Korean J Ophthalmol.
2012 Aug;26(4):290-296. 10.3341/kjo.2012.26.4.290.
Safety of Bevacizumab on Extraocular Muscle in a Rabbit Model
- Affiliations
-
- 1Department of Ophthalmology, Pusan National University Yangsan Hospital, Yangsan, Korea.
- 2Medical Research Institute, Pusan National University, Busan, Korea. hychoi@pusan.ac.kr
- 3Department of Ophthalmology, Busan Medical Center, Busan, Korea.
- 4Department of Ophthalmology, Pusan National University Hospital, Busan, Korea.
- KMID: 1387397
- DOI: http://doi.org/10.3341/kjo.2012.26.4.290
Abstract
- PURPOSE
The purpose of this study was to investigate the myotoxicity of bevacizumab on extraocular muscles in a rabbit model.
METHODS
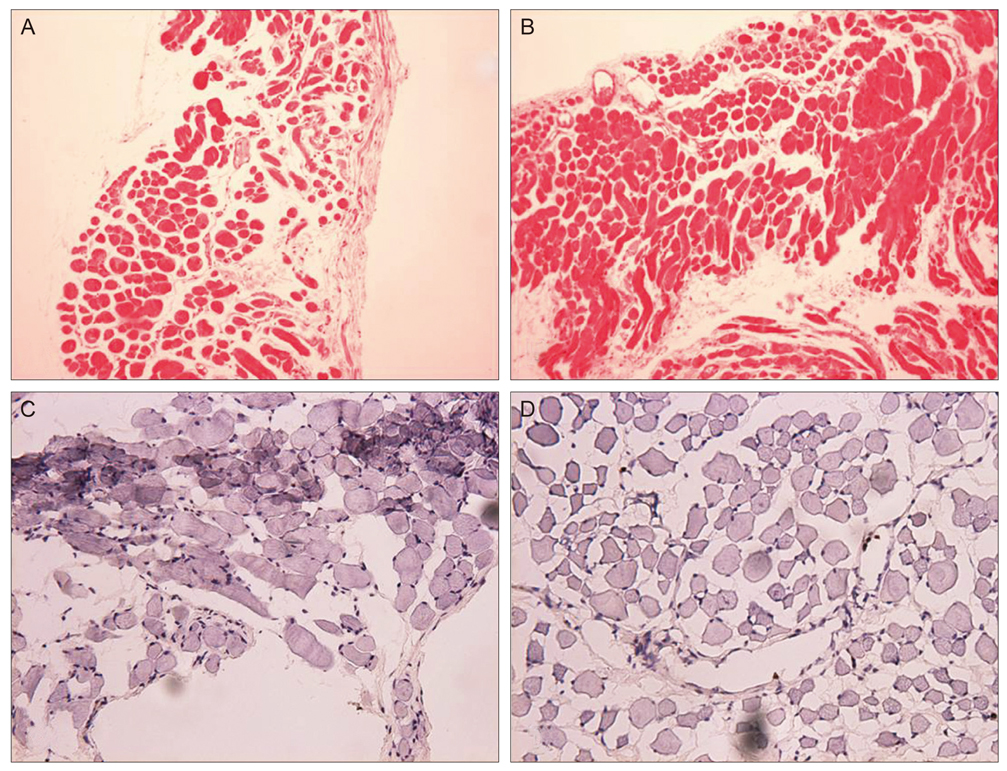
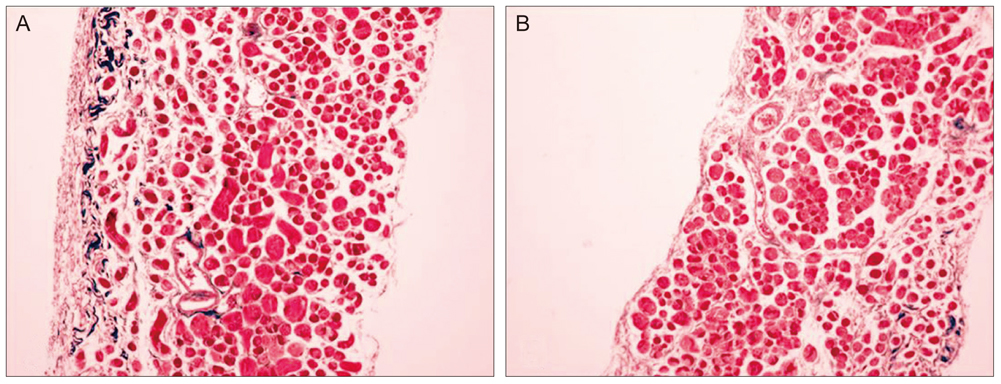
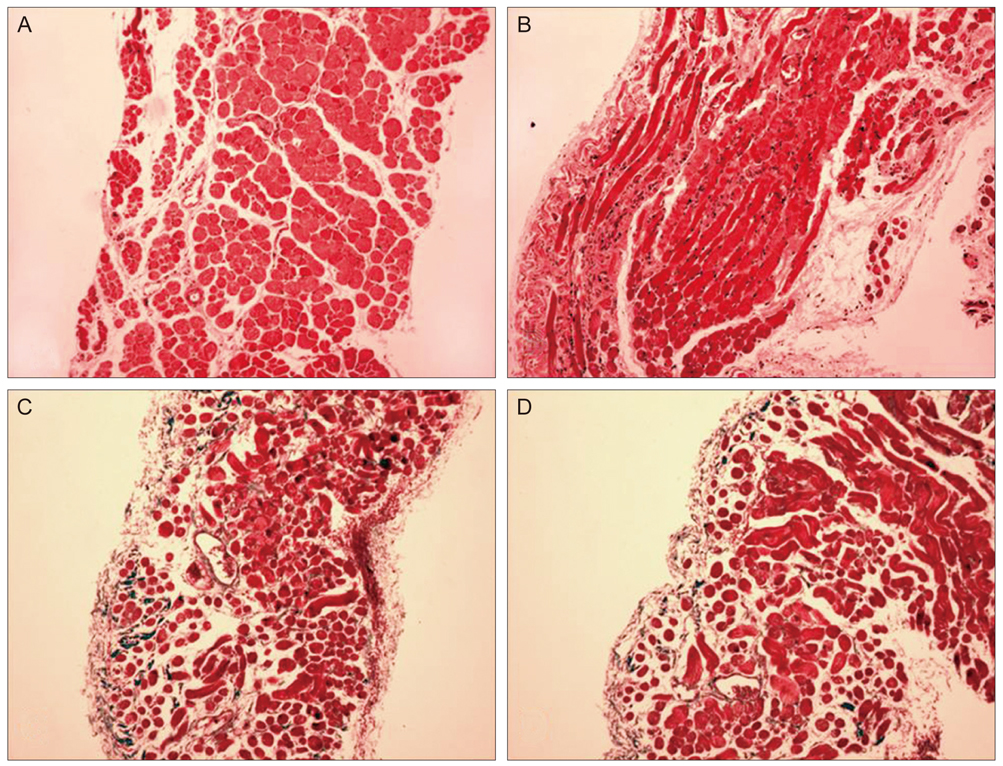
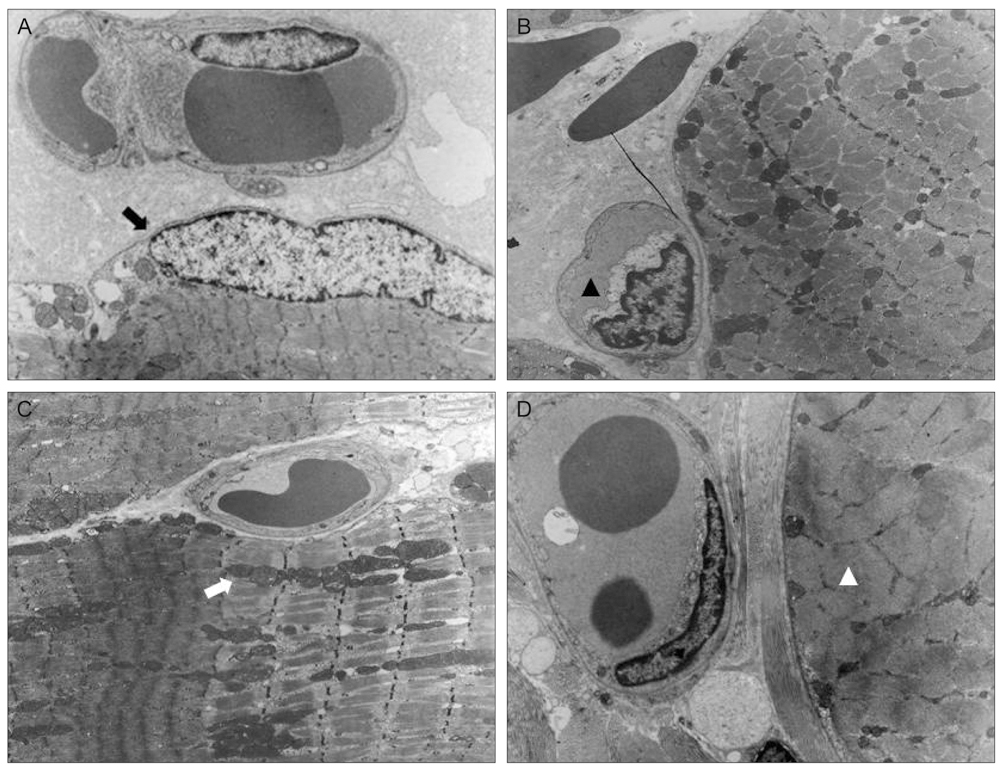
Thirty New Zealand white rabbits were used for this study. The animals were evenly divided into two groups. In the first group, 15 rabbits were treated with intramuscular injections of bevacizumab (1.25 mg/0.05 mL) in the right superior rectus muscle and normal saline solution (0.05 mL) in the left superior rectus muscle. In the second group, 15 rabbits were treated with subconjunctival injections of bevacizumab (2.5 mg/0.1 mL) in the right superior subconjunctival area and normal saline solution (0.1 mL) in the left superior subconjunctival area. Five rabbits in each group were sacrificed at one day, two weeks and four weeks after the injections. Extraocular muscle samples were prepared for light microscopic (LM) and electron microscopic (EM) examination. Degrees of acute inflammation were evaluated via CD-11b immunohistochemistry, and global muscle change was investigated using hematoxylin and eosin stains. Intensity of fibrosis was evaluated using Masson trichrome stains, and ultrastructural changes were observed on EM.
RESULTS
We observed no significant inflammatory cell infiltration, muscle necrosis or fibrotic change in treated and control eyes. EM findings revealed no significant damage to muscle or vascular tissue after bevacizumab injection.
CONCLUSIONS
We found no signs of extraocular muscle toxicity after LM and EM intramuscular and subconjunctival bevacizumab injections in a rabbit model.
Keyword
MeSH Terms
Figure
Reference
-
1. Hurwitz H, Fehrenbacher L, Novotny W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004. 350:2335–2342.2. Gunther JB, Altaweel MM. Bevacizumab (Avastin) for the treatment of ocular disease. Surv Ophthalmol. 2009. 54:372–400.3. Banifatemi M, Razeghinejad MR, Hosseini H, Gholampour A. Bevacizumab and ocular wound healing after primary pterygium excision. J Ocul Pharmacol Ther. 2011. 27:17–21.4. Oh JY, Kim MK, Wee WR. Subconjunctival and intracorneal bevacizumab injection for corneal neovascularization in lipid keratopathy. Cornea. 2009. 28:1070–1073.5. Saravia M, Zapata G, Ferraiolo P, et al. Anti-VEGF monoclonal antibody-induced regression of corneal neovascularization and inflammation in a rabbit model of herpetic stromal keratitis. Graefes Arch Clin Exp Ophthalmol. 2009. 247:1409–1416.6. Dastjerdi MH, Al-Arfaj KM, Nallasamy N, et al. Topical bevacizumab in the treatment of corneal neovascularization: results of a prospective, open-label, noncomparative study. Arch Ophthalmol. 2009. 127:381–389.7. Memarzadeh F, Varma R, Lin LT, et al. Postoperative use of bevacizumab as an antifibrotic agent in glaucoma filtration surgery in the rabbit. Invest Ophthalmol Vis Sci. 2009. 50:3233–3237.8. Okland S, Komorowski TE, Carlson BM. Ultrastructure of mepivacaine-induced damage and regeneration in rat extraocular muscle. Invest Ophthalmol Vis Sci. 1989. 30:1643–1651.9. Kytta J, Heinonen E, Rosenberg PH, et al. Effects of repeated bupivacaine administration on sciatic nerve and surrounding muscle tissue in rats. Acta Anaesthesiol Scand. 1986. 30:625–629.10. Chapman JM, Abdelatif OM, Cheeks L, Green K. Subconjunctival gentamicin induction of extraocular toxic muscle myopathy. Ophthalmic Res. 1992. 24:189–196.11. O'Day DM, Smith R, Stevens JB, et al. Toxicity and pharmacokinetics of subconjunctival amphotericin B: an experimental study. Cornea. 1991. 10:411–417.12. Lim TH, Bae SH, Cho YJ, et al. Concentration of vascular endothelial growth factor after intracameral bevacizumab injection in eyes with neovascular glaucoma. Korean J Ophthalmol. 2009. 23:188–192.13. Kernt M, Welge-Lussen U, Yu A, et al. Bevacizumab is not toxic to human anterior- and posterior-segment cultured cells. Ophthalmologe. 2007. 104:965–971.14. Yoeruek E, Spitzer MS, Tatar O, et al. Safety profile of bevacizumab on cultured human corneal cells. Cornea. 2007. 26:977–982.15. Bakri SJ, Cameron JD, McCannel CA, et al. Absence of histologic retinal toxicity of intravitreal bevacizumab in a rabbit model. Am J Ophthalmol. 2006. 142:162–164.16. Manzano RP, Peyman GA, Khan P, Kivilcim M. Testing intravitreal toxicity of bevacizumab (Avastin). Retina. 2006. 26:257–261.17. Kaempf S, Johnen S, Salz AK, et al. Effects of bevacizumab (Avastin) on retinal cells in organotypic culture. Invest Ophthalmol Vis Sci. 2008. 49:3164–3171.18. Cakmak HB, Toklu Y, Yorgun MA, Simsek S. Isolated sixth nerve palsy after intravitreal bevacizumab injection. Strabismus. 2010. 18:18–20.19. Park HJ, Guy J. Sixth nerve palsy post intravitreal bevacizumab for AMD: a new possibly causal relationship and complication? Binocul Vis Strabismus Q. 2007. 22:209.20. Mora JS, Sprunger DT, Helveston EM, Evan AP. Intraoperative sponge 5-fluorouracil to reduce postoperative scarring in strabismus surgery. J AAPOS. 1997. 1:92–97.21. Ruff R, Kaminski H, Maas E, Spiegel P. Ocular muscles: physiology and structure-function correlations. Bull Soc Belge Ophtalmol. 1989. 237:321–352.22. Chiarandini DJ, Davidowitz J. Structure and function of extraocular muscle fibers. Curr Top Eye Res. 1979. 1:91–142.23. Merrillees NC, Sunderland S, Hayhow W. Neuromuscular spindles in the extraocular muscles in man. Anat Rec. 1950. 108:23–30.24. Tengroth B, Hamberger A. Oxygen consumption studies on guinea pigs after administration of D- and L-Thyroxine: effect on the whole animal compared with that on its eye muscles. Acta Ophthalmol (Copenh). 1963. 41:524–530.25. Komorowski TE, Shepard B, Okland S, Carlson BM. An electron microscopic study of local anesthetic-induced skeletal muscle fiber degeneration and regeneration in the monkey. J Orthop Res. 1990. 8:495–503.26. Zink W, Graf BM. Local anesthetic myotoxicity. Reg Anesth Pain Med. 2004. 29:333–340.27. Tidball JG. Inflammatory cell response to acute muscle injury. Med Sci Sports Exerc. 1995. 27:1022–1032.28. Cotran RS, Kumar V, Robbins SL. Robbins SL, Cotran RS, Kumar V, editors. Inflammation and repair. Robbins' pathologic basis of disease. 1989. 4th ed. Philadelphia: Saunders;39–71.29. Hoeben A, Landuyt B, Highley MS, et al. Vascular endothelial growth factor and angiogenesis. Pharmacol Rev. 2004. 56:549–580.30. Wilgus TA, Ferreira AM, Oberyszyn TM, et al. Regulation of scar formation by vascular endothelial growth factor. Lab Invest. 2008. 88:579–590.31. Matos K, Manso PG, Marback E, et al. Protein expression of VEGF, IGF-1 and FGF in retroocular connective tissues and clinical correlation in Graves' ophthalmopathy. Arq Bras Oftalmol. 2008. 71:486–492.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Distribution of the Extraocular Pulley Smooth Muscle in the Rabbit
- The Effect of Subconjunctival Injection of Bevacizumab After Resection of Muscle in Rabbit Models
- Evaluation of Extraocular Muscle Contractibility after Recession and resection by EMG in Rabbit
- The Resection of Extraocular Muscle without Scleral Suture in the Rabbit
- Prevention of Adhesion Syndrome after Extraocular Muscle Surgery in Rabbit