J Korean Med Sci.
2013 Oct;28(10):1474-1481. 10.3346/jkms.2013.28.10.1474.
S-Fas Urinary Excretion Helps to Predict the Immunosuppressive Treatment Outcomes in Patients with Proliferative Primary Glomerulonephritis
- Affiliations
-
- 1Department of Kidney Transplantation, Medical University of Lodz, Lodz, Poland. rzwiech@mp.pl
- KMID: 1777684
- DOI: http://doi.org/10.3346/jkms.2013.28.10.1474
Abstract
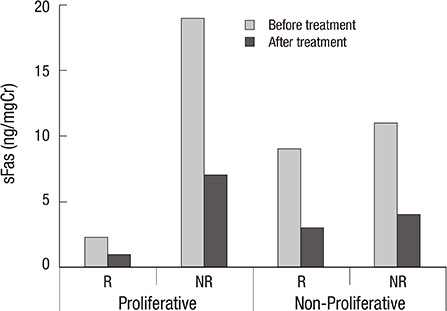
- Deregulation of soluble apoptosis stimulating fragment (sFas) plays an important role in glomerulonephritis (GN). The study assed the influence of immunosuppressive treatment on serum and urine sFas in patients with proliferative (PGN) and non-proliferative (NPGN) GN, and evaluated the potential of sFas measurements in predicting outcomes. Eighty-four patients with GN (45 males and 39 females) were included. Serum concentration (ng/mL) and urinary excretion (ng/mg of urinary creatinine) of sFas were measured before and after the treatment. After 12 months of therapy with steroids and cyclophosphamide, patients were divided into two subgroups according to the treatment results: Responders (R) and Non-Responders (NR). The sFas urinary excretion was reduced after treatment in both PGN and NPGN (from 17.12 +/- 15 to 5.3 +/- 4.2, P = 0.008 and from 10.11 +/- 6.1 to 3.4 +/- 3.0, P = 0.039; respectively) whereas the sFas serum concentration remained unchanged. In PGN, pre-treatment urinary sFas concentration was significantly lower in the Responders than in Non-Responders (2.3 +/- 3.1 vs 19.4 +/- 14.1, P = 0.003), and was lower still than in both R (P = 0.044) and NR (P = 0.042) subgroups with NPGN. The immunosuppressive treatment reduced sFas urinary excretion in proliferative and non-proliferative GN and results suggest that the lower urinary sFas may be linked with favorable therapy outcomes in patients with PGN.
MeSH Terms
Figure
Reference
-
1. Sato M, Konuma T, Yanagisawa N, Haizuka H, Asakura H, Nakashima Y. Fas-Fas ligand system in the peripheral blood of patients with renal diseases. Nephron. 2000; 85:107–113.2. Cheng J, Zhou T, Liu C, Shapiro JP, Brauer MJ, Kiefer MC, Barr PJ, Mountz JD. Protection from Fas-mediated apoptosis by a soluble form of the Fas molecule. Science. 1994; 263:1759–1762.3. Fujinaka H, Yamamoto T, Feng L, Nameta M, Garcia G, Chen S, El-shemi AA, Ohshiro K, Katsuyama K, Yoshida Y, et al. Anti-perforin antibody treatment ameliorates experimental crescentic glomerulonephritis in WKY rats. Kidney Int. 2007; 72:823–830.4. Lorz C, Ortiz A, Justo P, González-Cuadrado S, Duque N, Gómez-Guerrero C, Egido J. Proapoptotic Fas ligand is expressed by normal kidney tubular epithelium and injured glomeruli. J Am Soc Nephrol. 2000; 11:1266–1277.5. Remuzzi G, Bertani T. Is glomerulosclerosis a consequence of altered glomerular permeability to macromolecules? Kidney Int. 1990; 38:384–394.6. Couser WG. Glomerulonephritis. Lancet. 1999; 353:1509–1515.7. Chatziantoniou C, Boffa JJ, Tharaux PL, Flamant M, Ronco P, Dussaule JC. Progression and regression in renal vascular and glomerular fibrosis. Int J Exp Pathol. 2004; 85:1–11.8. Strutz F, Zeisberg M. Renal fibroblasts and myofibroblasts in chronic kidney disease. J Am Soc Nephrol. 2006; 17:2992–2998.9. Coppo R, Ponticelli C. Minimal change nephropathy. In : Ponticelli C, Glassock RJ, editors. Treatment of primary glomerulonephritis. 2nd ed. Oxford: Oxford University Press;2009. p. 179–210.10. Scolari F, Ponticelli C. Focal and segmental glomerulosclerosis. In : Ponticelli C, Glassock RJ, editors. Treatment of primary glomerulonephritis. 2nd ed. Oxford: Oxford University Press;2009. p. 215–256.11. Passerini P, Ponticelli C. Membranous nephropathy. In : Ponticelli C, Glassock RJ, editors. Treatment of primary glomerulonephritis. 2nd ed. Oxford: Oxford University Press;2009. p. 261–308.12. Glassock RJ, Lee G. Immunoglobulin A nephropathy. In : Ponticelli C, Glassock RJ, editors. Treatment of primary glomerulonephritis. 2nd ed. Oxford: Oxford University Press;2009. p. 313–361.13. Glassock RJ. Membranoprolipherative glomerulonephritis. In : Ponticelli C, Glassock RJ, editors. Treatment of primary glomerulonephritis. 2nd ed. Oxford: Oxford University Press;2009. p. 375–393.14. D'Amico G. Tubulo-interstitial damage in glomerular diseases: its role in the progression of the renal damage. Nephrol Dial Transplant. 1998; 13:80–85.15. Okoń K, Sułowicz W, Smoleński O, Sydor A, Chruściel B, Kirker-Nowak A, Rosiek Z, Sysło K, Stachura J. Predicting kidney function from renal biopsy: semiquantitative versus quantitative approach. Pol J Pathol. 2007; 58:65–71.16. Fuiano G, Sund S, Mazza G, Rosa M, Caglioti A, Gallo G, Natale G, Andreucci M, Memoli B, De Nicola L, et al. Renal hemodynamic response to maximal vasodilating stimulus in healthy older subjects. Kidney Int. 2001; 59:1052–1058.17. Glassock RJ, Cohen AH. The primary glomerulopathies. Dis Mon. 1996; 42:329–383.18. Ogihara T, Kikuchi K, Matsuoka H, Fujita T, Higaki J, Horiuchi M, Imai Y, Imaizumi T, Ito S, Iwao H, et al. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2009). Hypertens Res. 2009; 32:3–107.19. Fujita T, Ando K, Nishimura H, Ideura T, Yasuda G, Isshiki M, Takahashi K. Cilnidipine versus Amlodipine Randomised Trial for Evaluation in Renal Desease(CARTER) Study Investigators. Antiproteinuric effect of the calcium channel blocker cilnidipine added to renin-angiotensin inhibition in hypertensive patients with chronic renal disease. Kidney Int. 2007; 72:1543–1549.20. National Kidney Foundation. K/DOQI clinical practice guidelines for managing dyslipidemias in chronic kidney disease. Am J Kidney Dis. 2003; 41:S1–S91.21. Yuan J, Fang W, Zhang W, Ni Z, Qian J. Treatment of nephrotic idiopathic membranous nephropathy with monthly i.v. pulse cyclophosphamide and oral steroids: a single centre's retrospective study. Nephrology (Carlton). 2011; 16:440–445.22. Menon S, Valentini RP. Membranous nephropathy in children: clinical presentation and therapeutic approach. Pediatr Nephrol. 2010; 25:1419–1428.23. Cucer F, Miron I, Müller R, Iliescu Halitchi C, Mihaila D. Treatment with Cyclophosphamide for steroid-resistant nephrotic syndrome in children. Maedica (Buchar). 2010; 5:167–170.24. Van Husen M, Kemper MJ. New therapies in steroid-sensitive and steroid-resistant idiopathic nephrotic syndrome. Pediatr Nephrol. 2011; 26:881–892.25. Shenoy M, Plant ND, Lewis MA, Bradbury MG, Lennon R, Webb NJ. Intravenous methylprednisolone in idiopathic childhood nephrotic syndrome. Pediatr Nephrol. 2010; 25:899–903.26. Mitra N, Abbas M, Nematollah A. Correlation between prognosis and response to treatment in children with FSGS. Acta Med Iran. 2009; 47:93–96.27. Sümegi V, Haszon I, Bereczki C, Papp F, Túri S. Long-term follow-up after cyclophosphamide and cyclosporine-A therapy in steroid-dependent and -resistant nephrotic syndrome. Pediatr Nephrol. 2008; 23:1085–1092.28. Kawasaki Y. The pathogenesis and treatment of pediatric Henoch-Schönlein purpura nephritis. Clin Exp Nephrol. 2011; 15:648–657.29. Edström Halling S, Söderberg MP, Berg UB. Treatment of severe Henoch-Schönlein and immunoglobulin A nephritis: a single center experience. Pediatr Nephrol. 2009; 24:91–97.30. Oshima S, Kawamura O. Long-term follow-up of patients with IgA nephropathy treated with prednisolone and cyclophosphamide therapy. Clin Exp Nephrol. 2008; 12:264–269.31. Gulati S, Sharma AP, Sharma RK, Gupta A, Gupta RK. Do current recommendations for kidney biopsy in nephrotic syndrome need modifications? Pediatr Nephrol. 2002; 17:404–408.32. Yagi K, Yanagida H, Sugimoto K, Kuwajima H, Tabata N, Morita K, Okada M, Takemura T. Clinicopathologic features, outcome, and therapeutic interventions in four children with isolated C3 mesangial proliferative glomerulonephritis. Pediatr Nephrol. 2005; 20:1273–1278.33. Kobayashi Y, Tateno S, Hiki Y, Shigematsu H. IgA nephropathy: prognostic significance of proteinuria and histological alterations. Nephron. 1983; 34:146–153.34. McQuarrie EP, Shakerdi L, Jardine AG, Fox JG, Mackinnon B. Fractional excretions of albumin and IgG are the best predictors of progression in primary glomerulonephritis. Nephrol Dial Transplant. 2011; 26:1563–1569.35. Shou I, Tashiro K, Kurusu A, Kaneko S, Hayashi T, Fukui M, Shirato I, Kubota K, Tomino Y. Serum levels of soluble Fas and disease activity in patients with IgA nephropathy. Nephron. 1999; 81:387–392.36. Sano H, Asano K, Minatoguchi S, Hiraoka J, Fujisawa K, Nishigaki K, Yasuda N, Kumada H, Takemura M, Ohashi H, et al. Plasma soluble fas and soluble fas ligand in chronic glomerulonephritis. Nephron. 1998; 80:153–161.37. Kacprzyk F. Serum level and urinary excretion of soluble Fas (sFas) in patients with primary glomerulopathies. Pol Arch Med Wewn. 2002; 108:843–847.38. Polanco N, Gutiérrez E, Covarsí A, Ariza F, Carreño A, Vigil A, Baltar J, Fernández-Fresnedo G, Martín C, Pons S, et al. Spontaneous remission of nephrotic syndrome in idiopathic membranous nephropathy. J Am Soc Nephrol. 2010; 21:697–704.39. Berthoux F, Mohey H, Laurent B, Mariat C, Afiani A, Thibaudin L. Predicting the risk for dialysis or death in IgA nephropathy. J Am Soc Nephrol. 2011; 22:752–761.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Predictive Value of Conjointly Examined IL-1ra, TNF-R I, TNF-R II, and RANTES in Patients with Primary Glomerulonephritis
- Assessment of urinary endothelin-1 and nitric oxide levels and their relationship with clinical and pathologic types in primary glomerulonephritis
- Effect of Losartan Treatment on Proteinuria and Urinary Transforming Growth Factor-beta1 in Human Chronic Glomerulonephritis
- Significance of Urinary Interleukin 6 Level in Mesangial Proliferative Glomerulonephritis in Children
- Two Cases of Nephrolithiasis Following Administration of Cyclosporine