J Vet Sci.
2006 Sep;7(3):211-216. 10.4142/jvs.2006.7.3.211.
Altered expression of thioredoxin reductase-1 in dysplastic bile ducts and cholangiocarcinoma in a hamster model
- Affiliations
-
- 1School of Veterinary Medicine and Institute of Veterinary Science, Kangwon National University, Chuncheon 200-701, Korea. byoon@kangwon.ac.kr
- 2Department of Veterinary Pathology, College of Veterinary Medicine, Seoul National University, Seoul 151-742, Korea.
- 3Department of Pathology, College of Medicine, Seoul National University, Seoul 110-799, Korea.
- KMID: 1089903
- DOI: http://doi.org/10.4142/jvs.2006.7.3.211
Abstract
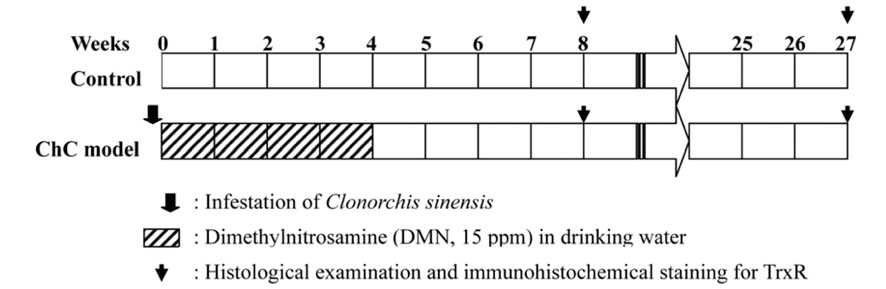
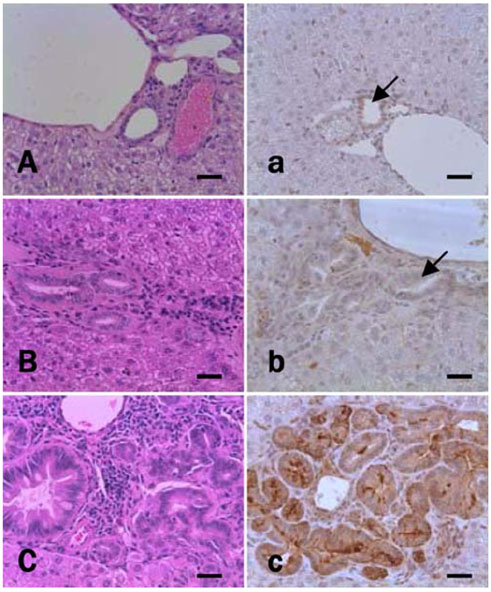
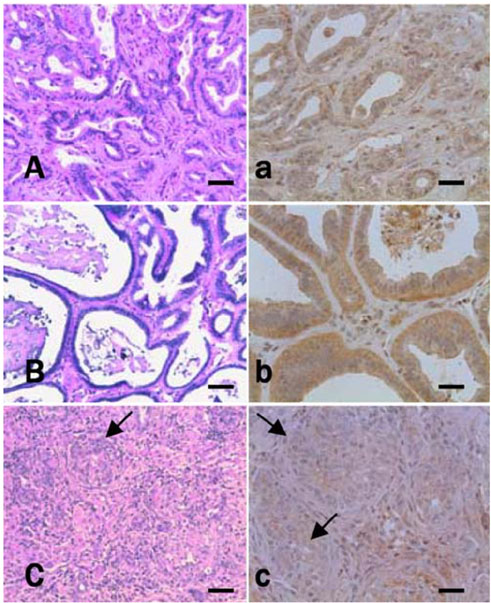
- Thioredoxin reductase 1 (TrxR) is a homodimeric selenoenzyme catalyzing thioredoxin (Trx) in an NADPHdependent manner. With regard to carcinogenesis, these redox proteins have been implicated in cell proliferation, transformation and anti-apoptosis. In the present study, using a hamster cholangiocarcinoma (ChC) model, we evaluated the immunohistochemical expression pattern of TrxR in precancerous lesions and ChCs as well as in normal bile ducts. The goal of this study was to determine the potential role and importance of TrxR in cholangiocarcinogenesis. For the ChC model, we obtained liver tissue specimens with dysplastic bile ducts prior to the development of ChC 8 weeks after initiation of the experiment and ChC samples at 27 weeks. The immunohistochemical analysis showed diffuse cytoplasmic overexpression of TrxR in the dysplastic bile duct epithelial cells as well as in cholangiocarcinoma; this was comparable to the negative or weakly positive in normal and type 1 hyperplastic bile ducts. However, TrxR appeared to be considerably down-regulated in the ChCs when compared to the higher expression observed in the dysplastic bile ducts. Therefore, these results suggest that TrxR overexpression followed by down-regulation might be an important event in cholangiocarcinogenesis, especially at early stages including the cellular transformation of candidate bile ducts. Further studies are however required to determine whether TrxR may be a potential target molecule for chemoprevention against cholangiocarcinogenesis. In addition, the molecular mechanism as well as the importance of the loss of TrxR in the development of cholangiocarcinoma, following dysplastic transformation of bile duct cells, also remains to be clarified.
MeSH Terms
Figure
Reference
-
1. Becker K, Gromer S, Schirmer RH, Müller S. Thioredoxin reductase as a pathophysiological factor and drug target. Eur J Biochem. 2000. 267:6118–6125.
Article2. Berggren M, Gallegos A, Gasdaska JR, Gasdaska PY, Warneke J, Powis G. Thioredoxin and thioredoxin reductase gene expression in human tumors and cell lines, and the effects of serum stimulation and hypoxia. Anticancer Res. 1996. 16:3459–3466.3. Biaglow JE, Miller RA. The thioredoxin reductase/thioredoxin system: Novel redox targets for cancer therapy. Cancer Biol Ther. 2005. 4:6–13.
Article4. Choi JH, Kim TN, Kim S, Baek SH, Kim JH, Lee SR, Kim JR. Overexpression of mitochondrial thioredoxin reductase and peroxiredoxin III in hepatocellular carcinomas. Anticancer Res. 2002. 22:3331–3335.5. Engman L, Al-Maharik N, McNaughton M, Birmingham A, Powis G. Thioredoxin reductase and cancer cell growth inhibition by organotellurium compounds that could be selectively incorporated into tumor cells. Bioorg Med Chem. 2003. 11:5091–5100.
Article6. Engman L, Al-Maharik N, McNaughton M, Birmingham A, Powis G. Thioredoxin reductase and cancer cell growth inhibition by organotellurium antioxidants. Anticancer Drugs. 2003. 14:153–161.
Article7. Gius D, Botero A, Shah S, Curry HA. Intracellular oxidation/reduction status in the regulation of transcription factors NF-kappa B and AP-1. Toxicol Lett. 1999. 106:93–106.
Article8. Kahlos K, Soini Y, Saily M, Koistinen P, Kakko S, Paakko P, Holmgren A, Kinnula VL. Up-regulation of thioredoxin and thioredoxin reductase in human malignant pleural mesothelioma. Int J Cancer. 2001. 95:198–204.
Article9. Khan SA, Thomas HC, Davidson BR, Taylor-Robison SD. Cholangiocarcinoma. Lancet. 2005. 366:1303–1314.
Article10. Kunkel MW, Kirkpatrick DL, Johnson JI, Powis G. Cell line-directed screening assay for inhibitors of thioredoxin reductase signaling as potential anti-cancer drugs. Anticancer Drug Des. 1997. 12:659–670.11. Lechner S, Müller-Ladner U, Neumann E, Spöttl T, Schlottmann K, Rüschoff J, Schölmerich J, Kullmann F. Thioredoxin reductase 1 expression in colon cancer: discrepancy between in vitro and in vivo findings. Lab Invest. 2003. 83:1321–1331.
Article12. Lee JH, Rim HJ, Sell S. Heterogeneity of the "oval-cell" response in the hamster liver during cholangiocarcinogenesis following Clonorchis sinensis infection and dimethylnitrosamine treatment. J Hepatol. 1997. 26:1313–1323.
Article13. Lincoln DT, Ali Emadi EM, Tonissen KF, Clarke FM. The thioredoxin-thioredoxin reductase system: over-expression in human cancer. Anticancer Res. 2003. 23:2425–2433.14. Mau BL, Powis G. Inhibition of cellular thioredoxin reductase by diaziquone and doxorubicin. Biochem Pharmacol. 1992. 43:1621–1627.
Article15. Mau BL, Powis G. Mechanism-based inhibition of thioredoxin reductase by antitumor quinoid compounds. Biochem Pharmacol. 1992. 43:1613–1620.
Article16. Mustacich D, Powis G. Thioredoxin reductase. Biochem J. 2000. 346:1–8.
Article17. Pinlaor S, Hiraku Y, Ma N, Yongvanit P, Semba R, Oikawa S, Murata M, Sripa B, Sithithaworn P, Kawanishi S. Mechanism of NO-mediated oxidative and nitrative DNA damage in hamsters infected with Opisthorchis viverrini: a model of inflammation-mediated carcinogenesis. Nitric Oxide. 2004. 11:175–183.
Article18. Powis G, Briehl M, Oblong J. Redox signalling and the control of cell growth and death. Pharmacol Ther. 1995. 68:149–173.
Article19. Powis G, Kirkpatrick DL, Angulo M, Baker A. Thioredoxin redox control of cell growth and death and the effects of inhibitors. Chem Biol Interact. 1998. 111-112:23–34.
Article20. Powis G, Montfort WR. Properties and biological activities of thioredoxins. Annu Rev Biophys Biomol Struct. 2001. 30:421–455.
Article21. Powis G, Oblong JE, Gasdaska PY, Berggren M, Hill SR, Kirkpatrick DL. The thioredoxin/thioredoxin reductase redox system and control of cell growth. Oncol Res. 1994. 6:539–544.22. Sirica AE. Cholangiocarcinoma: Molecular targeting strategies for chemoprevention and therapy. Hepatology. 2005. 41:5–15.
Article23. Sirica AE, Lai GH, Endo K, Zhang Z, Yoon BI. Cyclooxygenase-2 and ERBB-2 in cholangiocarcinoma: potential therapeutic targets. Semin Liver Dis. 2002. 22:303–313.
Article24. Smart DK, Ortiz KL, Mattson D, Bradbury CM, Bisht KS, Sieck LK, Brechbiel MW, Gius D. Thioredoxin reductase as a potential molecular target for anticancer agents that induce oxidative stress. Cancer Res. 2004. 64:6716–6724.
Article25. Soini Y, Kahlos K, Näpänkangas U, Kaarteenaho-Wiik R, Säily M, Koistinen P, Pääkkö P, Holmgren A, Kinnula VL. Widespread expression of thioredoxin and thioredoxin reductase in non-small cell lung carcinoma. Clin Cancer Res. 2001. 7:1750–1757.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Experimental Animal Model of Biliary Tract Diseases
- Hilar Cholangiocarcinoma
- Glutathione Reductase and Thioredoxin Reductase: Novel Antioxidant Enzymes from Plasmodium berghei
- Hilar Cholangiocarcinoma: Recent update of radiologic assessment
- Histopathology of a benign bile duct lesion in the liver: Morphologic mimicker or precursor of intrahepatic cholangiocarcinoma