Yonsei Med J.
2011 Nov;52(6):990-998. 10.3349/ymj.2011.52.6.990.
Immunotherapy of Malignant Melanoma with Tumor Lysate-Pulsed Autologous Monocyte-Derived Dendritic Cells
- Affiliations
-
- 1Department of Dermatology and Cutaneous Biology Research Institute, Brain Korea 21 Project for Medical Science, Yonsei University College of Medicine, Seoul, Korea. mglee@yuhs.ac
- 2Department of Dermatology, Bundang CHA Medical Center, CHA University, Seongnam, Korea.
- 3Department of Laboratory Medicine, Yonsei Cell Therapy Center, Yonsei University College of Medicine, Seoul, Korea.
- 4Clinical Research Institute, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 5Division of Nuclear Medicine, Department of Diagnostic Radiology, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 1058821
- DOI: http://doi.org/10.3349/ymj.2011.52.6.990
Abstract
- PURPOSE
Dendritic cell (DC) vaccination for melanoma was introduced because melanoma carries distinct tumor-associated antigens. The purpose of this study was to investigate the efficacy and safety of DC vaccination for melanoma in Korea.
MATERIALS AND METHODS
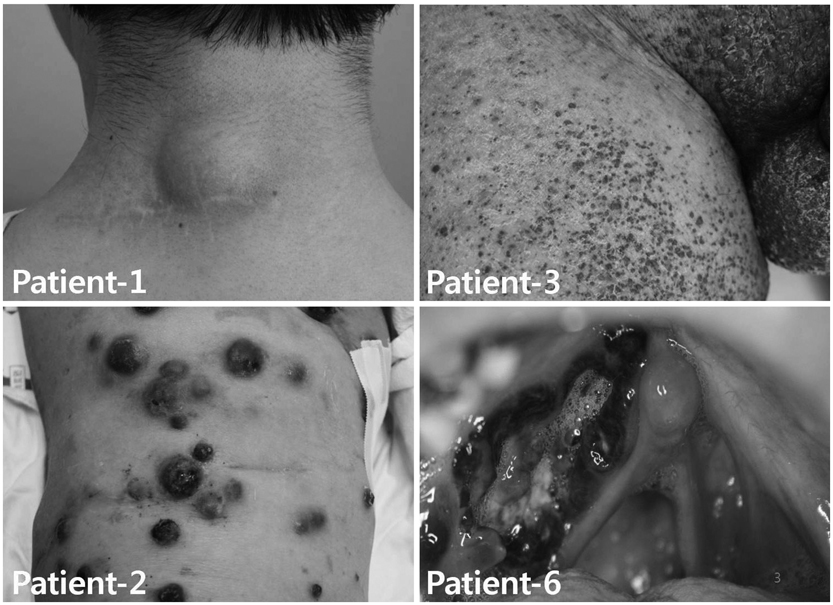
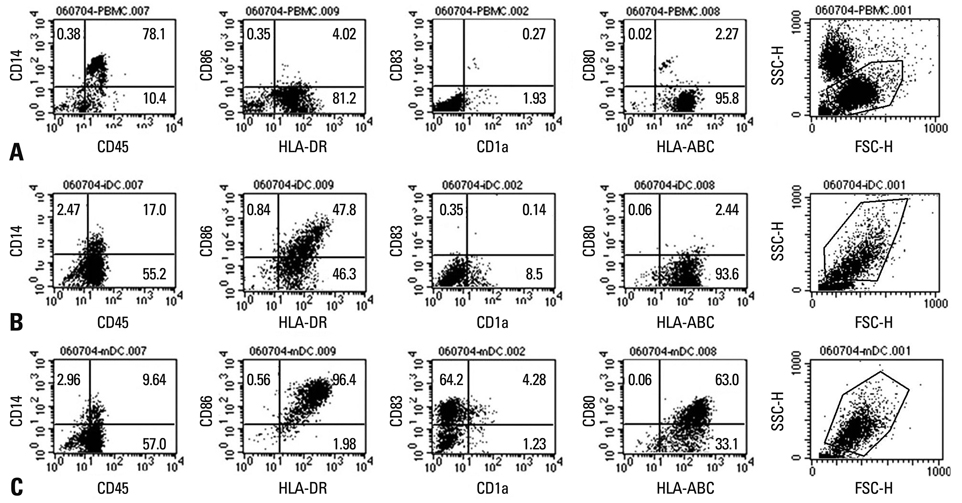
Five patients with stage IV and one with stage II were enrolled. Autologous monocyte-derived DCs (MoDCs) were cultured and pulsed with tumor-lysate, keyhole limpet hemocyanin, and cytokine cocktail for mature antigen-loaded DC. DC vaccination was repeated four times at 2-week intervals and 2-4x107 DC were injected each time.
RESULTS
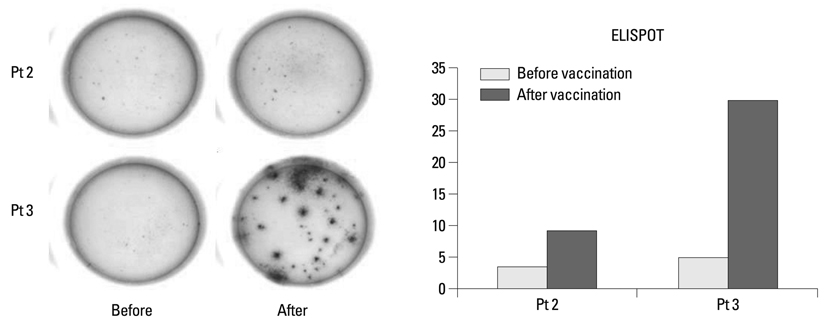
Reduced tumor volume was observed by PET-CT in three patients after DC vaccination. Delayed type hypersensitivity responses against tumor antigen were induced in five patients. Tumor antigen-specific IFN-gamma-producing peripheral blood mononuclear cells were detected with enzyme-linked immunosorbent spot in two patients. However, the overall clinical outcome showed disease progression in all patients.
CONCLUSION
In this study, DC vaccination using tumor antigen-loaded, mature MoDCs led to tumor regression in individual melanoma patients. Further standardization of DC vaccination protocol is required to determine which parameters lead to better anti-tumor responses and clinical outcomes.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Centennial History of Yonsei University Dermatology in Korea: 1917 to 2017
Jihee Kim, Tae-Gyun Kim, Si Hyung Lee, Min Kyung Lee, Jong Hoon Kim, Sang Eun Lee, Do Young Kim, Mi Ryung Roh, Chang Ook Park, Ju Hee Lee, Min-Geol Lee, Dongsik Bang, Sang Ho Oh, Kee Yang Chung
Ann Dermatol. 2018;30(5):513-521. doi: 10.5021/ad.2018.30.5.513.
Reference
-
1. Hall HI, Miller DR, Rogers JD, Bewerse B. Update on the incidence and mortality from melanoma in the United States. J Am Acad Dermatol. 1999. 40:35–42.
Article2. Houghton AN, Gold JS, Blachere NE. Immunity against cancer: lessons learned from melanoma. Curr Opin Immunol. 2001. 13:134–140.
Article3. de Braud F, Khayat D, Kroon BB, Valdagni R, Bruzzi P, Cascinelli N. Malignant melanoma. Crit Rev Oncol Hematol. 2003. 47:35–63.
Article4. Hsu FJ, Benike C, Fagnoni F, Liles TM, Czerwinski D, Taidi B, et al. Vaccination of patients with B-cell lymphoma using autologous antigen-pulsed dendritic cells. Nat Med. 1996. 2:52–58.
Article5. Schuler G, Schuler-Thurner B, Steinman RM. The use of dendritic cells in cancer immunotherapy. Curr Opin Immunol. 2003. 15:138–147.
Article6. Kim S, Kim HO, Baek EJ, Choi Y, Kim HS, Lee MG. Monocyte enrichment from leukapheresis products by using the Elutra cell separator. Transfusion. 2007. 47:2290–2296.
Article7. Eubel J, Enk AH. Dendritic cell vaccination as a treatment modality for melanoma. Expert Rev Anticancer Ther. 2009. 9:1631–1642.
Article8. Nakai N, Asai J, Ueda E, Takenaka H, Katoh N, Kishimoto S. Vaccination of Japanese patients with advanced melanoma with peptide, tumor lysate or both peptide and tumor lysate-pulsed mature, monocyte-derived dendritic cells. J Dermatol. 2006. 33:462–472.
Article9. López MN, Pereda C, Segal G, Muñoz L, Aguilera R, González FE, et al. Prolonged survival of dendritic cell-vaccinated melanoma patients correlates with tumor-specific delayed type IV hypersensitivity response and reduction of tumor growth factor beta-expressing T cells. J Clin Oncol. 2009. 27:945–952.
Article10. Jonuleit H, Schmitt E, Stassen M, Tuettenberg A, Knop J, Enk AH. Identification and functional characterization of human CD4(+)CD25(+) T cells with regulatory properties isolated from peripheral blood. J Exp Med. 2001. 193:1285–1294.
Article11. Liyanage UK, Moore TT, Joo HG, Tanaka Y, Herrmann V, Doherty G, et al. Prevalence of regulatory T cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J Immunol. 2002. 169:2756–2761.
Article12. Curiel TJ, Coukos G, Zou L, Alvarez X, Cheng P, Mottram P, et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med. 2004. 10:942–949.
Article13. Mahnke K, Schmitt E, Bonifaz L, Enk AH, Jonuleit H. Immature, but not inactive: the tolerogenic function of immature dendritic cells. Immunol Cell Biol. 2002. 80:477–483.
Article14. Dannull J, Su Z, Rizzieri D, Yang BK, Coleman D, Yancey D, et al. Enhancement of vaccine-mediated antitumor immunity in cancer patients after depletion of regulatory T cells. J Clin Invest. 2005. 115:3623–3633.
Article15. Mahnke K, Schönfeld K, Fondel S, Ring S, Karakhanova S, Wiedemeyer K, et al. Depletion of CD4+CD25+ human regulatory T cells in vivo: kinetics of Treg depletion and alterations in immune functions in vivo and in vitro. Int J Cancer. 2007. 120:2723–2733.
Article16. Rasku MA, Clem AL, Telang S, Taft B, Gettings K, Gragg H, et al. Transient T cell depletion causes regression of melanoma metastases. J Transl Med. 2008. 6:12.
Article17. Attia P, Maker AV, Haworth LR, Rogers-Freezer L, Rosenberg SA. Inability of a fusion protein of IL-2 and diphtheria toxin (Denileukin Diftitox, DAB389IL-2, ONTAK) to eliminate regulatory T lymphocytes in patients with melanoma. J Immunother. 2005. 28:582–592.
Article18. Lee TH, Cho HK, Cho YH, Lee MG. Development of an effective method for dendritic cell immunotherapy of mouse melanoma. Scand J Immunol. 2009. 70:85–92.
Article19. Jonuleit H, Giesecke-Tuettenberg A, Tuting T, Thurner-Schuler B, Stuge TB, Paragnik L, et al. A comparison of two types of dendritic cell as adjuvants for the induction of melanoma-specific T-cell responses in humans following intranodal injection. Int J Cancer. 2001. 93:243–251.
Article20. Tuettenberg A, Becker C, Huter E, Knop J, Enk AH, Jonuleit H. Induction of strong and persistent MelanA/MART-1-specific immune responses by adjuvant dendritic cell-based vaccination of stage II melanoma patients. Int J Cancer. 2006. 118:2617–2627.
Article21. Whiteside TL. The tumor microenvironment and its role in promoting tumor growth. Oncogene. 2008. 27:5904–5912.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Dendritic Cell Based Cancer Vaccine Using Allogeneic Tumor Cell Lysate in Melanoma Pulmonary Metastasis Model
- Immunotherapy for Malignant Melanoma using Dendritic Cells
- Anti-cancer Effect of Hematopoietic Stem Cell-derived Allogeneic-DC Vaccine in Melanoma Metastasis Model
- Induction of 90K-specific Cytotoxic T Lymphocytes for Colon Cancer Immunotherapy
- Immunocell Therapy for Lung Cancer: Dendritic Cell Based Adjuvant Therapy in Mouse Lung Cancer Model