Ann Lab Med.
2013 May;33(3):184-189. 10.3343/alm.2013.33.3.184.
Performance Evaluation of the OraQuick Hepatitis C Virus Rapid Antibody Test
- Affiliations
-
- 1Department of Laboratory Medicine, Chung-Ang University Healthcare System and Medical Device Clinical Trials Center, Seoul, Korea. chayoung@cau.ac.kr
- 2Department of Laboratory Medicine, Samsung Medical Center, Seoul, Korea.
- 3Department of Internal Medicine, Samsung Medical Center, Seoul, Korea.
- 4Department of Laboratory Medicine, Seoul National University Bundang Hospital, Seongnam, Korea.
- 5Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea.
- 6Hanmaeum Blood Center, Gwacheon, Korea.
- KMID: 1781323
- DOI: http://doi.org/10.3343/alm.2013.33.3.184
Abstract
- BACKGROUND
A reliable rapid assay for hepatitis C virus (HCV) may be helpful in various clinical settings. We evaluated the performance of the OraQuick HCV Rapid Antibody Test (OraSure Technologies Inc., Bethlehem, PA, USA).
METHODS
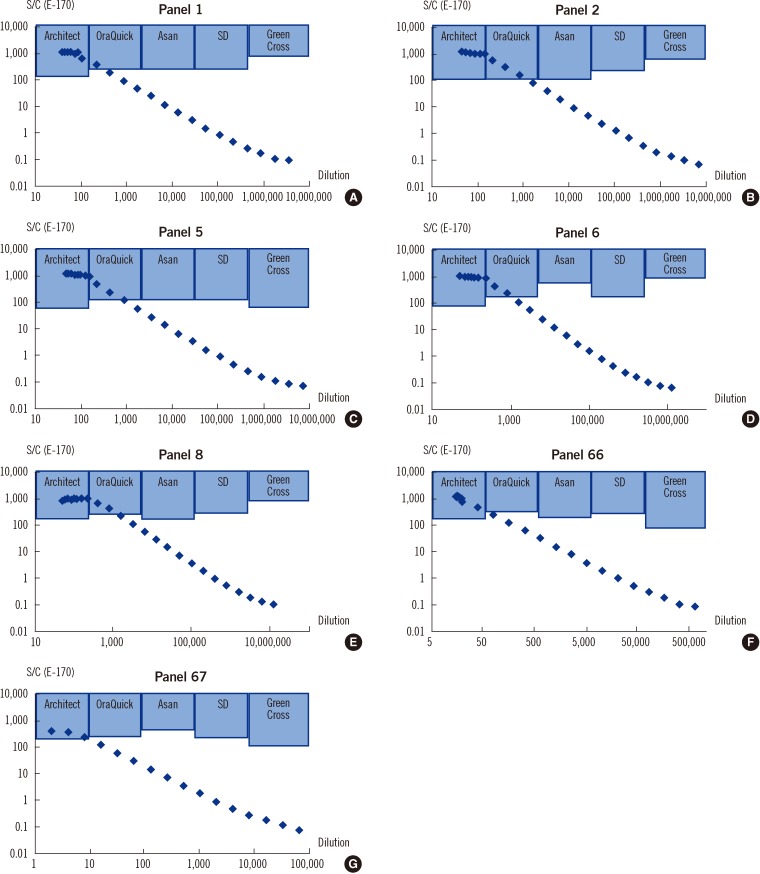
Clinical sensitivity and specificity were evaluated with oral fluids and sera from 137 patients diagnosed with hepatitis C and 300 healthy blood donors in a multi-center collaborative study. The stored sera of 200 proven HCV-infected patients and 200 healthy subjects were also evaluated. Analytical sensitivity was estimated with 4 commercial seroconversion panels and 7 Korean reference panels. The performance of 4 laboratory-based tests (3 chemiluminescence assays and 1 enzyme immunoassay) and 4 rapid test kits was compared. We also assessed the interference due to bilirubin, hemoglobin, lipid, rheumatoid factor, multipara, and several viral infections.
RESULTS
The clinical sensitivity and specificity of the OraQuick HCV test using oral fluid were 97.8% (95% confidence interval [CI], 93.2-99.4%) and 100% (95% CI, 98.4-100%), respectively. The clinical sensitivity using serum samples was 100%. Using the 4 seroconversion panels, the OraQuick HCV test showed results comparable to those of the laboratory-based assays; its analytical sensitivity was higher than that of the other rapid test kits. There was no cross-reactivity with common interfering factors.
CONCLUSIONS
The clinical performance of the OraQuick HCV Test is comparable to that of laboratory-based tests with both serum and oral fluid. This supports the supplementary use of rapid HCV testing using oral fluid in various medical and non-medical settings.
Keyword
MeSH Terms
Figure
Reference
-
1. Hepatitis C. Fact sheet No. 164. World Health Organization. http://www.who.int/mediacentre/factsheets/fs164/en/index.html (Update on Jun 2011).2. Shin HR. Epidemiology of hepatitis C virus in Korea. Intervirology. 2006; 49:18–22. PMID: 16166784.
Article3. Kim SJ. Viral hepatitis surveillance system and statue of C hepatitis sentinel surveillance in Korea. Public Health Wkly Rep. 2012; 5:214–219.4. Scheiblauer H, El-Nageh M, Nick S, Fields H, Prince A, Diaz S. Evaluation of the performance of 44 assays used in countries with limited resources for the detection of antibodies to hepatitis C virus. Transfusion. 2006; 46:708–718. PMID: 16686838.
Article5. Smith BD, Drobeniuc J, Jewett A, Branson BM, Garfein RS, Teshale E, et al. Evaluation of three rapid screening assays for detection of antibodies to hepatitis C virus. J Infect Dis. 2011; 204:825–831. PMID: 21849279.
Article6. Lee SR, Kardos KW, Schiff E, Berne CA, Mounzer K, Banks AT, et al. Evaluation of a new, rapid test for detecting HCV infection, suitable for use with blood or oral fluid. J Virol Methods. 2011; 172:27–31. PMID: 21182871.
Article7. Smith BD, Teshale E, Jewett A, Weinbaum CM, Neaigus A, Hagan H, et al. Performance of premarket rapid hepatitis C virus antibody assays in 4 national human immunodeficiency virus behavioral surveillance system sites. Clin Infect Dis. 2011; 53:780–786. PMID: 21921221.
Article8. Jewett A, Smith BD, Garfein RS, Cuevas-Mota J, Teshale EH, Weinbaum CM. Field-based performance of three pre-market rapid hepatitis C virus antibody assays in STAHR (Study to Assess Hepatitis C Risk) among young adults who inject drugs in San Diego, CA. J Clin Virol. 2012; 54:213–217. PMID: 22560051.
Article9. O'Connell RJ, Merritt TM, Malia JA, VanCott TC, Dolan MJ, Zahwa H, et al. Performance of the OraQuick rapid antibody test for diagnosis of human immunodeficiency virus type 1 infection in patients with various levels of exposure to highly active antiretroviral therapy. J Clin Microbiol. 2003; 41:2153–2155. PMID: 12734265.10. Kee KM, Wang JH, Hung CH, Chen CH, Lee CM, Chang KC, et al. Decreased anti-hepatitis C virus titer and associated factors in chronic hepatitis C patients after sustained virological response: a prospective study. J Gastroenterol Hepatol. 2012; 27:1106–1111. PMID: 22004331.
Article11. Thompson ND, Perz JF, Moorman AC, Holmberg SD. Nonhospital health care-associated hepatitis B and C virus transmission: United States, 1998-2008. Ann Intern Med. 2009; 150:33–39. PMID: 19124818.
Article12. Kim JY, Won JE, Jeong SH, Park SJ, Hwang SG, Kang SK, et al. Acute hepatitis C in Korea: different modes of infection, high rate of spontaneous recovery, and low rate of seroconversion. J Med Virol. 2011; 83:1195–1202. PMID: 21567423.
Article13. Smith BD, Morgan RL, Beckett GA, Falck-Ytter Y, Holtzman D, Teo CG, et al. Recommendations for the identification of chronic hepatitis C virus infection among persons born during 1945-1965. MMWR Recomm Rep. 2012; 61:1–32. PMID: 22895429.14. Hepatitis C virus transmission at an outpatient hemodialysis unit--New York, 2001-2008. MMWR Morb Mortal Wkly Rep. 2009; 58:189–194. PMID: 19265779.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Evaluation of the OraQuick HCV Rapid Antibody Test as a Screening Test for Hepatitis C Virus Infection
- HIV Screening Using the OraQuick(R) ADVANCE(TM) Rapid HIV-1/2 Antibody Test (OraQuick Test) in Dentistry: A Literature Review
- Rapid Diagnosis of Human Immunodeficiency Virus Infection Using OraQuick(r) Advance Rapid HIV-1/2 Antibody Test
- Evaluation of OraQuick Advance Rapid HIV-1/2 Antibody Test as a Screening Test for HIV Infection
- Prevention of Viral Hepatitis and Vaccination