J Korean Med Sci.
2014 Mar;29(3):370-377. 10.3346/jkms.2014.29.3.370.
The Relationship among Complex Fractionated Electrograms, Wavebreak, Phase Singularity, and Local Dominant Frequency in Fibrillation Wave-Dynamics: a Modeling Comparison Study
- Affiliations
-
- 1Department of Convergence Biomedical Engineering, Daelim University College, Anyang, Korea.
- 2Division of Cardiology, Yonsei University Health System, Seoul, Korea.
- 3Department of Biomedical Engineering, Chonnam National University, Yeosu, Korea.
- 4Department of Mechanical and Biomedical Engineering, Kangwon National University, Chuncheon, Korea. ebshim@kangwon.ac.kr
- KMID: 1734924
- DOI: http://doi.org/10.3346/jkms.2014.29.3.370
Abstract
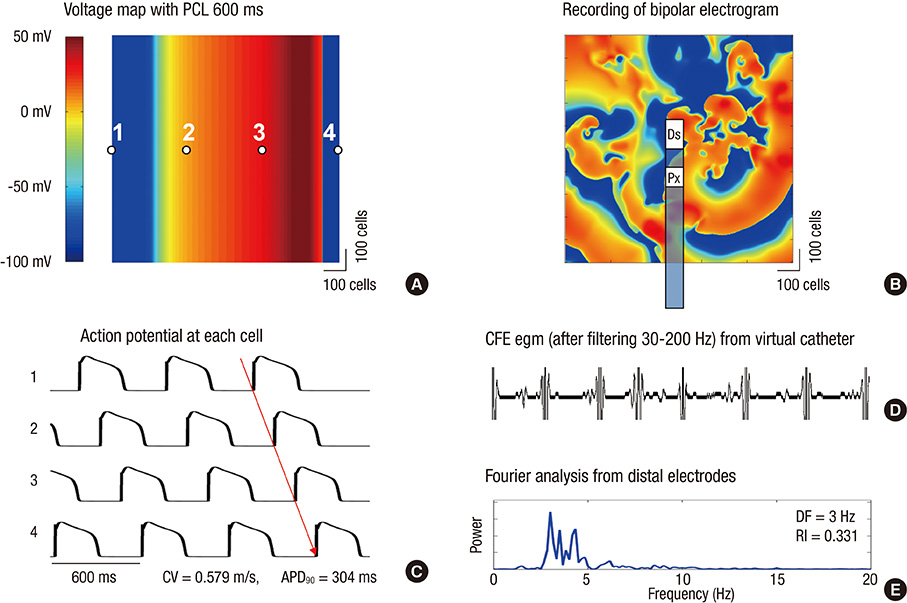
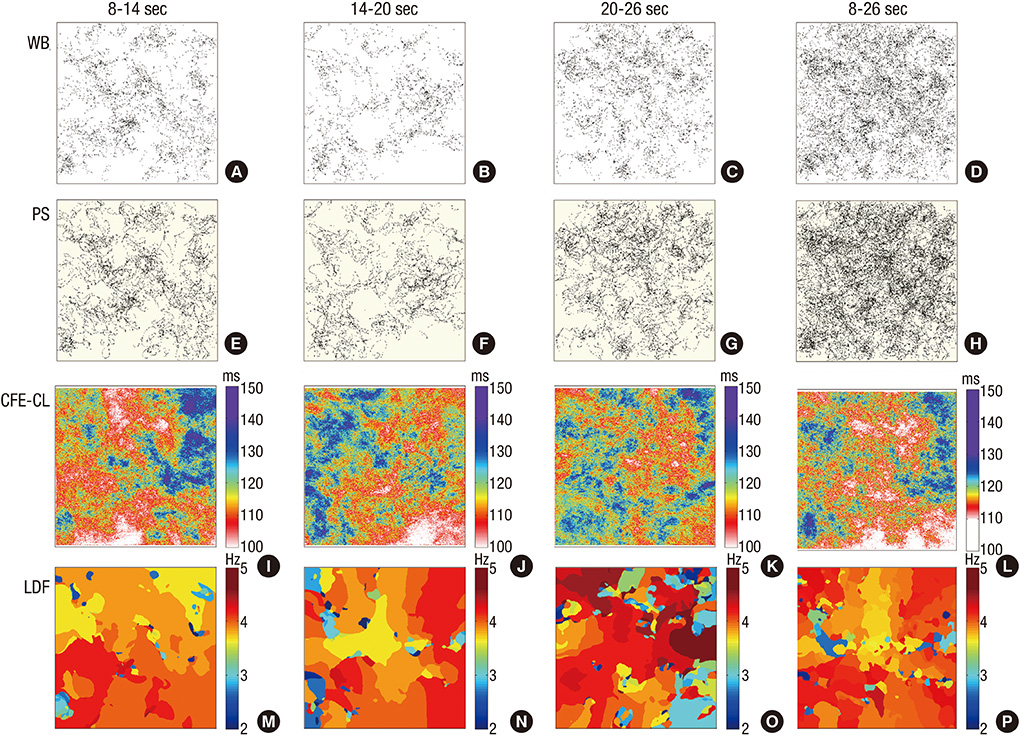
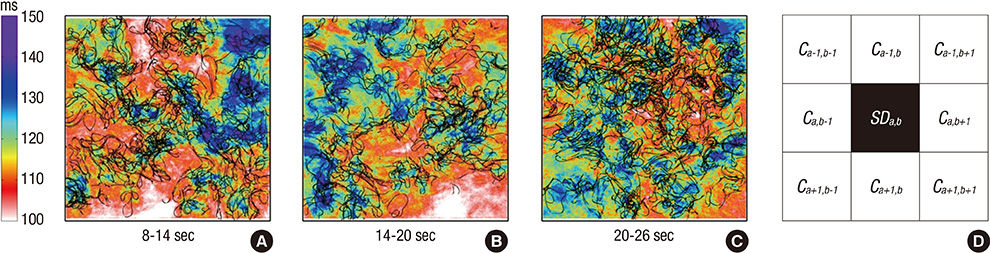
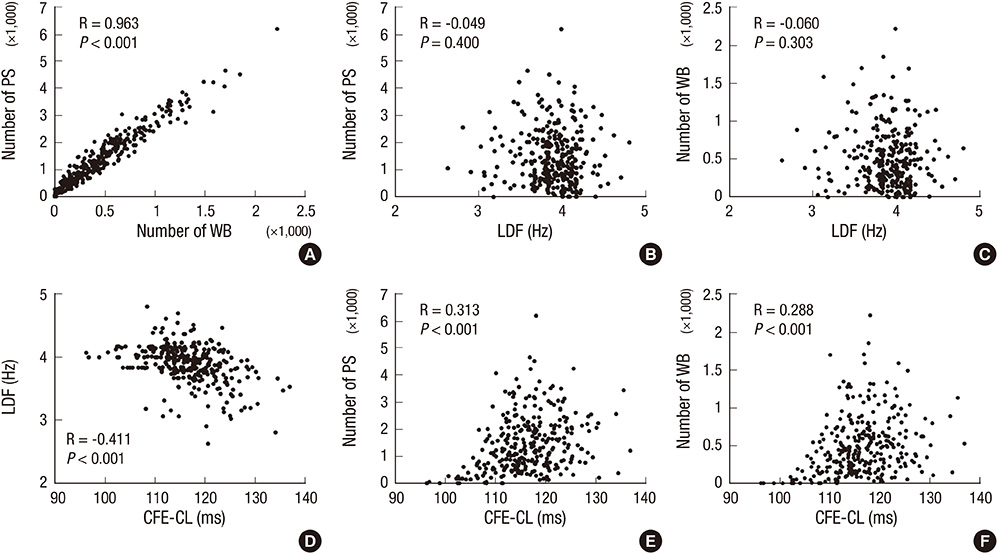
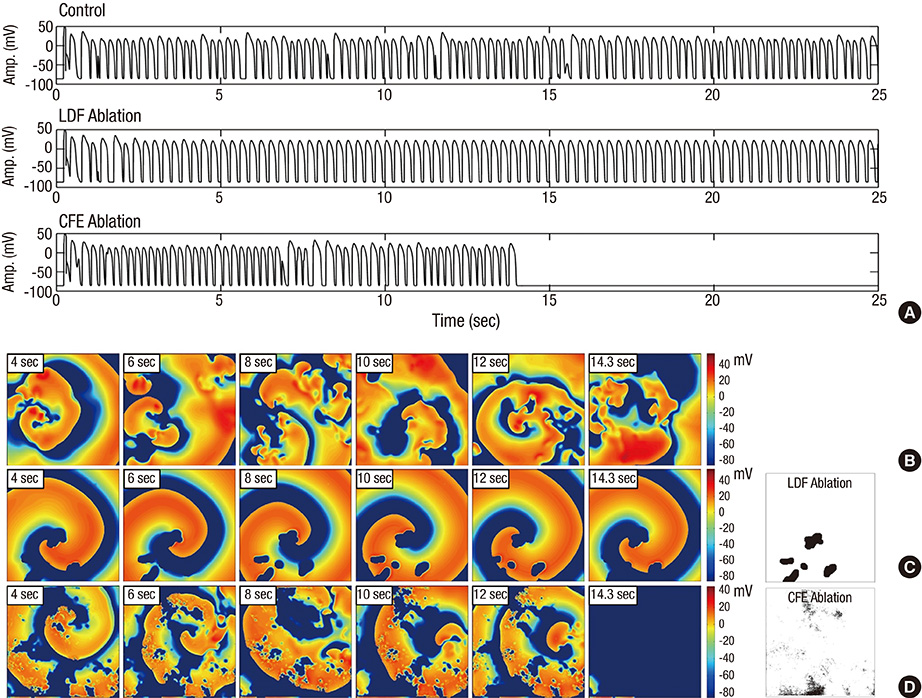
- Although complex fractionated electrogram (CFE) is known to be a target for catheter ablation of fibrillation, its physiological meaning in fibrillation wave-dynamics remains to be clarified. We evaluated the spatiotemporal relationships among the parameters of fibrillation wave-dynamics by simulation modeling. We generated maps of CFE-cycle length (CFE-CL), local dominant frequency (LDF), wave break (WB), and phase singularity (PS) of fibrillation in 2-dimensional homogeneous bidomain cardiac modeling (1,000 x 1,000 cells ten Tusscher model). We compared spatiotemporal correlations by dichotomizing each maps into 10 x 10 lattice zones. In spatial distribution, WB and PS showed excellent correlation (R = 0.963, P < 0.001). CFE-CL had weak correlations with WB (R = 0.288, P < 0.001), PS (R = 0.313, P < 0.001), and LDF (R = -0.411, P < 0.001). However, LDF did not show correlation with PS or WB. PSs were mostly distributed at the periphery of low CFE-CL area. Virtual ablation (5% of critical mass) of CFE-CL < 100 ms terminated fibrillation at 14.3 sec, and high LDF ablation (5% of critical mass) changed fibrillation to organized tachycardia, respectively. In homogeneous 2D fibrillation modeling, CFE-CL was weakly correlated with WB, PS, and LDF, spatiotemporally. PSs are mostly positioned at the periphery of low CFE-CL areas, and virtual ablation targeting low CFE-CL regions terminated fibrillation successfully.
MeSH Terms
Figure
Cited by 1 articles
-
Spatial reproducibility of complex fractionated atrial electrogram depending on the direction and configuration of bipolar electrodes: an
in-silico modeling study
Jun-Seop Song, Young-Seon Lee, Minki Hwang, Jung-Kee Lee, Changyong Li, Boyoung Joung, Moon-Hyoung Lee, Eun Bo Shim, Hui-Nam Pak
Korean J Physiol Pharmacol. 2016;20(5):507-514. doi: 10.4196/kjpp.2016.20.5.507.
Reference
-
1. Nademanee K, McKenzie J, Kosar E, Schwab M, Sunsaneewitayakul B, Vasavakul T, Khunnawat C, Ngarmukos T. A new approach for catheter ablation of atrial fibrillation: mapping of the electrophysiologic substrate. J Am Coll Cardiol. 2004; 43:2044–2053.2. Nademanee K, Veerakul G, Chandanamattha P, Chaothawee L, Ariyachaipanich A, Jirasirirojanakorn K, Likittanasombat K, Bhuripanyo K, Ngarmukos T. Prevention of ventricular fibrillation episodes in Brugada syndrome by catheter ablation over the anterior right ventricular outflow tract epicardium. Circulation. 2011; 123:1270–1279.3. Verma A, Sanders P, Macle L, Deisenhofer I, Morillo CA, Chen J, Jiang CY, Ernst S, Mantovan R. Substrate and trigger ablation for reduction of atrial fibrillation trial-part II (STAR AF II): design and rationale. Am Heart J. 2012; 164:1–6.e6.4. Calkins H, Kuck KH, Cappato R, Brugada J, Camm AJ, Chen SA, Crijns HJ, Damiano RJ Jr, Davies DW, DiMarco J, et al. 2012 HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation: recommendations for patient selection, procedural techniques, patient management and follow-up, definitions, endpoints, and research trial design: a report of the Heart Rhythm Society (HRS) Task Force on Catheter and Surgical Ablation of Atrial Fibrillation: developed in partnership with the European Heart Rhythm Association (EHRA), a registered branch of the European Society of Cardiology (ESC) and the European Cardiac Arrhythmia Society (ECAS); and in collaboration with the American College of Cardiology (ACC), American Heart Association (AHA), the Asia Pacific Heart Rhythm Society (APHRS), and the Society of Thoracic Surgeons (STS): endorsed by the governing bodies of the American College of Cardiology Foundation, the American Heart Association, the European Cardiac Arrhythmia Society, the European Heart Rhythm Association, the Society of Thoracic Surgeons, the Asia Pacific Heart Rhythm Society, and the Heart Rhythm Society. Heart Rhythm. 2012; 9:632–696.e21.5. Park JH, Pak HN, Kim SK, Jang JK, Choi JI, Lim HE, Hwang C, Kim YH. Electrophysiologic characteristics of complex fractionated atrial electrograms in patients with atrial fibrillation. J Cardiovasc Electrophysiol. 2009; 20:266–272.6. Shim EB, Leem CH, Abe Y, Noma A. A new multi-scale simulation model of the circulation: from cells to system. Philos Trans A Math Phys Eng Sci. 2006; 364:1483–1500.7. Shim EB, Amano A, Takahata T, Shimayoshi T, Noma A. The cross-bridge dynamics during ventricular contraction predicted by coupling the cardiac cell model with a circulation model. J Physiol Sci. 2007; 57:275–285.8. Im UB, Kwon SS, Kim K, Lee YH, Park YK, Youn CH, Shim EB. Theoretical analysis of the magnetocardiographic pattern for reentry wave propagation in a three-dimensional human heart model. Prog Biophys Mol Biol. 2008; 96:339–356.9. Ashihara T, Namba T, Ito M, Ikeda T, Nakazawa K, Trayanova N. Spiral wave control by a localized stimulus: a bidomain model study. J Cardiovasc Electrophysiol. 2004; 15:226–233.10. Ten Tusscher KH, Panfilov AV. Alternans and spiral breakup in a human ventricular tissue model. Am J Physiol Heart Circ Physiol. 2006; 291:H1088–H1100.11. Priebe L, Beuckelmann DJ. Simulation study of cellular electric properties in heart failure. Circ Res. 1998; 82:1206–1223.12. Iyer V, Mazhari R, Winslow RL. A computational model of the human left-ventricular epicardial myocyte. Biophys J. 2004; 87:1507–1525.13. Ten Tusscher KH, Bernus O, Hren R, Panfilov AV. Comparison of electrophysiological models for human ventricular cells and tissues. Prog Biophys Mol Biol. 2006; 90:326–345.14. Ern A, Guermond J. Theory and practice of finite elements. New York: Springer;2004.15. Clayton RH, Holden AV. A method to quantify the dynamics and complexity of re-entry in computational models of ventricular fibrillation. Phys Med Biol. 2002; 47:225–238.16. Park JH, Park SW, Kim JY, Kim SK, Jeoung B, Lee MH, Hwang C, Kim YH, Kim SS, Pak HN. Characteristics of complex fractionated atrial electrogram in the electroanatomically remodeled left atrium of patients with atrial fibrillation. Circ J. 2010; 74:1557–1563.17. Moe G. On the multiple wavelet hypothesis of atrial fibrillation. Arch Int Pharmacodyn Ther. 1962; 140:183–188.18. Iyer AN, Gray RA. An experimentalist's approach to accurate localization of phase singularities during reentry. Ann Biomed Eng. 2001; 29:47–59.19. Oppenheim AV, Schafer RW. Discrete-time signal processing. Upper Saddle River: Prentice-Hall;1989.20. Konings KT, Smeets JL, Penn OC, Wellens HJ, Allessie MA. Configuration of unipolar atrial electrograms during electrically induced atrial fibrillation in humans. Circulation. 1997; 95:1231–1241.21. Kienzle MG, Miller J, Falcone RA, Harken A, Josephson ME. Intraoperative endocardial mapping during sinus rhythm: relationship to site of origin of ventricular tachycardia. Circulation. 1984; 70:957–965.22. Haqqani HM, Kalman JM, Roberts-Thomson KC, Balasubramaniam RN, Rosso R, Snowdon RL, Sparks PB, Vohra JK, Morton JB. Fundamental differences in electrophysiologic and electroanatomic substrate between ischemic cardiomyopathy patients with and without clinical ventricular tachycardia. J Am Coll Cardiol. 2009; 54:166–173.23. Jalife J, Berenfeld O, Mansour M. Mother rotors and fibrillatory conduction: a mechanism of atrial fibrillation. Cardiovasc Res. 2002; 54:204–216.24. Wu TJ, Lin SF, Weiss JN, Ting CT, Chen PS. Two types of ventricular fibrillation in isolated rabbit hearts: importance of excitability and action potential duration restitution. Circulation. 2002; 106:1859–1866.25. Pak HN, Oh YS, Liu YB, Wu TJ, Karagueuzian HS, Lin SF, Chen PS. Catheter ablation of ventricular fibrillation in rabbit ventricles treated with beta-blockers. Circulation. 2003; 108:3149–3156.26. Lee G, Roberts-Thomson K, Madry A, Spence S, Teh A, Heck PM, Kumar S, Kistler PM, Morton JB, Sanders P, et al. Relationship among complex signals, short cycle length activity, and dominant frequency in patients with long-lasting persistent AF: a high-density epicardial mapping study in humans. Heart Rhythm. 2011; 8:1714–1719.27. Hodgkin AL, Huxley AF. A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol. 1952; 117:500–544.28. Pak HN, Kim YH, Lim HE, Chou CC, Miyauchi Y, Fang YH, Sun K, Hwang C, Chen PS. Role of the posterior papillary muscle and purkinje potentials in the mechanism of ventricular fibrillation in open chest dogs and Swine: effects of catheter ablation. J Cardiovasc Electrophysiol. 2006; 17:777–783.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Wavebreak Mechanism During Ventricular Fibrillation in Isolated Swine Right Ventricle
- Association of Complex Fractionated Electrograms with Atrial Myocardial Thickness and Fibrosis
- The Characteristics of Unipolar and Bipolar Electrograms at the Origin of Ventricular Tachycardia
- Spatial reproducibility of complex fractionated atrial electrogram depending on the direction and configuration of bipolar electrodes: an in-silico modeling study
- Computational Fluid Dynamics Modeling in Aortic Diseases