J Vet Sci.
2009 Sep;10(3):177-180. 10.4142/jvs.2009.10.3.177.
Immunohistochemical localization of galectin-3 in the granulomatous lesions of paratuberculosis-infected bovine intestine
- Affiliations
-
- 1Department of Veterinary Anatomy, College of Veterinary Medicine and Applied Radiological Science Research Institute, Jeju National University, Jeju 690-756, Korea. shint@cheju.ac.kr
- 2Department of Veterinary Anatomy, College of Veterinary Medicine and Animal Medical Institute, Chonnam National University, Gwangju 500-757, Korea.
- 3Department of Microbiology, College of Medicine, Jeju National University, Jeju 690-756, Korea.
- KMID: 1726915
- DOI: http://doi.org/10.4142/jvs.2009.10.3.177
Abstract
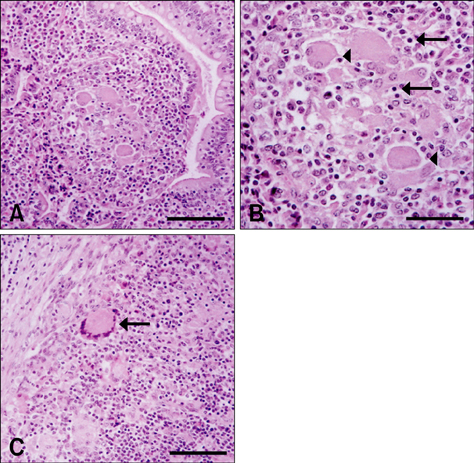
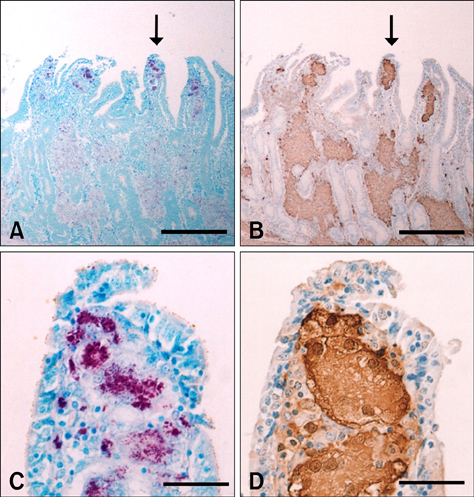
- The presence of galectin-3 was immunohistochemically quantified in bovine intestines infected with paratuberculosis (Johne's disease) to determine whether galectin-3 was involved in the formation of granulation tissue associated with the disease. Mycobacterium avium subsp. paratuberculosis infection was histochemically confirmed using Ziehl-Neelsen staining and molecularly diagnosed through rpoB DNA sequencing. Galectin-3 was detected in the majority of inflammatory cells, possibly macrophages, in the granulomatous lesions within affected tissues, including the ileum. These findings suggest that galectin-3 is associated with the formation of chronic granulation tissues in bovine paratuberculosis, probably through cell adhesion and anti-apoptosis mechanisms.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Investigation of the Roles of Cyclooxygenase-2 and Galectin-3 Expression in the Pathogenesis of Premenopausal Endometrial Polyps
Esin Kasap, Serap Karaarslan, Esra Bahar Gur, Mine Genc, Nur Sahin, Serkan Güclü
J Pathol Transl Med. 2016;50(3):225-230. doi: 10.4132/jptm.2016.03.08.
Reference
-
1. Almkvist J, Karlsson A. Galectins as inflammatory mediators. Glycoconj J. 2004. 19:575–581.
Article2. Barondes SH, Cooper DN, Gitt MA, Leffler H. Galectins. Structure and function of a large family of animal lectins. J Biol Chem. 1994. 269:20807–20810.
Article3. Beatty WL, Rhoades ER, Hsu DK, Liu FT, Russell DG. Association of a macrophage galactoside-binding protein with Mycobacterium-containing phagosomes. Cell Microbiol. 2002. 4:167–176.
Article4. Boor KJ, Duncan ML, Price CW. Genetic and transcriptional organization of the region encoding the beta subunit of Bacillus subtilis RNA polymerase. J Biol Chem. 1995. 270:20329–20336.
Article5. Fowler M, Thomas RJ, Atherton J, Roberts IS, High NJ. Galectin-3 binds to Helicobacter pylori O-antigen: it is upregulated and rapidly secreted by gastric epithelial cells in response to H. pylori adhesion. Cell Microbiol. 2006. 8:44–54.
Article6. Harris NB, Barletta RG. Mycobacterium avium subsp. paratuberculosis in Veterinary Medicine. Clin Microbiol Rev. 2001. 14:489–512.
Article7. Hsu DK, Yang RY, Pan Z, Yu L, Samomon DR, Fung-Leung WP, Liu FT. Targeted disruption of the galectin-3 gene results in attenuated peritoneal inflammatory responses. Am J Pathol. 2000. 156:1073–1083.
Article8. Hughes RC. Galectins as modulators of cell adhesion. Biochimie. 2001. 83:667–676.
Article9. Kaltner H, Seyrek K, Heck A, Sinowatz F, Gabius H. Galectin-1 and galectin-3 in fetal development of bovine respiratory and digestive tracts. Comparison of cell type-specific expression profiles and subcellular localization. Cell Tissue Res. 2002. 307:35–46.
Article10. Kim BJ, Lee SH, Lyu MA, Kim SJ, Bai GH, Chae GT, Kim EC, Cha CY, Kook YH. Identification of mycobacterial species by comparative sequence analysis of the RNA polymerase gene (rpoB). J Clin Microbiol. 1999. 37:1714–1720.
Article11. Kim H, Kang TY, Joo HG, Shin T. Immunohistochemical localization of galectin-3 in boar testis and epididymis. Acta Histochem. 2006. 108:481–485.
Article12. Kim H, Lee J, Hyun JW, Park JW, Joo HG, Shin T. Expression and immunohistochemical localization of galectin-3 in various mouse tissues. Cell Biol Int. 2007. 31:655–662.
Article13. Kim H, Ahn M, Moon C, Kim S, Jee Y, Joo HG, Shin T. Immunohistochemical study of galectin-3 in mature and immature bull testis and epididymis. J Vet Sci. 2008. 9:339–344.
Article14. Liu FT. Galectins: a new family of regulators of inflammation. Clin Immunol. 2000. 97:79–88.
Article15. Liu FT, Frigeri LG, Gritzmacher CA, Hsu DK, Robertson MW, Zuberi RI. Expression and function of an IgE-binding animal lectin (epsilon BP) in mast cells. Immunopharmacology. 1993. 26:187–195.
Article16. Liu FT, Patterson RJ, Wang JL. Intracellular functions of galectins. Biochim Biophys Acta. 2002. 1572:263–273.
Article17. Luna LG. Manual of Histologic Staining Methods of the Armed Forced Institute of Pathology. 1968. 3rd ed. New York: McGraw-Hill Book Company;220.18. McGavin MD, Zachary JF. Pathologic Basis of Veterinary Disease. 2006. 4th ed. Philadelphia: Elsevier;372–374.19. Müller S, Schaffer T, Flogerzi B, Fleetwood A, Weimann R, Schoepfer AM, Seibold F. Galectin-3 modulates T cell activity and is reduced in the inflamed intestinal epithelium in IBD. Inflamm Bowel Dis. 2006. 12:588–597.
Article20. Nio J, Kon Y, Iwanaga T. Differential cellular expression of galectin family mRNAs in the epithelial cells of the mouse digestive tract. J Histochem Cytochem. 2005. 53:1323–1334.
Article21. Oka N, Nakahara S, Takenaka Y, Fukumori T, Hogan V, Kanayama HO, Yanagawa T, Raz A. Galectin-3 inhibits tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis by activating Akt in human bladder carcinoma cells. Cancer Res. 2005. 65:7546–7553.
Article22. Olsen I, Sigurğardóttir G, Djønne B. Paratuberculosis with special reference to cattle. A review. Vet Q. 2002. 24:12–28.
Article23. Sano H, Hsu DK, Apgar JR, Yu L, Sharma BB, Kuwabara I, Izui S, Liu FT. Critical role of galectin-3 in phagocytosis by macrophages. J Clin Invest. 2003. 112:389–397.
Article24. Vray B, Camby I, Vercruysse V, Mijatovic T, Bovin NV, Ricciardi-Castagnoli P, Kaltner H, Salmon I, Gabius HJ, Kiss R. Up-regulation of galectin-3 and its ligands by Trypanosoma cruzi infection with modulation of adhesion and migration of murine dendritic cells. Glycobiology. 2004. 14:647–657.
Article25. Yang RY, Hsu DK, Liu FT. Expression of galectin-3 modulates T-cell growth and apoptosis. Proc Natl Acad Sci USA. 1996. 93:6737–6742.
Article26. Yu F, Finley RL Jr, Raz A, Kim HR. Galectin-3 translocates to the perinuclear membranes and inhibits cytochrome c release from the mitochondria. A role for synexin in galectin-3 translocation. J Biol Chem. 2002. 277:15819–15827.
Article27. Yun YJ, Lee KH, Haihua L, Ryu YJ, Kim BJ, Lee YH, Baek GH, Kim HJ, Chung MS, Lee MC, Lee SH, Choi IH, Cho TJ, Chang BS, Kook YH. Detection and identification of Mycobacterium tuberculosis in joint biopsy specimens by rpoB PCR cloning and sequencing. J Clin Microbiol. 2005. 43:174–178.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Immunohistochemical localization of galectin-3 in the brain with Theiler's murine encephalomyelitis virus (DA strain) infection
- Tactics of Mycobacterium avium subsp. paratuberculosis for intracellular survival in mononuclear phagocytes
- Is the expression of p16INK4A and galectin-3 correlated with disease progression of cervical neoplasia?
- The Significance of Immunohistochemical Staining in Thyroid Nodule: TPO and Galectin-3
- Case report of Johne's disease in Korean black goat (Capra hircus aegagrus)