J Vet Sci.
2012 Mar;13(1):73-79. 10.4142/jvs.2012.13.1.73.
Development and characterization of a potential diagnostic monoclonal antibody against capsid protein VP1 of the chicken anemia virus
- Affiliations
-
- 1Department of Veterinary Medicine, National Pingtung University of Science and Technology, Pingtung 91201, Taiwan.
- 2Graduate School of Biotechnology, Hungkuang University, Taichung 43302, Taiwan.
- 3Department of Bioresources, Dayeh University, Chunghwa 51591, Taiwan.
- 4Department of Food Science, National Pingtung University of Science and Technology, Pingtung 91201, Taiwan.
- 5Department of Medical Research, Tungs' Taichung MetroHarbor Hospital, Taichung 43503, Taiwan.
- 6Department of Safety, Health and Environmental Engineering, Mingchi University of Technology, Taipei 24301, Taiwan.
- 7School of Chinese Medicine Resources, China Medical University, Taichung 40402, Taiwan. leemengshiou@mail.cmu.edu.tw
- KMID: 1365006
- DOI: http://doi.org/10.4142/jvs.2012.13.1.73
Abstract
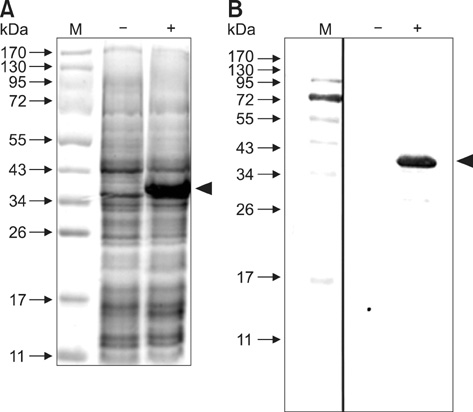
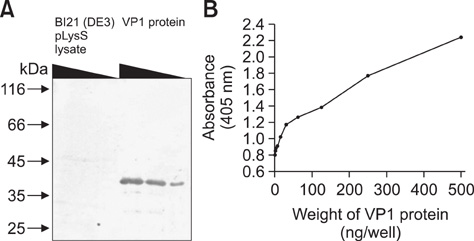
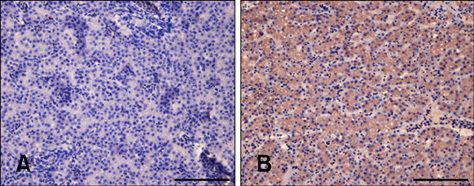
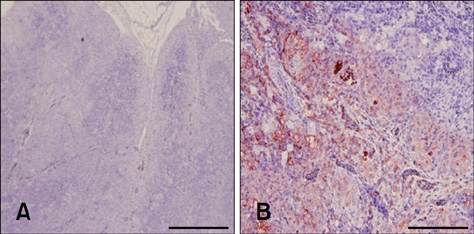
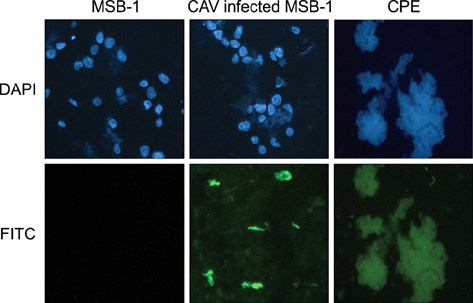
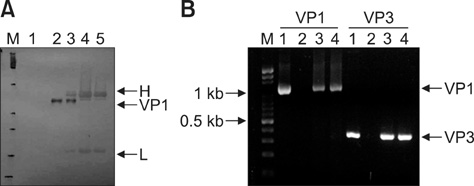
- Chicken anemia virus (CAV) is an important viral pathogen that causes anemia and severe immunodeficiency syndrome in chickens worldwide. In this study, a potential diagnostic monoclonal antibody against the CAV VP1 protein was developed which can precisely recognize the CAV antigen for diagnostic and virus recovery purposes. The VP1 gene of CAV encoding the N-terminus-deleted VP1 protein, VP1Nd129, was cloned into an Escherichia (E.) coli expression vector. After isopropyl-beta-D-thiogalactopyronoside induction, VP1Nd129 protein was shown to be successfully expressed in the E. coli. By performing an enzyme-linked immunoabsorbent assay using two coating antigens, purified VP1Nd129 and CAV-infected liver tissue lysate, E3 monoclonal antibody (mAb) was found to have higher reactivity against VP1 protein than the other positive clones according to the result of limiting dilution method from 64 clones. Using immunohistochemistry, the presence of the VP1-specific mAb, E3, was confirmed using CAV-infected liver and thymus tissues as positive-infected samples. Additionally, CAV particle purification was also performed using an immunoaffinity column containing E3 mAb. The monoclonal E3 mAb developed in this study will not only be very useful for detecting CAV infection and performing histopathology studies of infected chickens, but may also be used to purify CAV particles in the future.
MeSH Terms
-
Animals
Antibodies, Monoclonal/biosynthesis/genetics/*immunology
Antigens, Viral/analysis
Capsid Proteins/genetics/*immunology
Chicken anemia virus/genetics/*immunology
*Chickens
Circoviridae Infections/blood/immunology/*veterinary/virology
Escherichia coli/genetics
Immunohistochemistry/veterinary
Liver/virology
Mice
Mice, Inbred BALB C
Microscopy, Fluorescence/veterinary
Poultry Diseases/blood/immunology/*virology
Specific Pathogen-Free Organisms
Thymus Gland/virology
Figure
Reference
-
1. Brentano L, Lazzarin S, Bassi SS, Klein TAP, Schat KA. Detection of chicken anemia virus in the gonads and in the progeny of broiler breeder hens with high neutralizing antibody titers. Vet Microbiol. 2005. 105:65–72.
Article2. Daum I, Finsterbusch T, Härtle S, Göbel TW, Mankertz A, Korbel R, Grund C. Cloning and expression of a truncated pigeon circovirus capsid protein suitable for antibody detection in infected pigeons. Avian Pathol. 2009. 38:135–141.
Article3. Ducatez MF, Owoade AA, Abiola JO, Muller CP. Molecular epidemiology of chicken anemia virus in Nigeria. Arch Virol. 2006. 151:97–111.
Article4. Huang CH, Lai GH, Lee MS, Lin WH, Lien YY, Hsueh SC, Kao JY, Chang WT, Lu TC, Lin WN, Chen HJ, Lee MS. Development and evaluation of a loop-mediated isothermal amplification assay for rapid detection of chicken anaemia virus. J Appl Microbiol. 2010. 108:917–924.
Article5. Iwata N, Fujino M, Tuchiya K, Iwata A, Otaki Y, Ueda S. Development of an enzyme-linked immunosorbent assay using recombinant chicken anemia virus proteins expressed in a baculovirus vector system. J Vet Med Sci. 1998. 60:175–180.
Article6. Köhler G, Milstein C. Derivation of specific antibodyproducing tissue culture and tumor lines by cell fusion. Eur J Immunol. 1976. 6:511–519.
Article7. Lacorte C, Lohuis H, Goldbach R, Prins M. Assessing the expression of chicken anemia virus proteins in plants. Virus Res. 2007. 129:80–86.
Article8. Lee MS, Lien YY, Feng SH, Huang RL, Tsai MC, Chang WT, Chen HJ. Production of chicken anemia virus (CAV) VP1 and VP2 protein expressed by recombinant Escherichia coli. Process Biochem. 2009. 44:390–395.
Article9. Lou Z, Li X, Li Z, Yin X, Li B, Lan X, Yang B, Zhang Y, Liu J. Expression and antigenicity characterization for truncated capsid protein of porcine circovirus type 2. Can J Vet Res. 2011. 75:61–64.10. Lucio B, Schat KA, Shivaprasad HL. Identification of the chicken anemia agent, reproduction of the disease, and serological survey in the United States. Avian Dis. 1990. 34:146–153.
Article11. McNulty MS, Connor TJ, McNeilly F, Spackman D. Chicken anemia agent in the United States: isolation of the virus and detection of antibody in broiler breeder flocks. Avian Dis. 1989. 33:691–694.
Article12. McNulty MS, Curran WL, Todd D, Mackie DP. Chicken anemia agent: an electron microscopic study. Avian Dis. 1990. 34:736–743.
Article13. Meehan BM, Todd D, Creelan JL, Earle JA, Hoey EM, McNulty MS. Characterization of viral DNAs from cells infected with chicken anaemia agent: sequence analysis of the cloned replicative form and transfection capabilities of cloned genome fragments. Arch Virol. 1992. 124:301–319.
Article14. Noteborn MHM, de Boer GF, van Roozelaar DJ, Karreman C, Kranenburg O, Vos JG, Jeurissen SHM, Hoeben RC, Zantema A, Koch G, van Ormondt H, van der Eb AJ. Characterization of cloned chicken anemia virus DNA that contains all elements for the infectious replication cycle. J Virol. 1991. 65:3131–3139.
Article15. Pallister J, Fahey KJ, Sheppard M. Cloning and sequencing of the chicken anaemia virus (CAV) ORF-3 gene, and the development of an ELISA for the detection of serum antibody to CAV. Vet Microbiol. 1994. 39:167–178.
Article16. Schat KA. Chicken anemia virus. Curr Top Microbiol Immunol. 2009. 331:151–183.
Article17. Todd D, Creelan JL, McNulty MS. Dot blot hybridization assay for chicken anemia agent using a cloned DNA probe. J Clin Microbiol. 1991. 29:933–939.
Article18. Todd D, Mawhinney KA, McNulty MS. Detection and differentiation of chicken anemia virus isolates by using the polymerase chain reaction. J Clin Microbiol. 1992. 30:1661–1666.
Article19. Wang X, Gao H, Gao Y, Fu C, Wang Z, Lu G, Cheng Y, Wang X. Mapping of epitopes of VP2 protein of chicken anemia virus using monoclonal antibodies. J Virol Methods. 2007. 143:194–199.
Article20. Wewetzer K, Seilheimer B. Establishment of a single-step hybridoma cloning protocol using an automated cell transfer system: comparison with limiting dilution. J Immunol Methods. 1995. 179:71–76.
Article21. Yuasa N, Taniguchi T, Imada T, Hihara H. Distribution of chicken anemia agent (CAA) and detection of neutralizing antibody in chicks experimentally inoculated with CAA. Natl Inst Anim Health Q (Tokyo). 1983. 23:78–81.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Development of Peptide Antibody against Coxsackievirus B3 VP2
- Molecular characterization of chicken anemia virus in Guangxi Province, southern China, from 2018 to 2020
- Protein Expression of the Human Norovirus Capsid Gene using the Baculovirus Expression System
- Expression of human norovirus VP1 gene and VP1-specific monoclonal antibodies
- Intracellular Localization of the Japanese Encephalitis Virus Capsid Protein