J Korean Med Sci.
2013 Dec;28(12):1774-1780. 10.3346/jkms.2013.28.12.1774.
Change in Somatostatinergic Tone of Acromegalic Patients according to the Size of Growth Hormone-Producing Pituitary Tumors
- Affiliations
-
- 1Department of Endocrinology and Metabolism, Kyung Hee University School of Medicine, Seoul, Korea. igf1@unitel.co.kr
- KMID: 1779419
- DOI: http://doi.org/10.3346/jkms.2013.28.12.1774
Abstract
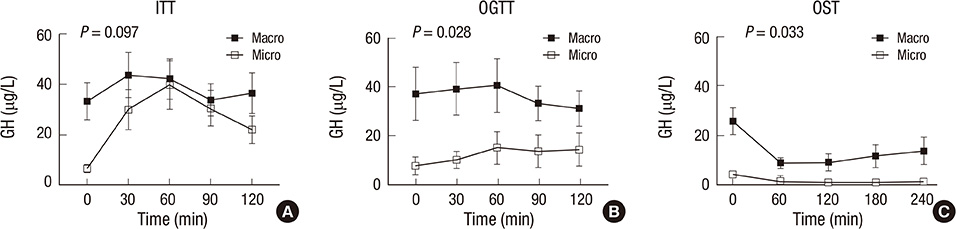
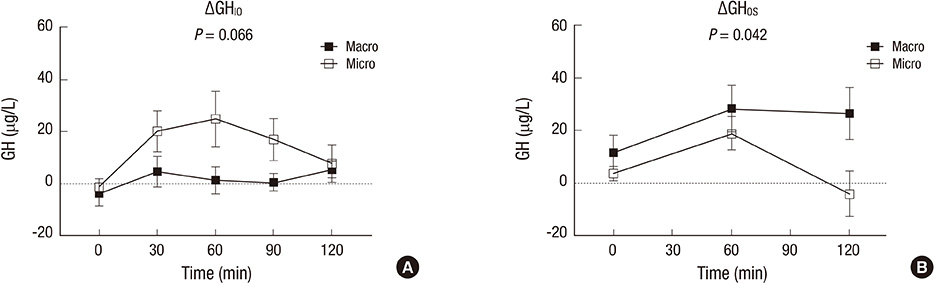
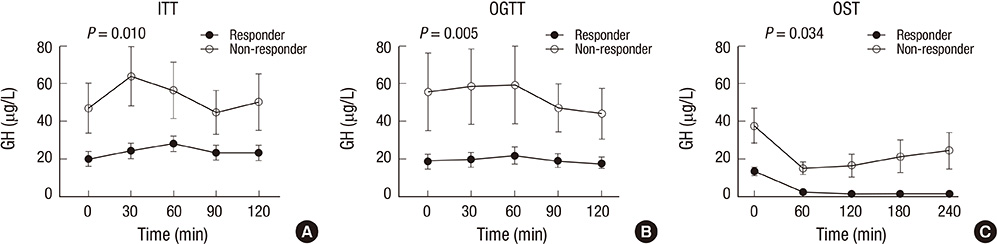
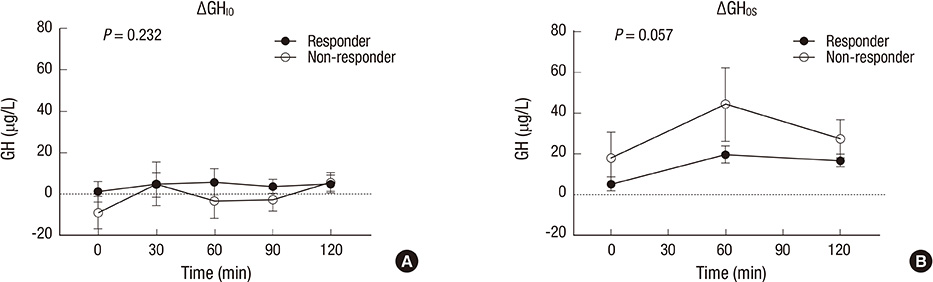
- The aim of this study was to investigate the relationship between somatostatinergic tone (SST) and the size of growth hormone (GH)-producing pituitary tumors. GH levels of 29 patients with newly diagnosed acromegaly were measured using a 75-gram oral glucose tolerance test (OGTT), an insulin tolerance test (ITT), and an octreotide suppression test (OST). Differences between GH levels during the ITT and the OGTT (DeltaGH(IO)), and between the OGTT and the OST at the same time point (DeltaGH(OS)) were compared according to the size of the tumor and the response pattern to the OST. DeltaGH(IO) of macroadenomas (n=22) was non-significantly higher than those of microadenomas while DeltaGH(OS) of macroadenomas were significantly higher than those of microadenomas. According to further analyses of macroadenomas based on the response pattern to the OST, GH levels during the ITT were significantly higher in non-responders. DeltaGH(OS) showed near-significant differences between responders and non-responders. In conclusion, as the size of the pituitary tumor increases, the effect of glucose on SST appears to be attenuated. Macroadenomas that are non-responders to the OST possess a portion of GH secretion exceeding the range of regulation by SST.
MeSH Terms
-
Acromegaly/*diagnosis/*pathology
Adenoma/drug therapy/*pathology
Adult
Aged
Antineoplastic Agents, Hormonal/therapeutic use
Female
Glucose Tolerance Test
Human Growth Hormone/*blood/secretion
Humans
Insulin/blood
Insulin-Like Growth Factor I/analysis
Male
Middle Aged
Octreotide/therapeutic use
Pituitary Neoplasms/drug therapy/*pathology
Antineoplastic Agents, Hormonal
Human Growth Hormone
Insulin
Insulin-Like Growth Factor I
Octreotide
Figure
Reference
-
1. Lissett CA, Shalet SM. Management of pituitary tumours: strategy for investigation and follow-up. Horm Res. 2000; 53:65–70.2. Mori R, Inoshita N, Takahashi-Fujigasaki J, Joki T, Nishioka H, Abe T, Fujii T, Yamada S. Clinicopathological features of growth hormone-producing pituitary adenomas in 242 acromegaly patients: classification according to hormone production and cytokeratin distribution. ISRN Endocrinol. 2013; 2013:723432.3. Lamberts SW, Uitterlinden P, Schuijff PC, Klijn JG. Therapy of acromegaly with sandostatin: the predictive value of an acute test, the value of serum somatomedin-C measurements in dose adjustment and the definition of a biochemical 'cure'. Clin Endocrinol (Oxf). 1988; 29:411–420.4. Colao A, Ferone D, Lastoria S, Marzullo P, Cerbone G, Di Sarno A, Longobardi S, Merola B, Salvatore M, Lombardi G. Prediction of efficacy of octreotide therapy in patients with acromegaly. J Clin Endocrinol Metab. 1996; 81:2356–2362.5. Giustina A, Veldhuis JD. Pathophysiology of the neuroregulation of growth hormone secretion in experimental animals and the human. Endocr Rev. 1998; 19:717–797.6. Park C, Yang I, Woo J, Kim S, Kim J, Kim Y, Park S. Acute hyperglycemia and activation of the beta-adrenergic system exhibit synergistic inhibitory actions on growth hormone (GH) releasing hormone-induced GH release. Eur J Endocrinol. 2003; 148:635–640.7. Hartman ML, Pincus SM, Johnson ML, Matthews DH, Faunt LM, Vance ML, Thorner MO, Veldhuis JD. Enhanced basal and disorderly growth hormone secretion distinguish acromegalic from normal pulsatile growth hormone release. J Clin Invest. 1994; 94:1277–1288.8. Petersenn S, Quabbe HJ, Schöfl C, Stalla GK, von Werder K, Buchfelder M. The rational use of pituitary stimulation tests. Dtsch Arztebl Int. 2010; 107:437–443.9. Gilbert JA, Miell JP, Chambers SM, McGregor AM, Aylwin SJ. The nadir growth hormone after an octreotide test dose predicts the long-term efficacy of somatostatin analogue therapy in acromegaly. Clin Endocrinol (Oxf). 2005; 62:742–747.10. Carrozza C, Lapolla R, Canu G, Annunziata F, Torti E, Baroni S, Zuppi C. Human growth hormone (GH) immunoassay: standardization and clinical implications. Clin Chem Lab Med. 2011; 49:851–853.11. Hardy J. Transsphenoidal surgery of hypersecreting pituitary tumors. In : Kohler PO, Ross GT, editors. The diagnosis and treatment of pituitary tumors. Amsterdam: Excerpta Medica;1973. p. 179–188.12. Cryer PE. Diverse causes of hypoglycemia-associated autonomic failure in diabetes. N Engl J Med. 2004; 350:2272–2279.13. Mancini A, Zuppi P, Fiumara C, Valle D, Conte G, Fabrizi ML, Sammartano L, Anile C, Maira G, De Marinis L. GH response to oral and intravenous glucose load in acromegalic patients. Horm Metab Res. 1995; 27:322–325.14. Bruns C, Lewis I, Briner U, Meno-Tetang G, Weckbecker G. SOM230: a novel somatostatin peptidomimetic with broad somatotropin release inhibiting factor (SRIF) receptor binding and a unique antisecretory profile. Eur J Endocrinol. 2002; 146:707–716.15. Reubi JC, Landolt AM. The growth hormone responses to octreotide in acromegaly correlate with adenoma somatostatin receptor status. J Clin Endocrinol Metab. 1989; 68:844–850.16. Ghigo E, Bellone J, Aimaretti G, Bellone S, Loche S, Cappa M, Bartolotta E, Dammacco F, Camanni F. Reliability of provocative tests to assess growth hormone secretory status: study in 472 normally growing children. J Clin Endocrinol Metab. 1996; 81:3323–3327.17. Aimaretti G, Corneli G, Razzore P, Bellone S, Baffoni C, Arvat E, Camanni F, Ghigo E. Comparison between insulin-induced hypoglycemia and growth hormone (GH)-releasing hormone + arginine as provocative tests for the diagnosis of GH deficiency in adults. J Clin Endocrinol Metab. 1998; 83:1615–1618.18. Blake AD, Smith RG. Desensitization studies using perifused rat pituitary cells show that growth hormone-releasing hormone and His-D-Trp-Ala-Trp-D-Phe-Lys-NH2 stimulate growth hormone release through distinct receptor sites. J Endocrinol. 1991; 129:11–19.19. Mayr B, Buslei R, Theodoropoulou M, Stalla GK, Buchfelder M, Schöfl C. Molecular and functional properties of densely and sparsely granulated GH-producing pituitary adenomas. Eur J Endocrinol. 2013; 169:391–400.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Relationship between the Expression of Growth Hormone-Releasing Hormone Receptor Gene and Endocrinologic Profiles in GH-Secreting Pituitary Adenomas
- Comparison of Anterior Pituitary Function between Patients with GH-secreting Macroadenoma and those with Nonfunctioning Macroadenoma
- An Approach for Endotracheal Intubation in Acromegaly Patient
- Analysis of Factors Affecting Surgical Outcome in Acromegaly
- The Role of Gamma Knife Radiosurgery for the Treatment of Pituitary Adenomas