatality in mice due to oversaturation of cellular microRNA/short hairpin RNA pathways. Nature. 2006. 441:537–541.
Article85. Aronin N. Target selectivity in mRNA silencing. Gene Ther. 2006. 13:509–516.
Article86. Nguyen T, Hamby A, Massa SM. Clioquinol down-regulates mutant huntingtin expression in vitro and mitigates pathology in a Huntington's disease mouse model. Proc Natl Acad Sci USA. 2005. 102:11840–11845.
Article