Yonsei Med J.
2010 Nov;51(6):838-844. 10.3349/ymj.2010.51.6.838.
Ethyl Pyruvate Has Anti-Inflammatory and Delayed Myocardial Protective Effects after Regional Ischemia/Reperfusion Injury
- Affiliations
-
- 1Department of Cardiothoracic and Vascular Surgery, Institute of Health Sciences, Gyeongsang National University, Jinju, Korea.
- 2Department of Anesthesiology and Pain Medicine, Institute of Health Sciences, Gyeongsang National University, Jinju, Korea. ilwooshin@gnu.ac.kr
- KMID: 1779628
- DOI: http://doi.org/10.3349/ymj.2010.51.6.838
Abstract
- PURPOSE
Ethyl pyruvate has anti-inflammatory properties and protects organs from ischemia/reperfusion (I/R)-induced tissue injury. The aim of this study was to determine whether ethyl pyruvate decreases the inflammatory response after regional I/R injury and whether ethyl pyruvate protects against delayed regional I/R injury in an in vivo rat heart model after a 24 hours reperfusion.
MATERIALS AND METHODS
Rats were randomized to receive lactated Ringer's solution or ethyl pyruvate dissolved in Ringer's solution, which was given by intraperitoneal injection 1 hour prior to ischemia. Rats were subjected to 30 min of ischemia followed by reperfusion of the left coronary artery territory. After a 2 hours reperfusion, nuclear factor kappaB, myocardial myeloperoxidase activity, and inflammatory cytokine levels were determined. After the 24 hours reperfusion, the hemodynamic function and myocardial infarct size were evaluated.
RESULTS
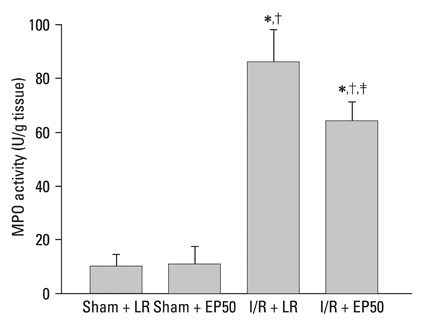
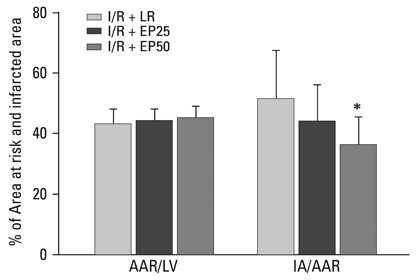
At 2 hours after I/R injury, ethyl pyruvate attenuated I/R-induced nuclear factor kappaB translocation and reduced myeloperoxidase activity in myocardium. The plasma circulating levels of inflammatory cytokines decreased significantly in the ethyl pyruvate-treated group. At 24 hours after I/R injury, ethyl pyruvate significantly improved cardiac function and reduced infarct size after regional I/R injury.
CONCLUSION
Ethyl pyruvate has the ability to inhibit neutrophil activation, inflammatory cytokine release, and nuclear factor kappaB translocation. Ethyl pyruvate is associated with a delayed myocardial protective effect after regional I/R injury in an in vivo rat heart model.
Keyword
MeSH Terms
-
Animals
Anti-Inflammatory Agents/*pharmacology
Cell Nucleus/metabolism
Cytoplasm/metabolism
Heart/physiopathology
Inflammation
Male
Myocardial Infarction/prevention & control
Myocardium/*metabolism
NF-kappa B/metabolism
Peroxidase/metabolism
Pyruvates/*pharmacology
Rats
Rats, Sprague-Dawley
Reperfusion Injury/*drug therapy/*metabolism
Figure
Cited by 2 articles
-
Myocardial protective effect by ulinastatin via an anti-inflammatory response after regional ischemia/reperfusion injury in an in vivo rat heart model
Il-Woo Shin, In-Seok Jang, Seung-Min Lee, Kyeong-Eon Park, Seong-Ho Ok, Ju-Tae Sohn, Heon-Keun Lee, Young-Kyun Chung
Korean J Anesthesiol. 2011;61(6):499-505. doi: 10.4097/kjae.2011.61.6.499.Lipofundin® MCT/LCT 20% increase left ventricular systolic pressure in an
ex vivo rat heart model via increase of intracellular calcium level
Jiyoung Park, Yeon A Kim, Jeong Yeol Han, Sangkyu Jin, Seong-Ho Ok, Ju-Tae Sohn, Heon-Keun Lee, Young-Kyun Chung, Il-Woo Shin
Korean J Anesthesiol. 2016;69(1):57-62. doi: 10.4097/kjae.2016.69.1.57.
Reference
-
1. Mallet RT, Sun J, Knott EM, Sharma AB, Olivencia-Yurvati AH. Metabolic cardioprotection by pyruvate: recent progress. Exp Biol Med (Maywood). 2005. 230:435–443.
Article2. Brand KA, Hermfisse U. Aerobic glycolysis by proliferating cells: a protective strategy against reactive oxygen species. FASEB J. 1997. 11:388–395.
Article3. Dobsak P, Courderot-Masuyer C, Zeller M, Vergely C, Laubriet A, Assem M, et al. Antioxidative properties of pyruvate and protection of the ischemic rat heart during cardioplegia. J Cardiovasc Pharmacol. 1999. 34:651–659.
Article4. Yu YM, Kim JB, Lee KW, Kim SY, Han PL, Lee JK. Inhibition of the cerebral inchemic injury by ethyl pyruvate with a wide therapeutic window. Stroke. 2005. 36:2238–2243.
Article5. Cicalese L, Lee K, Schraut W, Watkins S, Borle A, Stanko R. Pyruvate prevents ischemia-reperfusion mucosal injury of rat small intestine. Am J Surg. 1996. 171:97–100.6. Sileri P, Schena S, Morini S, Rastellini C, Pham S, Benedetti E, et al. Pyruvate inhibits hepatic ischemia-reperfusion injury in rats. Transplantation. 2001. 72:27–30.
Article7. Fink MP. Ethyl pyruvate. Curr Opin Anaesthesiol. 2008. 21:160–167.
Article8. Taylor MD, Grand TJ, Cohen JE, Hsu V, Liao GP, Zentko S, et al. Ethyl pyruvate enhances ATP levels, reduces oxidative stress and preserves cardiac function in a rat model of off-pump coronary bypass. Heart Lung Circ. 2005. 14:25–31.
Article9. Woo YJ, Taylor MD, Cohen JE, Jayasankar V, Bish LT, Burdick J, et al. Ethyl pyruvate preserves cardiac function and attenuates oxidative injury after prolonged myocardial ischemia. J Thorac Cardiovasc Surg. 2004. 127:1262–1269.
Article10. Kim HS, Cho IH, Kim JE, Shin YJ, Jeon JH, Kim Y, et al. Ethyl pyruvate has an anti-inflammatory effect by inhibiting ROS-dependent STAT signaling in activated microglia. Free Radic Biol Med. 2008. 45:950–963.11. Kim JB, Yu YM, Kim SW, Lee JK. Anti-inflammatory mechanism is involved in ethyl pyruvate-mediated efficacious neuroprotection in the postischemic brain. Brain Res. 2005. 1060:188–192.12. Yang R, Han X, Delude RL, Fink MP. Ethyl pyruvate ameliorates acute alcohol-induced liver injury and inflammation in mice. J Lab Clin Med. 2003. 142:322–331.
Article13. Shin IW, Jang IS, Lee SH, Baik JS, Park KE, Sohn JT, et al. Propofol has delayed myocardial protective effects after a regional ischemia/reperfusion injury in an in vivo rat heart model. Korean J Anesthesiol. 2010. 58:378–382.
Article14. Jin YC, Kim CW, Kim YM, Nizamutdinova IT, Ha YM, Kim HJ, et al. Cryptotanshinone, a lipophilic compound of Salvia miltiorrhiza root, inhibits TNF-alpha-induced expression of adhesion molecules in HUVEC and attenuates rat myocardial ischemia/ reperfusion injury in vivo. Eur J Pharmacol. 2009. 614:91–97.
Article15. Gwechenberger M, Mendoza LH, Youker KA, Frangogiannis NG, Smith CW, Michael LH, et al. Cardiac myocytes produce interleukin-6 in culture and in viable border zone of reperfused infarctions. Circulation. 1999. 99:546–551.
Article16. Ulloa L, Ochani M, Yang H, Tanovic M, Halperin D, Yang R, et al. Ethyl pyruvate prevents lethality in mice with established lethal sepsis and systemic inflammation. Proc Natl Acad Sci U S A. 2002. 99:12351–12356.
Article17. Zingarelli B, Hake PW, Yang Z, O'Connor M, Denenberg A, Wong HR. Absence of inducible nitric oxide synthase modulates early reperfusion-induced NF-kappaB and AP-1 activation and enhances myocardial damage. FASEB J. 2002. 16:327–342.18. Chandrasekar B, Freeman GL. Induction of nuclear factor kappaB and activation protein 1 in postischemic myocardium. FEBS Lett. 1997. 401:30–34.
Article19. Barnes PJ, Karin M. Nuclear factor-kappaB; a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med. 1997. 336:1066–1071.
Article20. Han Y, Englert JA, Yang R, Delude RL, Fink MP. Ethyl pyruvate inhibits nuclear factor-kappaB-dependent signaling by directly targeting p65. J Pharmacol Exp Ther. 2005. 312:1097–1105.
Article21. Tsung A, Kaizu T, Nakao A, Shao L, Bucher B, Fink MP, et al. Ethyl pyruvate ameliorates liver ischemia-reperfusion injury by decreasing hepatic necrosis and apoptosis. Transplantation. 2005. 79:196–204.
Article22. Reade MC, Fink MP. Bench-to-bedside review: Amelioration of acute renal impairment using ethyl pyruvate. Crit Care. 2005. 9:556–560.23. Hansen PR. Role of neutrophils in myocardial ischemia and reperfusion. Circulation. 1995. 91:1872–1885.
Article24. Frangogiannis NG, Smith CW, Entman ML. The inflammatory response in myocardial infarction. Cardiovasc Res. 2002. 53:31–47.
Article25. Goldmann BU, Rudolph V, Rudolph TK, Holle AK, Hillebrandt M, Meinertz T, et al. Neutrophil activation precedes myocardial injury in patients with acute myocardial infarction. Free Radic Biol Med. 2009. 47:79–83.
Article26. Zhao ZQ, Nakamura M, Wang NP, Velez DA, Hewan-Lowe KO, Guyton RA, et al. Dynamic progression of contractile and endothelial dysfunction and infarct extension in the late phase of reperfusion. J Surg Res. 2000. 94:133–144.
Article27. Zhao ZQ, Velez DA, Wang NP, Hewan-Lowe KO, Nakamura M, Guyton RA, et al. Progressively developed myocardial apoptotic cell death during late phase of reperfusion. Apoptosis. 2001. 6:279–290.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Myocardial protective effect by ulinastatin via an anti-inflammatory response after regional ischemia/reperfusion injury in an in vivo rat heart model
- Ethyl Pyruvate Ameliorates Renal Ischemia- reperfusion Injury
- Propofol has delayed myocardial protective effects after a regional ischemia/reperfusion injury in an in vivo rat heart model
- Flavonoids as therapeutics for myocardial ischemia-reperfusion injury: a comprehensive review on preclinical studies
- The Role of HMGB1 and the Effect of Ethyl Pyruvate on Ischemia and Reperfusion Injury of Rat Kidney