Endocrinol Metab.
2013 Jun;28(2):149-152. 10.3803/EnM.2013.28.2.149.
Transformation of Nonfunctioning Pancreatic Neuroendocrine Carcinoma Cells into Insulin Producing Cells after Treatment with Sunitinib
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea. junghs@snu.ac.kr
- 2Division of Hematology and Medical Oncology, Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea.
- KMID: 1973815
- DOI: http://doi.org/10.3803/EnM.2013.28.2.149
Abstract
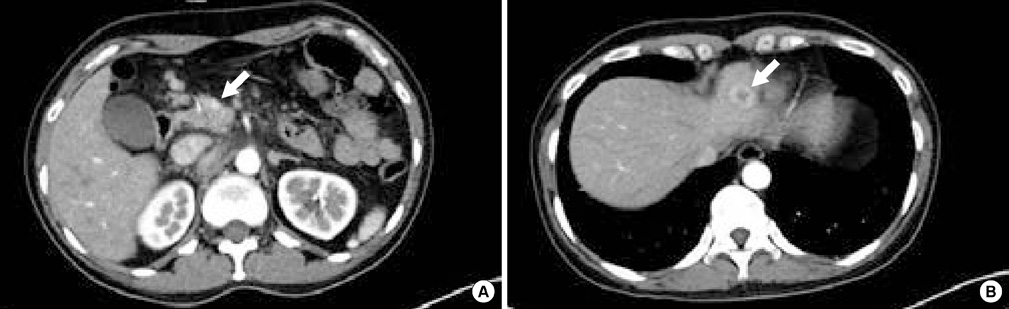
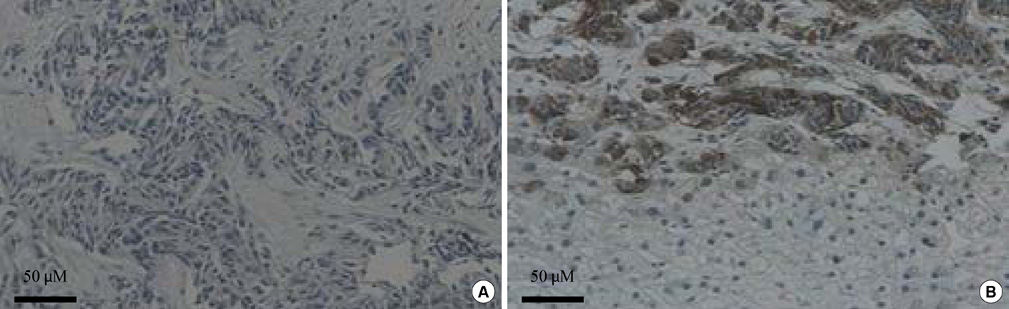
- We report a rare case of severe hypoglycemia after sunitinib treatment for pancreatic neuroendocrine carcinoma. We describe the initial clinical presentation, laboratory results, pathologic findings, and managment in a patient with a nonfunctioning pancreatic neuroendocrine carcinoma with liver metastases who developed life threatening hypoglycemia after 2 months of sunitinib therapy. A 46-year-old woman presented to the emergency department with loss of consciousness from hypoglycemia. Serum C-peptide and insulin levels at fasting state revealed that the hypoglycemia resulted from endogenous hyperinsulinemia. She had been diagnosed with nonfunctioning pancreatic neuroendocrine carcinoma based on a biopsy of metastatic cervical lymph node and was being treated with sunitinib, a small molecule tyrosine kinase inhibitor. Immunohistochemical stain of the metastatic liver mass demonstrated that the initially nonfunctioning neuroendocrine carcinoma cells had changed into insulin-producing cells after sunitinib therapy. Transarterial chemoembolization of the liver masses and systemic chemotherapy with streptozotocin/adriamycin relieved the hypoglycemia. A nonfunctioning pancreatic neuroendocrine carcinoma was transformed into an insulin-producing tumor after treatment with sunitinib, causing endogenous hyperinsulinemia and severe hypoglycemia.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Brief Review of Articles in '
Endocrinology and Metabolism ' in 2013
Won-Young Lee
Endocrinol Metab. 2014;29(3):251-256. doi: 10.3803/EnM.2014.29.3.251.
Reference
-
1. Billemont B, Medioni J, Taillade L, Helley D, Meric JB, Rixe O, Oudard S. Blood glucose levels in patients with metastatic renal cell carcinoma treated with sunitinib. Br J Cancer. 2008. 99:1380–1382.2. Mokhtari D, Welsh N. Potential utility of small tyrosine kinase inhibitors in the treatment of diabetes. Clin Sci (Lond). 2009. 118:241–247.3. Templeton A, Brandle M, Cerny T, Gillessen S. Remission of diabetes while on sunitinib treatment for renal cell carcinoma. Ann Oncol. 2008. 19:824–825.4. Lee Y, Jung HS, Choi HJ, Kim MJ, Kim TM, Park KS, Kim SY. Life-threatening hypoglycemia induced by a tyrosine kinase inhibitor in a patient with neuroendocrine tumor: a case report. Diabetes Res Clin Pract. 2011. 93:e68–e70.5. Vashi PG, Gupta D, Dahlk S. A unique case of a nonfunctional metastatic pancreatic neuroendocrine tumor transforming into an insulin-secreting tumor with an unusual clinical course. Pancreas. 2011. 40:781–784.6. Hagerkvist R, Jansson L, Welsh N. Imatinib mesylate improves insulin sensitivity and glucose disposal rates in rats fed a high-fat diet. Clin Sci (Lond). 2008. 114:65–71.7. Hagerkvist R, Makeeva N, Elliman S, Welsh N. Imatinib mesylate (Gleevec) protects against streptozotocin-induced diabetes and islet cell death in vitro. Cell Biol Int. 2006. 30:1013–1017.8. Hagerkvist R, Sandler S, Mokhtari D, Welsh N. Amelioration of diabetes by imatinib mesylate (Gleevec): role of beta-cell NF-kappaB activation and anti-apoptotic preconditioning. FASEB J. 2007. 21:618–628.9. Han MS, Chung KW, Cheon HG, Rhee SD, Yoon CH, Lee MK, Kim KW, Lee MS. Imatinib mesylate reduces endoplasmic reticulum stress and induces remission of diabetes in db/db mice. Diabetes. 2009. 58:329–336.10. Louvet C, Szot GL, Lang J, Lee MR, Martinier N, Bollag G, Zhu S, Weiss A, Bluestone JA. Tyrosine kinase inhibitors reverse type 1 diabetes in nonobese diabetic mice. Proc Natl Acad Sci U S A. 2008. 105:18895–18900.11. Chen H, Gu X, Liu Y, Wang J, Wirt SE, Bottino R, Schorle H, Sage J, Kim SK. PDGF signalling controls age-dependent proliferation in pancreatic beta-cells. Nature. 2011. 478:349–355.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of ACTH-producing Pancreatic Neuroendocrine Tumor Presenting with Cushing Syndrome
- Cell Replacement and Regeneration Therapy for Diabetes
- Hypertriglyceridemia Associated with Use of Sunitinib to Treat a Metastatic Pancreatic Neuroendocrine Tumor
- Nonfunctioning Neuroendocrine Tumor of the Pancreas in a 15-year-old Girl: a Case Report
- Adenoviruses Expressing PDX-1, BETA2/NeuroD and MafA Induces the Transdifferentiation of Porcine Neonatal Pancreas Cell Clusters and Adult Pig Pancreatic Cells into Beta-Cells