J Vet Sci.
2008 Mar;9(1):39-44. 10.4142/jvs.2008.9.1.39.
The combination of deoxynivalenol and zearalenone at permitted feed concentrations causes serious physiological effects in young pigs
- Affiliations
-
- 1College of Animal Science, South China Agricultural University, Guangzhou 510642, P R China.
- 2State Key Laboratory of Biocontrol, College of Life Sciences, Sun Yat-sen University, Guangzhou 510275, P R China. caoych@mail.sysu.edu.cn
- KMID: 1102952
- DOI: http://doi.org/10.4142/jvs.2008.9.1.39
Abstract
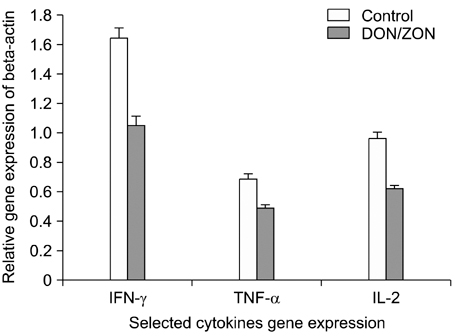
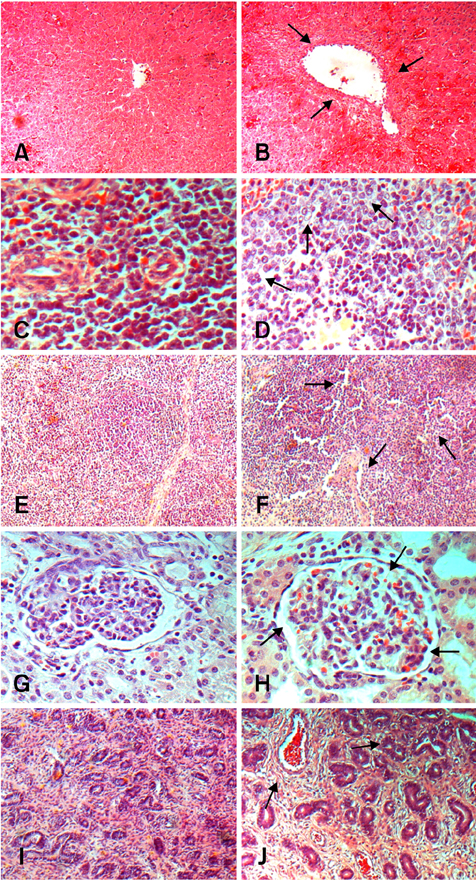
- This study was to investigate the effects of the combination of deoxynivalenol (DON) and zearalenone (ZON) on pigs. Twenty-four weaning piglets were divided into a control group fed a diet free of mycotoxins and a toxin group fed a diet containing 1 mg/kg DON and 250 microgram/kg ZON. The results showed that supplementation of DON and ZON in diets had extensive effects on pigs. More specifically, DON and ZON caused levels of total protein, albumin, and globulin in sera to decrease (p < 0.05) by 14.5%, 6.5% and 11.3%, respectively, and at the same time increased (p < 0.05) the serum enzyme activities of gamma-glutamyltransferase, aspartate aminotransferase and alanine aminotransferase by 72.0%, 32.6% and 36.6%, respectively. In addition, DON and ZON decreased (p < 0.05) the level of anticlassical swine fever antibody titers by 14.8%. Real-time PCR showed that DON and ZON caused the mRNA expression levels of IFN-gamma, TNF-alpha, IL-2, to decrease (p < 0.05) by 36.0%, 29.0% and 35.4%, respectively. Histopathological studies demonstrated that DON and ZON caused abnormalities in the liver, spleen, lymph nodes, uterus, and kidney. The concentrations of DON and ZON used in this study are in line with the published critical values permitted by BML. Our study clearly put the standard and adequacy of safety measures for these toxins into question. The authors suggest that with the increasing availability of cellular and molecular technologies, it is time to revisit the safety standards for toxins in feeds so as to make feeds safer, providing consumers with safer products.
Keyword
MeSH Terms
-
Animal Feed/*analysis
Animal Nutritional Physiological Phenomena
Animals
Diet/veterinary
Drug Therapy, Combination
Female
Swine
Swine Diseases/blood/*chemically induced/*physiopathology
Trichothecenes/*administration & dosage/*adverse effects/pharmacology
Zearalenone/*administration & dosage/*adverse effects/pharmacology
Figure
Cited by 1 articles
-
Induction of apoptotic lesions in liver and lymphoid tissues and modulation of cytokine mRNA expression by acute exposure to deoxynivalenol in piglets
Osamu Mikami, Hiroyuki Yamaguchi, Hideo Murata, Yasuyuki Nakajima, Shigeru Miyazaki
J Vet Sci. 2010;11(2):107-113. doi: 10.4142/jvs.2010.11.2.107.
Reference
-
1. Bergsjø B, Langseth W, Nafstad I, Jansen JH, Larsen HJ. The effects of naturally deoxynivalenol-contaminated oats on the clinical condition, blood parameters, performance and carcass composition of growing pigs. Vet Res Commun. 1993. 17:283–294.
Article2. Budesministerium für Ernahrung, Landwirtschaft und Forsten (BML). Orientierungswerte fur die beurteilung der gehalte an deoxynivalenol und zearalenon in futtermitteln im rahmen des § 3 des futtermittelgesetzes. 2000. Berlin: BML;1–2.3. Chang K, Kurtz HJ, Mirocha CJ. Effects of the mycotoxin zearalenone on swine reproduction. Am J Vet Res. 1979. 40:1260–1267.4. Cheng YH, Shen TF, Pang VF, Chen BJ. Effects of aflatoxin and carotenoids on growth performance and immune response in mule ducklings. Comp Biochem Physiol C Toxicol Pharmacol. 2001. 128:19–26.
Article5. Döll S, Dänicke S, Ueberschär KH, Valenta H, Schnurrbusch U, Ganter M, Klobasa F, Flachowsky G. Effects of graded levels of fusarium toxin contaminated maize in diets for female weaned piglets. Arch Tierernahr. 2003. 57:311–334.
Article6. Green ML, Stouffer DK, Scheidt AB, Long GG, Diekman MA. Evaluation of use of progesterone to counteract zearalenone toxicosis during early pregnancy in gilts. Am J Vet Res. 1991. 52:1871–1874.7. Harvey JW, Bruss M, Kaneko JJ. Clinical biochemistry of domestic animals. 1997. 5th ed. San Diego: Academic Press.8. Ihara T, Sugamata M, Sekijima M, Okumura H, Yoshino N, Ueno Y. Apoptotic cellular damage in mice after t-2 toxin-induced acute toxicosis. Nat Toxins. 1997. 5:141–145.
Article9. James LJ, Smith TK. Effect of dietary alfalfa on zearalenone toxicity and metabolism in rats and swine. J Anim Sci. 1982. 55:110–118.
Article10. Kinser S, Jia Q, Li M, Laughter A, Cornwell P, Corton JC, Pestka J. Gene expression profiling in spleens of deoxynivalenol-exposed mice: Immediate early genes as primary targets. J Toxicol Environ Health A. 2004. 67:1423–1441.
Article11. Kubena LF, Huff WE, Harvey RB, Corrier DE, Phillips TD, Creger CR. Influence of ochratoxin and deoxynivalenol on growing broiler chicks. Poult Sci. 1988. 67:253–260.12. Overnes G, Matre T, Sivertsen T, Larsen HJ, Langseth W, Reitan LJ, Jansen JH. Effects of diets with graded levels of naturally deoxynivalenol-contaminated oats on immune response in growing pigs. Zentralbl Veterinarmed A. 1997. 44:539–550.
Article13. Rotter BA, Prelusky DB, Pestka JJ. Toxicology of deoxynivalenol (vomitoxin). J Toxicol Environ Health. 1996. 48:1–34.14. Rotter BA, Thompson BK, Clarkin S, Owen TC. Rapid colorimetric bioassay for screening of fusarium mycotoxins. Nat Toxins. 1993. 1:303–307.
Article15. Rotter BA, Thompson BK, Lessard M, Trenholm HL, Tryphonas H. Influence of low-level exposure to fusarium mycotoxins on selected immunological and hematological parameters in young swine. Fundam Appl Toxicol. 1994. 23:117–124.
Article16. Shifrin VI, Anderson P. Trichothecene mycotoxins trigger a ribotoxic stress response that activates c-jun n-terminal kinase and p38 mitogen-activated protein kinase and induces apoptosis. J Biol Chem. 1999. 274:13985–13992.
Article17. Tiemann U, Brüssow KP, Küchenmeister U, Jonas L, Kohlschein P, Pöhland R, Dänicke S. Influence of diets with cereal grains contaminated by graded levels of two fusarium toxins on selected enzymatic and histological parameters of liver in gilts. Food Chem Toxicol. 2006. 44:1228–1235.
Article18. Ueno Y. Ueno Y, editor. General toxicology. Trichothecene - Chemical, Biological and Toxicological Aspects. 1983. Amsterdam: Elsevier;135–146.19. Zhou HR, Yan D, Pestka JJ. Differential cytokine mrna expression in mice after oral exposure to the trichothecene vomitoxin (deoxynivalenol): Dose response and time course. Toxicol Appl Pharmacol. 1997. 144:294–305.
Article20. Zhou HR, Yan D, Pestka JJ. Induction of cytokine gene expression in mice after repeated and subchronic oral exposure to vomitoxin (deoxynivalenol): Differential toxin-induced hyporesponsiveness and recovery. Toxicol Appl Pharmacol. 1998. 151:347–358.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of barley and barley bran contaminated with Fusarium spp. on the growth and feed efficiency of fattening and growing pigs
- A novel mycotoxin purification system using magnetic nanoparticles for the recovery of aflatoxin B1 and zearalenone from feed
- Effects of a herbal compound, KIOM-C, on growth performance and immune response in commercial pigs
- Effects of gender and gonadectomy on growth and plasma cholesterol levels in pigs
- Production of a highly group-specific monoclonal antibody against zearalenone and its application in an enzyme-linked immunosorbent assay