J Vet Sci.
2006 Jun;7(2):105-109. 10.4142/jvs.2006.7.2.105.
Topical application of epidermal growth factor accelerates wound healing by myofibroblast proliferation and collagen synthesis in rat
- Affiliations
-
- 1Department of Pharmacology, Chonbuk National University Medical School, Jeonju 561-756, Korea.
- 2Department of Veterinary Physiology, College of Veterinary Medicine and School of Agricultural Biotechnology, Seoul National University, Seoul 151-742, Korea. JHL1101@snu.ac.kr
- 3Department of Plastic Surgery, College of Medicine, Seoul National University, Seoul 110-744, Korea.
- 4R&D Center, Daewoong Pharmaceutical Company, Yongin 449-814, Korea.
- 5Department of Agricultural Biology, National Institute of Agricultural Science and Technology, Suwon 441-100, Korea.
- 6School of Biological Resources and Materials Engineering, College of Agriculture and Life Sciences, Seoul National University, Seoul 151-742, Korea.
- KMID: 1059193
- DOI: http://doi.org/10.4142/jvs.2006.7.2.105
Abstract
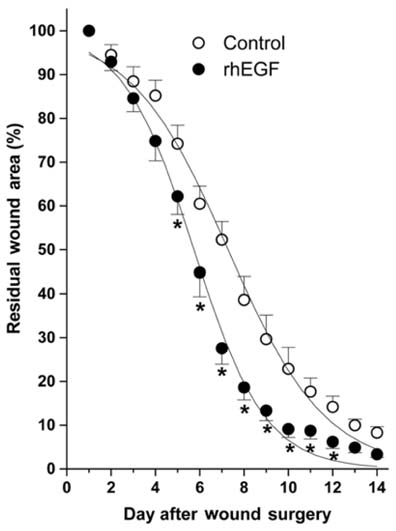
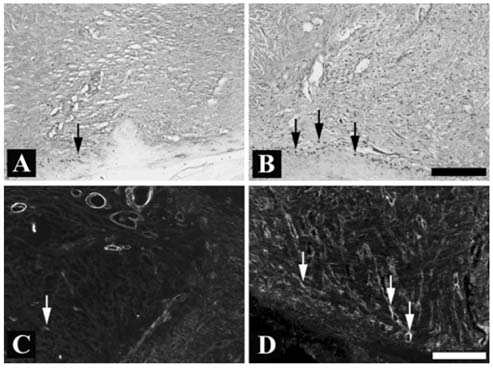
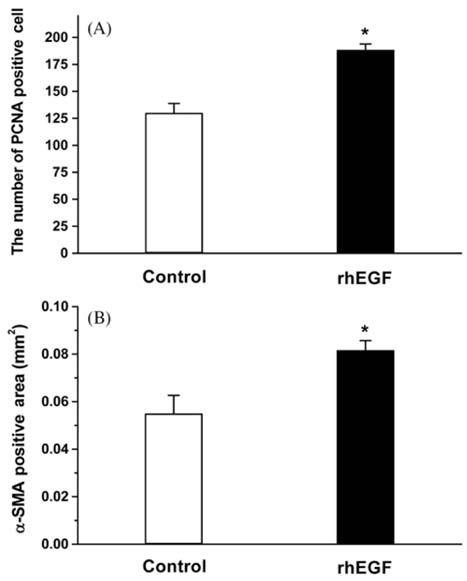
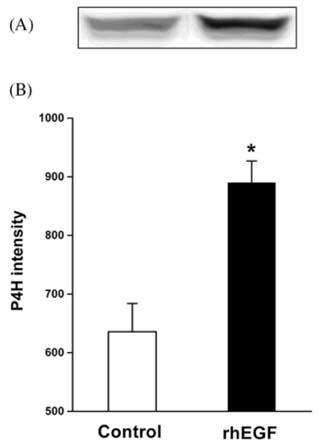
- Recombinant human epidermal growth factor (rhEGF) stimulates the proliferation and migration of epithelial cells in human cell culture systems and animal models of partial-thickness skin wounds. This study investigated the effect of a topical rhEGF ointment on the rate of wound healing and skin re-epithelialization in a rat full thickness wound model, and verified whether or not the rhEGF treatment affected both myofibroblast proliferation and collagen synthesis in the dermis. When rhEGF (10 microgram/g ointment) was applied topically twice a day for 14 days, there was significantly enhanced wound closure from the 5th to the 12th day compared with the control (ointment base treatment) group. A histological examination at the postoperative 7th day revealed that the rhEGF treatment increased the number of proliferating nuclear antigen immunoreactive cells in the epidermis layer. In addition, the immunoreactive area of alpha-smooth muscle actin and the expression of prolyl 4-hydroxylase were significantly higher than those of the control group. Overall, a topical treatment of rhEGF ointment promotes wound healing by increasing the rate of epidermal proliferation and accelerating the level of wound contraction related to myofibroblast proliferation and collagen deposition.
Keyword
MeSH Terms
-
Actins/genetics/metabolism
Administration, Topical
Animals
Cell Proliferation/drug effects
Collagen/*biosynthesis
Epidermal Growth Factor/*administration&dosage/*pharmacology
Gene Expression Regulation
Male
Myoblasts, Skeletal/*drug effects
Proliferating Cell Nuclear Antigen/genetics/metabolism
Rats
Rats, Sprague-Dawley
Wound Healing/*drug effects
Figure
Reference
-
1. Babul A, Gonul B, Dincer S, Erdogan D, Ozogul C. The effect of EGF application in gel form on histamine content of experimentally induced wound in mice. Amino Acids. 2004. 27:321–326.
Article2. Breuing K, Andree C, Helo G, Slama J, Liu PY, Eriksson E. Growth factors in the repair of partial thickness porcine skin wounds. Plast Reconstr Surg. 1997. 100:657–664.
Article3. Brown GL, Curtsinger L, Brightwell JR, Ackerman DM, Tobin GR, Polk HC Jr, George-Nascimento C, Valenzuela P, Schultz GS. Enhancement of epidermal regeneration by biosynthetic epidermal growth factor. J Exp Med. 1986. 163:1319–1324.
Article4. Buckley A, Davidson JM, Kamerath CD, Woodward SC. Epidermal growth factor increases granulation tissue formation dose dependently. J Surg Res. 1987. 43:322–328.
Article5. Buckley A, Davidson JM, Kamerath CD, Wolt TB, Woodward SC. Sustained release of epidermal growth factor accelerates wound repair. Proc Natl Acad Sci USA. 1985. 82:7340–7344.
Article6. Darby I, Skalli O, Gabbiani G. Alpha-smooth muscle actin is transiently expressed by myofibroblasts during experimental wound healing. Lab Invest. 1990. 63:21–29.7. Gabbiani G. The myofibroblast in wound healing and fibrocontractive diseases. J Pathol. 2003. 200:500–503.
Article8. Gope R. The effect of epidermal growth factor & platelet-derived growth factors on wound healing process. Indian J Med Res. 2002. 116:201–206.9. Hall PA, Levison DA, Woods AL, Yu CC, Kellock DB, Watkins JA, Barnes DM, Gillett CE, Camplejohn R, Dover R. Proliferating cell nuclear antigen (PCNA) immunolocalization in paraffin sections: an index of cell proliferation with evidence of deregulated expression in some neoplasms. J Pathol. 1990. 162:285–294.
Article10. Hergott GJ, Kalnins VI. Expression of proliferating cell nuclear antigen in migrating retinal pigment epithelial cells during wound healing in organ culture. Exp Cell Res. 1991. 195:307–314.
Article11. Kim HW, Kwon YB, Ham TW, Roh DH, Yoon SY, Lee HJ, Han HJ, Yang IS, Beitz AJ, Lee JH. Acupoint stimulation using bee venom attenuates formalin-induced pain behavior and spinal cord fos expression in rats. J Vet Med Sci. 2003. 65:349–355.
Article12. Kim JS, McKinnis VS, Adams K, White SR. Proliferation and repair of guinea pig tracheal epithelium after neuropeptide depletion and injury in vivo. Am J Physiol. 1997. 273:L1235–L1241.
Article13. Laato M. Effect of epidermal growth factor (EGF) on blood flow and albumin extravasation in experimental granulation tissue. Acta Chir Scand. 1986. 152:401–405.14. Lee J. Formulation development of epidermal growth factor. Pharmazie. 2002. 57:787–790.15. LeGrand EK, Burke JF, Costa DE, Kiorpes TC. Dose responsive effects of PDGF-BB, PDGF-AA, EGF, and bFGF on granulation tissue in a guinea pig partial thickness skin excision model. Growth Factors. 1993. 8:307–314.
Article16. Liu M, Warn JD, Fan Q, Smith PG. Relationships between nerves and myofibroblasts during cutaneous wound healing in the developing rat. Cell Tissue Res. 1999. 297:423–433.
Article17. Nanney LB. Epidermal and dermal effects of epidermal growth factor during wound repair. J Invest Dermatol. 1990. 94:624–629.
Article18. Okumura K, Kiyohara Y, Komada F, Iwakawa S, Hirai M, Fuwa T. Improvement in wound healing by epidermal growth factor (EGF) ointment. I. Effect of nafamostat, gabexate, or gelatin on stabilization and efficacy of EGF. Pharm Res. 1990. 7:1289–1293.19. Park JS, Kim JY, Cho JY, Kang JS, Yu YH. Epidermal growth factor (EGF) antagonizes transforming growth factor (TGF)-beta1-induced collagen lattice contraction by human skin fibroblasts. Biol Pharm Bull. 2000. 23:1517–1520.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Epidermal Growth Factor in Wound Healing
- Effect of Epidermal Growth Factor on the Wound Healing after Excimer Laser Photokeratectomy in Rabbits
- Effects of Topical Application of Halofuginone on Wound Healing
- The effect of epidermal growth factor in wound healing of skin defect
- The Accelerated Epithelization of Recombinant Epidermal Growth Factor on Partial-thickness Skin Wounds