J Korean Med Sci.
2009 Oct;24(5):782-788. 10.3346/jkms.2009.24.5.782.
Myocardial Protective Effect of Tezosentan, an Endothelin Receptor Antagonist, for Ischemia-Reperfusion Injury in Experimental Heart Failure Models
- Affiliations
-
- 1Department of Thoracic and Cardiovascular Surgery, College of Medicine, Kangwon National University, Chuncheon, Korea.
- 2Department of Thoracic and Cardiovascular Surgery, Korea University Guro Hospital, Korea University College of Medicine, Seoul, Korea.
- 3Department of Radiology, Korea University Anam Hospital, Korea University College of Medicine, Seoul, Korea.
- 4Department of Thoracic and Cardiovascular Surgery, Korea University Ansan Hospital, Korea University College of Medicine, Ansan, Korea. jowonmin@korea.ac.kr
- KMID: 1781994
- DOI: http://doi.org/10.3346/jkms.2009.24.5.782
Abstract
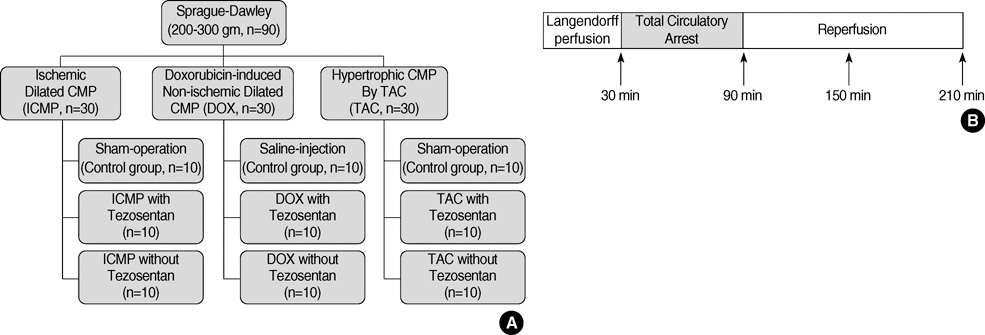
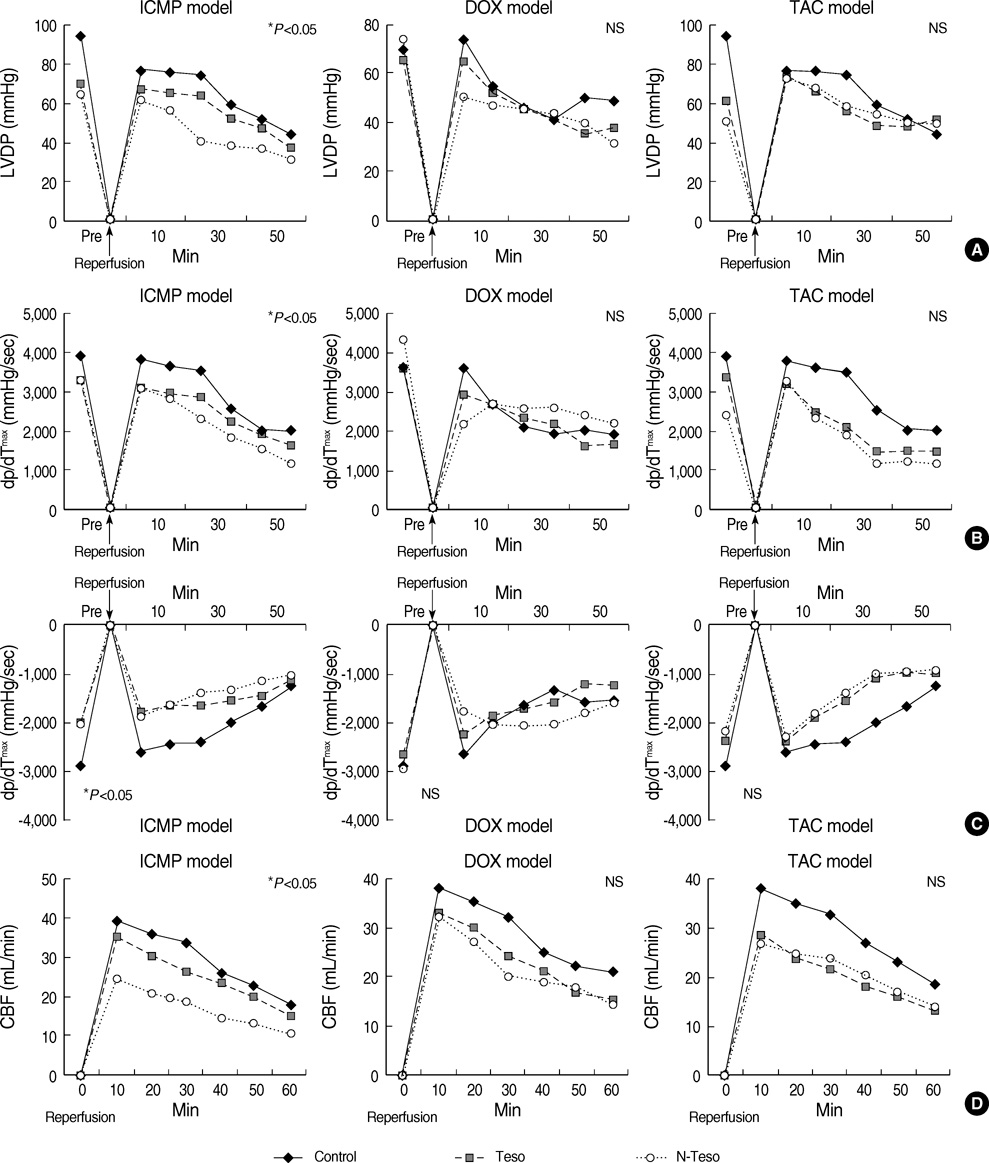
- The myocardial protective effects of endothelin antagonist in ischemic cardiomyopathy (ICMP), doxorubicin-induced cardiomyopathy (DOX) and pressure-overload hypertrophy by transverse aortic constriction (TAC) models have been predicted to be different. The objective of this experiment, therefore, is to evaluate the myocardial protective effect of tezosentan, an endothelin receptor antagonist, in various experimental heart failure models. Sprague-Dawley rats (6-8 weeks old, 200-300 g) were randomized to three experimental groups (n=30 each): ICMP; DOX; and TAC group. Each of these groups was randomly assigned further to the following subgroups (n=10 each): sham-operated ischemia-reperfusion subgroup (SHAM); tezosentan treated ischemia-reperfusion subgroup (Tezo); and tezosentan non-treated ischemia-reperfusion subgroup (N-Tezo). Total circulatory arrest was induced for 1 hr, followed by 2 hr of reperfusion. The left ventricular developed pressure, peak positive and negative first derivatives, and coronary blood flow were significantly different (P<0.05) among the SHAM, Tezo, and N-Tezo subgroups of the ICMP group at 30 min of reperfusion, but there were no statistically significant differences among the subgroups of the DOX and TAC groups. In conclusion, tezosentan, an endothelin receptor antagonist, showed myocardial protection effects only on the ischemic cardiomyopathy rat model, but not in the non-ischemic heart failure rat models.
MeSH Terms
-
Animals
Cardiomyopathies/chemically induced/drug therapy/physiopathology
Coronary Vessels/physiology
Disease Models, Animal
Doxorubicin/toxicity
Heart Failure/*drug therapy/physiopathology
Hypertrophy/drug therapy/physiopathology
Male
Pressure
Pyridines/*therapeutic use
Rats
Rats, Sprague-Dawley
Receptors, Endothelin/*antagonists & inhibitors/metabolism
Reperfusion Injury/*drug therapy/physiopathology/surgery
Tetrazoles/*therapeutic use
Vasodilator Agents/*therapeutic use
Ventricular Function, Left/physiology
Figure
Reference
-
1. Parker JD, Thiessen JJ, Reilly R, Tong JH, Stewart DJ, Pandey AS. Human endothelin-1 clearance kinetics revealed by a radiotracer technique. J Pharmacol Exp Ther. 1999. 289:261–265.2. Inada T, Fujiwara H, Hasegawa K, Araki M, Yamauchi-Kohno R, Yabana H, Fujiwara T, Tanaka M, Sasayama S. Upregulated expression of cardiac endothelin-1 participates in myocardial cell growth in Bio14.6 Syrian cardiomyopathic hamsters. J Am Coll Cardiol. 1999. 33:565–571.
Article3. Wittner M, Christ GJ, Huang H, Weiss LM, Hatcher VB, Morris SA, Orr GA, Berman JW, Zeballos GA, Douglas SA, Tanowitz HB. Trypanosoma cruzi induces endothelin release from endothelial cells. J Infect Dis. 1995. 171:493–497.
Article4. Cheng JW. Tezosentan in the management of decompensated heart failure. Cardiol Rev. 2005. 13:28–34.
Article5. Teerlink JR, McMurray JJ, Bourge RC, Cleland JG, Cotter G, Jondeau G, Krum H, Metra M, O'Connor CM, Parker JD, Torre-Amione G, Van Veldhuisen DJ, Frey A, Rainisio M, Kobrin I. Tezosentan in patients with acute heart failure: design of the Value of Endothelin Receptor Inhibition with Tezosentan in Acute heart failure Study (VERITAS). Am Heart J. 2005. 150:46–53.
Article6. Battistini B, Chailler P, D'Orleans-Juste P, Briere N, Sirois P. Growth regulatory properties of endothelins. Peptides. 1993. 14:385–399.
Article7. Kaddoura S, Firth JD, Boheler KR, Sugden PH, Poole-Wilson PA. Endothelin-1 is involved in norepinephrine-induced ventricular hypertrophy in vivo. Acute effects of bosentan, an orally active, mixed endothelin ETA and ETB receptor antagonist. Circulation. 1996. 93:2068–2079.8. Teerlink JR. The development of new medical treatments for acute decompensated heart failure. Heart Fail Monit. 2002. 2:129–137.9. Fishman WH, Kaur S, Singh I, Tamirisa P. Frishman WH, Sonnenblick EH, Sica DA, editors. Endothelin as a therapeutic target in the treatment of cardiovascular disease. Cardiovascular pharmacotherapeutics. 2003. 2nd ed. New York: McGraw-Hill, Medical Pub. Division;527–543.10. Webb DJ. Evidence for endothelin-1-mediated vasoconstriction in severe chronic heart failure. Endothelin antagonism in heart failure. Circulation. 1995. 92:3372.11. Margulies KB, Hildebrand FL Jr, Lerman A, Perrella MA, Burnett JC Jr. Increased endothelin in experimental heart failure. Circulation. 1990. 82:2226–2230.
Article12. Omland T, Lie RT, Aakvaag A, Aarsland T, Dickstein K. Plasma endothelin determination as a prognostic indicator of 1-year mortality after acute myocardial infarction. Circulation. 1994. 89:1573–1579.
Article13. Duchman SM, Thohan V, Kalra D, Torre-Amione G. Endothelin-1: a new target of therapeutic intervention for the treatment of heart failure. Curr Opin Cardiol. 2000. 15:136–140.
Article14. McMurdo L, Thiemermann C, Vane JR. The effects of the endothelin ETA receptor antagonist, FR 139317, on infarct size in a rabbit model of acute myocardial ischaemia and reperfusion. Br J Pharmacol. 1994. 112:75–80.
Article15. Richard V, Kaeffer N, Hogie M, Tron C, Blanc T, Thuillez C. Role of endogenous endothelin in myocardial and coronary endothelial injury after ischaemia and reperfusion in rats: studies with bosentan, a mixed ETA-ETB antagonist. Br J Pharmacol. 1994. 113:869–876.
Article16. Xia Z, Kuo KH, McNeill JH, Ansley DM. Endothelin A and B receptor antagonist bosentan reduces postischemic myocardial injury in the rat: critical timing of administration. Can J Physiol Pharmacol. 2005. 83:259–266.
Article17. Sakai S, Yorikane R, Miyauchi T, Sakurai T, Kasuya Y, Yamaguchi I, Sugishita Y, Goto K. Altered production of endothelin-1 in the hypertrophied rat heart. J Cardiovasc Pharmacol. 1995. 26:Suppl 3. S452–S455.
Article18. Cernacek P, Stewart DJ, Monge JC, Rouleau JL. The endothelin system and its role in acute myocardial infarction. Can J Physiol Pharmacol. 2003. 81:598–606.
Article19. Nguyen QT, Cernacek P, Calderoni A, Stewart DJ, Picard P, Sirois P, White M, Rouleau JL. Endothelin A receptor blockade causes adverse left ventricular remodeling but improves pulmonary artery pressure after infarction in the rat. Circulation. 1998. 98:2323–2330.
Article20. Nguyen QT, Cernacek P, Sirois MG, Calderone A, Lapointe N, Stewart DJ, Rouleau JL. Long-term effects of nonselective endothelin A and B receptor antagonism in postinfarction rat: importance of timing. Circulation. 2001. 104:2075–2081.21. Arola OJ, Saraste A, Pulkki K, Kallajoki M, Parvinen M, Voipio-Pulkki LM. Acute doxorubicin cardiotoxicity involves cardiomyocyte apoptosis. Cancer Res. 2000. 60:1789–1792.22. Watanabe K, Ohta Y, Kouda T, Sekiguchi T, Sato S, Nakazawa M, Hasegawa G, Naito M, Fuse K, Ito M, Hirono S, Tanabe N, Hanawa H, Kato K, Kodama M, Aizawa Y. Acute effects of endothelin-1 and TAK-044 (ET(A) and ET(B) receptor antagonist) in rats with dilated cardiomyopathy. J Cardiovasc Pharmacol. 2000. 36:Suppl 2. S49–S54.
Article23. Heineke J, Molkentin JD. Regulation of cardiac hypertrophy by intracellular signalling pathways. Nat Rev Mol Cell Biol. 2006. 7:589–600.
Article24. Houser SR, Margulies KB. Is depressed myocyte contractility centrally involved in heart failure? Circ Res. 2003. 92:350–358.
Article25. Olson EN. A decade of discoveries in cardiac biology. Nat Med. 2004. 10:467–474.
Article26. Yorikane R, Sakai S, Miyauchi T, Sakurai T, Sugishita Y, Goto K. Increased production of endothelin-1 in the hypertrophied rat heart due to pressure overload. FEBS Lett. 1993. 332:31–34.
Article27. Arai M, Yoguchi A, Iso T, Takahashi T, Imai S, Murata K, Suzuki T. Endothelin-1 and its binding sites are upregulated in pressure overload cardiac hypertrophy. Am J Physiol. 1995. 268:H2084–H2091.
Article28. Sirvio ML, Uhlenius N, Stewen P, Metsarinne K, Fyhrquist F. The effect of aortic coarctation on expression of endothelin-1 and endothelin receptors in heart and lungs. Blood Press. 1995. 4:320–323.29. Wei CM, Lerman A, Rodeheffer RJ, McGregor CG, Brandt RR, Wright S, Heublein DM, Kao PC, Edwards WD, Burnett JC Jr. Endothelin in human congestive heart failure. Circulation. 1994. 89:1580–1586.
Article30. Hayashi M, Tsutamoto T, Wada A, Maeda K, Mabuchi N, Tsutsui T, Horie H, Ohnishi M, Kinoshita M. Intravenous atrial natriuretic peptide prevents left ventricular remodeling in patients with first anterior acute myocardial infarction. J Am Coll Cardiol. 2001. 37:1820–1826.
Article31. Arnolda L, McGrath B, Cocks M, Sumithran E, Johnston C. Adriamycin cardiomyopathy in the rabbit: an animal model of low output cardiac failure with activation of vasoconstrictor mechanisms. Cardiovasc Res. 1985. 19:378–382.
Article32. Bauch M, Ester A, Kimura B, Victorica BE, Kedar A, Phillips MI. Atrial natriuretic peptide as a marker for doxorubicin-induced cardiotoxic effects. Cancer. 1992. 69:1492–1497.
Article33. Lako-Futo Z, Szokodi I, Sarman B, Foldes G, Tokola H, Ilves M, Leskinen H, Vuolteenaho O, Skoumal R, deChatel R, Ruskoaho H, Toth M. Evidence for a functional role of angiotensin II type 2 receptor in the cardiac hypertrophic process in vivo in the rat heart. Circulation. 2003. 108:2414–2422.
Article34. Gomez-Garre D, Guerra M, Gonzalez E, Lopez-Farre A, Riesco A, Caramelo C, Escanero J, Egido J. Aggregation of human polymorphonuclear leukocytes by endothelin: role of platelet-activating factor. Eur J Pharmacol. 1992. 224:167–172.35. Hansen PR. Role of neutrophils in myocardial ischemia and reperfusion. Circulation. 1995. 91:1872–1885.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Endothelin Antagonists on Myocardial Infarct Size after Coronary Artery Occlusion and Reperfusion in Rat
- The effects of hydrogen sulfide under sevoflurane administration against ischemia and reperfusion injury in isolated rat heart
- Effect of Rosiglitazone on Myocardial Ischemia-Reperfusion Injury in Rat Heart
- Influence of the Angiotensin II AT1 Receptor Antagonist on Reperfusion Injury in Rat Myocardial Ischemia Model
- Myocardial protective effect by ulinastatin via an anti-inflammatory response after regional ischemia/reperfusion injury in an in vivo rat heart model