J Korean Med Sci.
2013 Nov;28(11):1650-1656. 10.3346/jkms.2013.28.11.1650.
Clinical Features of Congenital Adrenal Insufficiency Including Growth Patterns and Significance of ACTH Stimulation Test
- Affiliations
-
- 1Department of Pediatrics, Dankook University College of Medicine, Cheonan, Korea. dryujs@dankook.ac.kr
- 2Medical Genetics Center, Asan Medical Center Children's Hospital, University of Ulsan College of Medicine, Seoul, Korea.
- KMID: 1777664
- DOI: http://doi.org/10.3346/jkms.2013.28.11.1650
Abstract
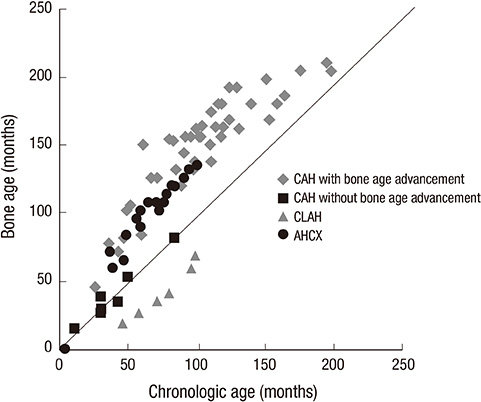
- Congenital adrenal insufficiency is caused by specific genetic mutations. Early suspicion and definite diagnosis are crucial because the disease can precipitate a life-threatening hypovolemic shock without prompt treatment. This study was designed to understand the clinical manifestations including growth patterns and to find the usefulness of ACTH stimulation test. Sixteen patients with confirmed genotyping were subdivided into three groups according to the genetic study results: congenital adrenal hyperplasia due to 21-hydroxylase deficiency (CAH, n=11), congenital lipoid adrenal hyperplasia (n=3) and X-linked adrenal hypoplasia congenita (n=2). Bone age advancement was prominent in patients with CAH especially after 60 months of chronologic age (n=6, 67%). They were diagnosed in older ages in group with bone age advancement (P<0.05). Comorbid conditions such as obesity, mental retardation, and central precocious puberty were also prominent in this group. In conclusion, this study showed the importance of understanding the clinical symptoms as well as genetic analysis for early diagnosis and management of congenital adrenal insufficiency. ACTH stimulation test played an important role to support the diagnosis and serum 17-hydroxyprogesterone levels were significantly elevated in all of the CAH patients. The test will be important for monitoring growth and puberty during follow up of patients with congenital adrenal insufficiency.
Keyword
MeSH Terms
-
17-alpha-Hydroxyprogesterone/blood
46, XY Disorders of Sex Development/drug therapy/*genetics
Adolescent
Adrenal Hyperplasia, Congenital/drug therapy/*genetics
Adrenal Insufficiency/*congenital/diagnosis/drug therapy/genetics
Adrenocorticotropic Hormone/*metabolism
Bone Development/genetics
Child
Child, Preschool
DAX-1 Orphan Nuclear Receptor/genetics
Female
Genetic Diseases, X-Linked/drug therapy/*genetics
Genotype
Glucocorticoids/therapeutic use
Humans
Intellectual Disability/complications
Male
Mineralocorticoids/therapeutic use
Obesity/complications
Phosphoproteins/genetics
Puberty, Precocious/complications
Retrospective Studies
Steroid 21-Hydroxylase/genetics
17-alpha-Hydroxyprogesterone
Adrenocorticotropic Hormone
Adrenal Insufficiency, Congenital
DAX-1 Orphan Nuclear Receptor
Glucocorticoids
Mineralocorticoids
Phosphoproteins
Steroid 21-Hydroxylase
Figure
Reference
-
1. Perrin CW. Adrenocortical insufficiency. In : Kliegman RM, Stanton BF, Schor NF, Geme J, Behrman RE, editors. Nelson textbook of pediatrics. 19th ed. Philadelphia: Saunders Elsevier;2011. p. 1924–1930.2. Speiser PW, Azziz R, Baskin LS, Ghizzoni L, Hensle TW, Merke DP, Meyer-Bahlburg HF, Miller WL, Montori VM, Oberfield SE, et al. Congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2010; 95:4133–4160.3. Kim JM, Choi JH, Lee JH, Kim GH, Lee BH, Kim HS, Shin JH, Shin CH, Kim CJ, Yu J, et al. High allele frequency of the p.Q258X mutation and identification of a novel mis-splicing mutation in the STAR gene in Korean patients with congenital lipoid adrenal hyperplasia. Eur J Endocrinol. 2011; 165:771–778.4. Zanaria E, Muscatelli F, Bardoni B, Strom TM, Guioli S, Guo W, Lalli E, Moser C, Walker AP, McCabe ER, et al. An unusual member of the nuclear hormone receptor superfamily responsible for X-linked adrenal hypoplasia congenita. Nature. 1994; 372:635–641.5. White PC. Neonatal screening for congenital adrenal hyperplasia. Nat Rev Endocrinol. 2009; 5:490–498.6. Oelkers W, Diederich S, Bähr V. Diagnosis and therapy surveillance in Addison's disease: rapid adrenocorticotropin (ACTH) test and measurement of plasma ACTH, renin activity, and aldosterone. J Clin Endocrinol Metab. 1992; 75:259–264.7. Greulich WW, Pyle SI. Radiographic atlas of skeletal development of the hand and wrist. 2nd ed. Stanford: Standford Univ. Press;1959.8. Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity and trends in body mass index among US children and adolescents, 1999-2010. JAMA. 2012; 307:483–490.9. Youngs EL, Henkhaus R, Hellings JA, Butler MG. IL1RAPL1 gene deletion as a cause of X-linked intellectual disability and dysmorphic features. Eur J Med Genet. 2012; 55:32–36.10. Fujieda K, Tajima T. Molecular basis of adrenal insufficiency. Pediatr Res. 2005; 57:62R–69R.11. Perry R, Kecha O, Paquette J, Huot C, Van Vliet G, Deal C. Primary adrenal insufficiency in children: twenty years experience at the Sainte-Justine Hospital, Montreal. J Clin Endocrinol Metab. 2005; 90:3243–3250.12. Han YJ, Lee DH, Kim JW. Newborn screening in Korea. Southeast Asian J Trop Med Public Health. 2003; 34:81–84.13. Lee DH. Newborn screening of inherited metabolic disease in Korea. Korean J Pediatr. 2006; 49:1125–1139.14. Allen DB, Hoffman GL, Fitzpatrick P, Laessig R, Maby S, Slyper A. Improved precision of newborn screening for congenital adrenal hyperplasia using weight-adjusted criteria for 17-hydroxyprogesterone levels. J Pediatr. 1997; 130:128–133.15. Ko HS, Lee S, Chae H, Choi SK, Kim M, Park IY, Suh BK, Shin JC. Prenatal diagnosis of congenital lipoid adrenal hyperplasia (CLAH) by molecular genetic testing in Korean siblings. Yonsei Med J. 2011; 52:1035–1038.16. Neary N, Nieman L. Adrenal insufficiency: etiology, diagnosis and treatment. Curr Opin Endocrinol Diabetes Obes. 2010; 17:217–223.17. Marik PE, Pastores SM, Annane D, Meduri GU, Sprung CL, Arlt W, Keh D, Briegel J, Beishuizen A, Dimopoulou I, et al. American College of Critical Care Medicine. Recommendations for the diagnosis and management of corticosteroid insufficiency in critically ill adult patients: consensus statements from an international task force by the American College of Critical Care Medicine. Crit Care Med. 2008; 36:1937–1949.18. Burris TP, Guo W, McCabe ER. The gene responsible for adrenal hypoplasia congenita, DAX-1, encodes a nuclear hormone receptor that defines a new class within the superfamily. Recent Prog Horm Res. 1996; 51:241–259.19. Reisch N, Arlt W, Krone N. Health problems in congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Horm Res Paediatr. 2011; 76:73–85.20. Jääskeläinen J, Voutilainen R. Growth of patients with 21-hydroxylase deficiency: an analysis of the factors influencing adult height. Pediatr Res. 1997; 41:30–33.21. Rasat R, Espiner EA, Abbott GD. Growth patterns and outcomes in congenital adrenal hyperplasia; effect of chronic treatment regimens. N Z Med J. 1995; 108:311–314.22. Joint LWPES/ESPE CAH Working Group. Consensus statement on 21-hydroxylase deficiency from the Lawson Wilkins Pediatric Endocrine Society and the European Society for Paediatric Endocrinology. J Clin Endocrinol Metab. 2002; 87:4048–4053.23. Thilén A, Woods KA, Perry LA, Savage MO, Wedell A, Ritzén EM. Early growth is not increased in untreated moderately severe 21-hydroxylase deficiency. Acta Paediatr. 1995; 84:894–898.24. Bonfig W, Schmidt H, Schwarz HP. Growth patterns in the first three years of life in children with classical congenital adrenal hyperplasia diagnosed by newborn screening and treated with low doses of hydrocortisone. Horm Res Paediatr. 2011; 75:32–37.25. Van der Kamp HJ, Otten BJ, Buitenweg N, De Muinck Keizer-Schrama SM, Oostdijk W, Jansen M, Delemarre-de Waal HA, Vulsma T, Wit JM. Longitudinal analysis of growth and puberty in 21-hydroxylase deficiency patients. Arch Dis Child. 2002; 87:139–144.26. Boepple PA, Mansfield MJ, Link K, Crawford JD, Crigler JF Jr, Kushner DC, Blizzard RM, Crowley WF Jr. Impact of sex steroids and their suppression on skeletal growth and maturation. Am J Physiol. 1988; 255:E559–E566.27. Trinh L, Nimkarn S, New MI, Lin-Su K. Growth and pubertal characteristics in patients with congenital adrenal hyperplasia due to 21-hydroxylase deficiency. J Pediatr Endocrinol Metab. 2007; 20:883–891.28. Völkl TM, Simm D, Beier C, Dörr HG. Obesity among children and adolescents with classic congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Pediatrics. 2006; 117:e98–e105.29. Bachelot A, Plu-Bureau G, Thibaud E, Laborde K, Pinto G, Samara D, Nihoul-Fékété C, Kuttenn F, Polak M, Touraine P. Long-term outcome of patients with congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Horm Res. 2007; 67:268–276.30. Jones HW Jr, Verkauf BS. Congenital adrenal hyperplasia: age at menarche and related events at puberty. Am J Obstet Gynecol. 1971; 109:292–298.31. Muscatelli F, Strom TM, Walker AP, Zanaria E, Récan D, Meindl A, Bardoni B, Guioli S, Zehetner G, Rabl W, et al. Mutations in the DAX-1 gene give rise to both X-linked adrenal hypoplasia congenita and hypogonadotropic hypogonadism. Nature. 1994; 372:672–676.32. Loke KY, Poh LK, Lee WW, Lai PS. A case of X-linked adrenal hypoplasia congenita, central precocious puberty and absence of the DAX-1 gene: implications for pubertal regulation. Horm Res. 2009; 71:298–304.33. Darcan S, Goksen D, Ozen S, Ozkinay F, Durmaz B, Lalli E. Gonadotropin-dependent precocious puberty in a patient with X-linked adrenal hypoplasia congenita caused by a novel DAX-1 mutation. Horm Res Paediatr. 2011; 75:153–156.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Role of Low-dose ACTH Stimulation Test in the Diagnosis of Adrenal Insufficiency
- The Effect of Low Dose and High Dose ACTH in the Evaluation of Adrenal Function
- A Case of Hypermelanosis in Congenital Adrenal Aplasia
- Usefullness of Urinary Free Cortisol Measurement in Diagnosis of Iatrogenic Cushing Syndrome
- Diagnostic Value of 1microgram Rapid ACTH Stimulation Test According to the Timing of Sampling of Serum Cortisol in Patients with Suspected Central Adrenal Insufficiency