J Vet Sci.
2010 Dec;11(4):333-340. 10.4142/jvs.2010.11.4.333.
Sequence and phylogenetic analysis of the gp200 protein of Ehrlichia canis from dogs in Taiwan
- Affiliations
-
- 1Department of Veterinary Medicine, College of Veterinary Medicine, National Chung Hsing University, Taichung 402, Taiwan. ytchung@dragon.nchu.edu.tw
- KMID: 1072183
- DOI: http://doi.org/10.4142/jvs.2010.11.4.333
Abstract
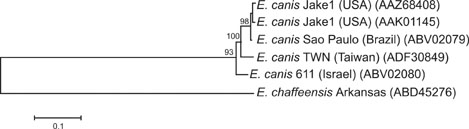
- Ehrlichia (E.) canis is a Gram-negative obligate intracellular bacterium responsible for canine monocytic ehrlichiosis. Currently, the genetic diversity of E. canis strains worldwide is poorly defined. In the present study, sequence analysis of the nearly full-length 16S rDNA (1,620 bp) and the complete coding region (4,269 bp) of the gp200 gene, which encodes the largest major immunoreactive protein in E. canis, from 17 Taiwanese samples was conducted. The resultant 16S rDNA sequences were found to be identical to each other and have very high homology (99.4~100%) with previously reported E. canis sequences. Additionally, phylogenetic analysis of gp200 demonstrated that the E. canis Taiwanese genotype was genetically distinct from other reported isolates obtained from the United States, Brazil, and Israel, and that it formed a separate clade. Remarkable variations unique to the Taiwanese genotype were found throughout the deduced amino acid sequence of gp200, including 15 substitutions occurring in two of five known species-specific epitopes. The gp200 amino acid sequences of the Taiwanese genotype bore 94.4~94.6 identities with those of the isolates from the United States and Brazil, and 93.7% homology with that of the Israeli isolate. Taken together, these results suggest that the Taiwanese genotype represents a novel strain of E. canis that has not yet been characterized.
MeSH Terms
Figure
Reference
-
1. de Castro MB, Machado RZ, de Aquino LP, Alessi AC, Costa MT. Experimental acute canine monocytic ehrlichiosis: clinicopathological and immunopathological findings. Vet Parasitol. 2004. 119:73–86.
Article2. Groves MG, Dennis GL, Amyx HL, Huxsoll DL. Transmission of Ehrlichia canis to dogs by ticks (Rhipicephalus sanguineus). Am J Vet Res. 1975. 36:937–940.3. Harrus S, Aroch I, Lavy E, Bark H. Clinical manifestations of infectious canine cyclic thrombocytopenia. Vet Rec. 1997. 141:247–250.
Article4. Harrus S, Waner T, Bark H, Jongejan F, Cornelissen AWCA. Recent advances in determining the pathogenesis of canine monocytic ehrlichiosis. J Clin Microbiol. 1999. 37:2745–2749.
Article5. Huxsoll DL, Hildebrandt PK, Nims RM, Walker JS. Tropical canine pancytopenia. J Am Vet Med Assoc. 1970. 157:1627–1632.6. Komnenou AA, Mylonakis ME, Kouti V, Tendoma L, Leontides L, Skountzou E, Dessiris A, Koutinas AF, Ofri R. Ocular manifestations of natural canine monocytic ehrlichiosis (Ehrlichia canis): a retrospective study of 90 cases. Vet Ophthalmol. 2007. 10:137–142.
Article7. Luo T, Zhang X, Nicholson WL, Zhu B, McBride JW. Molecular characterization of antibody epitopes of Ehrlichia chaffeensis ankyrin protein 200 and tandem repeat protein 47 and evaluation of synthetic immunodeterminants for serodiagnosis of human monocytotropic ehrlichiosis. Clin Vaccine Immunol. 2010. 17:87–97.
Article8. Mavromatis K, Doyle CK, Lykidis A, Ivanova N, Francino MP, Chain P, Shin M, Malfatti S, Larimer F, Copeland A, Detter JC, Land M, Richardson PM, Yu XJ, Walker DH, McBride JW, Kyrpides NC. The genome of the obligately intracellular bacterium Ehrlichia canis reveals themes of complex membrane structure and immune evasion strategies. J Bacteriol. 2006. 188:4015–4023.
Article9. McBride JW, Comer JE, Walker DH. Novel Immunoreactive glycoprotein orthologs of Ehrlichia spp. Ann N Y Acad Sci. 2003. 990:678–684.10. McBride JW, Corstvet RE, Gaunt SD, Boudreaux C, Guedry T, Walker DH. Kinetics of antibody response to Ehrlichia canis immunoreactive proteins. Infect Immun. 2003. 71:2516–2524.
Article11. Mylonakis ME, Koutinas AF, Breitschwerdt EB, Hegarty BC, Billinis CD, Leontides LS, Kontos VS. Chronic canine ehrlichiosis (Ehrlichia canis): a retrospective study of 19 natural cases. J Am Anim Hosp Assoc. 2004. 40:174–184.
Article12. Neer TM, Breitschwerdt EB, Greene RT, Lappin MR. Consensus statement on ehrlichial disease of small animals from the infectious disease study group of the ACVIM. J Vet Intern Med. 2002. 16:309–315.
Article13. Neer TM, Harrus S. Greene CE, editor. Canine monocytotropic ehrlichiosis and neorickettsiosis (E. canis, E. chaffeensis, E. ruminatium, N. sennetsu and N. risticii infection). Infectious Diseases of the Dog and Cat. 2006. St. Louis: Saunders;203–217.14. Nethery KA, Doyle CK, Zhang X, McBride JW. Ehrlichia canis gp200 contains dominant species-specific antibody epitopes in terminal acidic domains. Infect Immun. 2007. 75:4900–4908.
Article15. Pinyoowong D, Jittapalapong S, Suksawat F, Stich RW, Thamchaipenet A. Molecular characterization of Thai Ehrlichia canis and Anaplasma platys strains detected in dogs. Infect Genet Evol. 2008. 8:433–438.
Article16. Siarkou VI, Mylonakis ME, Bourtzi-Hatzopoulou E, Koutinas AF. Sequence and phylogenetic analysis of the 16S rRNA gene of Ehrlichia canis strains in dogs with clinical monocytic ehrlichiosis. Vet Microbiol. 2007. 125:304–312.
Article17. Tamura K, Dudley J, Nei M, Kumar S. MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol. 2007. 24:1596–1599.
Article18. Thompson JD, Higgins DG, Gibson TJ. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994. 22:4673–4680.
Article19. Unver A, Huang H, Rikihisa Y. Cytokine gene expression by peripheral blood leukocytes in dogs experimentally infected with a new virulent strain of Ehrlichia canis. Ann N Y Acad Sci. 2006. 1078:482–486.
Article20. Unver A, Rikihisa Y, Kawahara M, Yamamoto S. Analysis of 16S rRNA gene sequences of Ehrlichia canis, Anaplasma platys, and Wolbachia species from canine blood in Japan. Ann N Y Acad Sci. 2003. 990:692–698.
Article21. Vinasco J, Li O, Alvarado A, Diaz D, Hoyos L, Tabachi L, Sirigireddy K, Ferguson C, Moro MH. Molecular evidence of a new strain of Ehrlichia canis from South America. J Clin Microbiol. 2007. 45:2716–2719.
Article22. Walker JS, Rundquist JD, Taylor R, Wilson BL, Andrews MR, Barck J, Hogge AL Jr, Huxsoll DL, Hildebrandt PK, Nims RM. Clinical and clinicopathologic findings in tropical canine pancytopenia. J Am Vet Med Assoc. 1970. 157:43–55.23. Yu XJ, McBride JW, Walker DH. Restriction and expansion of Ehrlichia strain diversity. Vet Parasitol. 2007. 143:337–346.24. Zhang X, Luo T, Keysary A, Baneth G, Miyashiro S, Strenger C, Waner T, McBride JW. Genetic and antigenic diversities of major immunoreactive proteins in globally distributed Ehrlichia canis strains. Clin Vaccine Immunol. 2008. 15:1080–1088.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Isolation, in vitro propagation, genetic analysis, and immunogenic characterization of an Ehrlichia canis strain from southeastern Brazil
- Detection and molecular characterization of Hepatozoon canis, Babesia vogeli, Ehrlichia canis, and Anaplasma platys in dogs from Metro Manila, Philippines
- Serological evidence of Anaplasma spp., Borrelia burgdorferi and Ehrlichia canis in dogs from the Republic of Korea by rapid diagnostic test kits
- Prevalence of Toxocara canis in Dogs, North West Bank of Palestine
- Genetic Identification and Phylogenetic Analysis of Anaplasma and Ehrlichia Species in Haemaphysalis longicornis Collected from Jeju Island, Korea