Korean J Ophthalmol.
2005 Jun;19(2):116-121. 10.3341/kjo.2005.19.2.116.
Protective Effect of Heat Shock Proteins 70.1 and 70.3 on Retinal Photic Injury after Systemic Hyperthermia
- Affiliations
-
- 1Department of Ophthalmology, Seoul National University College of Medicine, Seoul, Korea.
- 2Seoul Artificial Eye Center and Clinical Research Institute, Seoul National University Hospital, Seoul, Korea. ysyu@snu.ac.kr
- 3Research Institute of Pharmaceutical Sciences, College of Pharmacy, Seoul National University, Seoul, Korea.
- KMID: 754408
- DOI: http://doi.org/10.3341/kjo.2005.19.2.116
Abstract
- PURPOSE
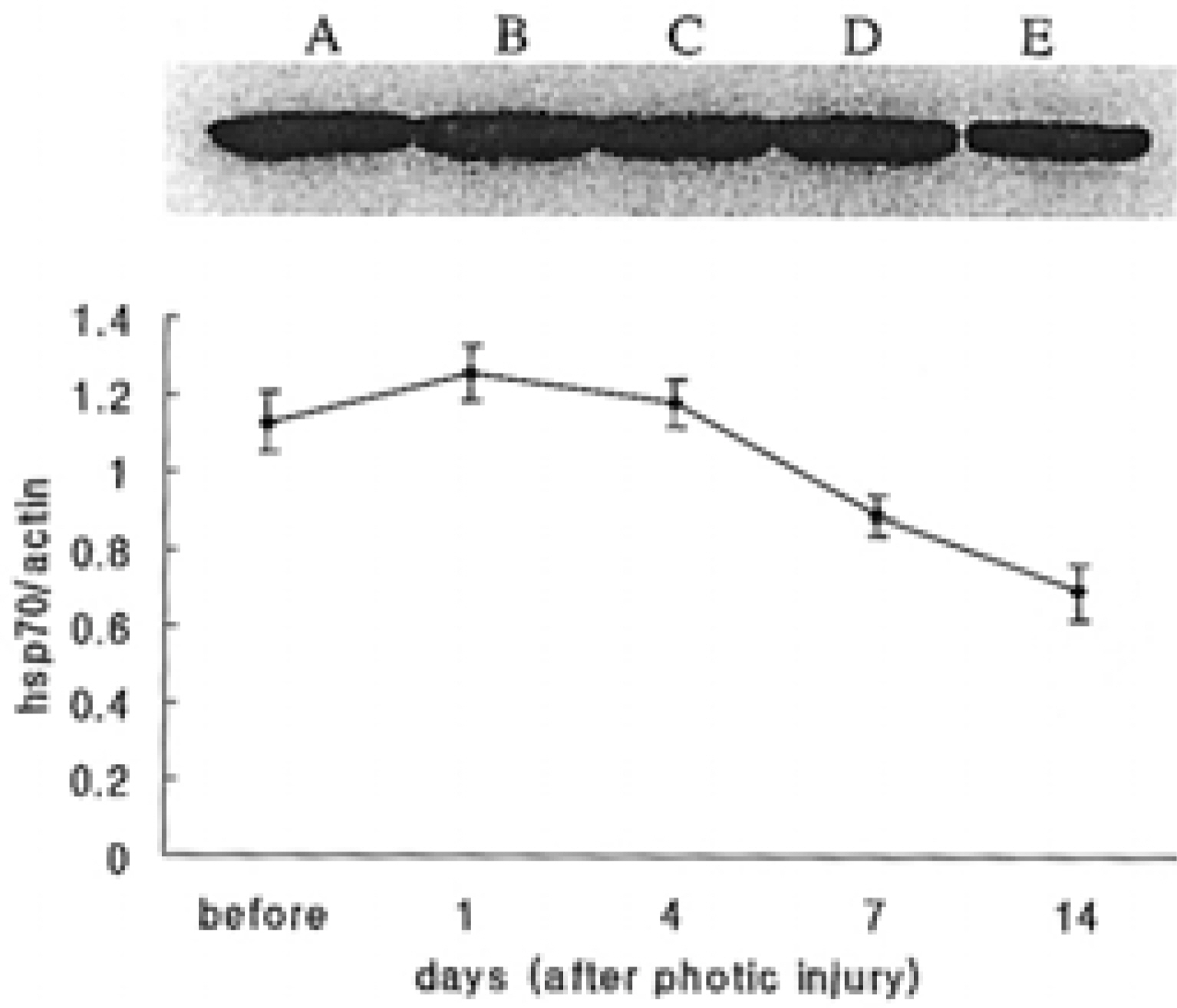
This study aimed to determine the relationship between the heat shock protein 70 from hsps70.1 and 70.3 on retinal photic injury after systemic hyperthermia. METHODS: Eight-week-old female C57BL/6 mice were kept at a constant temperature of 41~42 degrees C for 25~30 minutes. After dark-adaptation for 8 hours, intense light of 11000 lux was maintained for 6 hours. Histology and immunohistochemistry for the inducible heat shock protein 70 (hsp70), the constitutive heat shock protein 70 (hsc70), and western blot analysis, reverse transcriptase-polymerase chain reaction for hsp70.1 and hsp70.3 were performed just before photic injury and after 1, 4, 7, and 14 days. RESULTS: Light-induced retinal degeneration was prevented by thermotolerance. After hyperthermia, hsp70 was densely expressed in the inner segment of the photoreceptor layer on the photic injury. Hsp70 expression increased for 4 days after photic injury and slowly decreased thereafter. mRNA from hsp70.3 was induced earlier than that of hsp70.1. CONCLUSIONS: Retinal photic injury was prevented by hyperthermia-induced hsp70. Hsp70 from hsp70.3 may be a rapid and short-lived responder, and that from hsp70.1 is a slower and more sustained responder. Hsp70 from hsp70.3 may be an initial retinal chaperone while hsp70 from hsp70.1 may be a sustained chaperone.
Keyword
MeSH Terms
Figure
Reference
-
1. Lindquist S, Craig EA. The heat-shock proteins. Annu Rev Genetic. 1988; 22:631–77.
Article2. Hightower LE. Heat shock, stress protein, chaperone, and proteotoxicity. Cell. 1991; 66:191–7.3. Hartl FU. Molecular chaperones in cellular protein folding. Nature. 1996; 381:571–9.
Article4. Welch WJ. Mammalian stress response: cell physiology, structure/function of stress proteins, and implications for medicine and disease. Physiol Rev. 1992; 72:1063–81.
Article5. Morimoto RI, Santoro MG. Stress-inducible responses and heat shock proteins: new pharmacologic targets for cytoprotection. Nat Biotechnol. 1998; 16:833–8.
Article6. Rassow J, Von Ahsen O, Boemer U, Pfanner N. Molecular chaperones: towards a characterization of the heat-shock protein 70 family. Trends Cell Biol. 1997; 7:129–33.7. Beckmann RP, Mizzen LA, Welch WJ. Interaction of HSP 70 with newly synthesized proteins: implications for protein folding and assembly. Science. 1990; 248:850–4.
Article8. Yufu Y, Nishimura J, Ideguchi H, Nawata H. Enhanced synthesis of heat shock proteins and augmented thermotolerance after induction of differentiation in HL-60 human leukemia cells. FEBS Lett. 1990; 268:173–6.
Article9. Lee YJ, Hou Z-Z, Curetty L, Corry PM. Expression, synthesis, and phosphorylation of HSP 28 family during development and decay thermotolerance in CHO plateau-phase cells. J Cell Physiol. 1992; 150:441–6.10. Yokota S, Kitahara M, Nagata K. Benzylidene lactam compound, KNK437, a novel inhibitor of acquisition of thermotolerance and heat shock protein induction in human colon carcinoma cells. Cancer Res. 2000; 60:2942–8.11. Barbe MF, Tytell M, Gower DJ, Welch WJ. Hyperthermia protects against light damage in the rat retina. Science. 1988; 30:1817–20.
Article12. Landry J, Bernier D, Chretien P, et al. Synthesis and degradation of heat shock proteins during development and decay of thermotolerance. Cancer Res. 1982; 42:2457–61.13. Tytell M, Barbe MF, Brown IR. Induction of heat shock (stress) protein 70 and its m RNA in the normal and light-damaged rat retina after whole body hyperthermia. J Neurosci Res. 1994; 38:19–31.14. Dean DO, Kent CR, Tytell M. Constitutive and inducible heat shock protein 70 immunoreactivity in the normal rat eye. Invest Ophthalmol Vis Sci. 1999; 40:2952–62.15. Kim JH, Yu YS, Kim JH, et al. Immunoreactivity of constitutive and inducible heat shock protein 70 in human fetal retina. Korean J Ophthalmol. 2003; 17:14–8.
Article16. Yu YS, Heo JH, Hwang SW, et al. Effect of the absence of heat shock protein 70.1 (hsp70.1) on retinal photoreceptors in normal and rd mice. Korean J Ophthalmol. 2001; 15:67–73.17. Kim JH, Yu YS, Chung H, et al. Effect of the absence of heat shock protein 70.1 (hsp70.1) on retinal photic injury. Korean J Ophthalmol. 2003; 17:7–13.
Article18. Lewden O, Garcher C, Assem M, et al. Changes of the inducible heat shock protein70 mRNA level in rat retina after ischemia and reperfusion. Ophthalmic Res. 1998; 30:291–4.19. Milner CM, Campbell RD. Structure and expression of the three MHC- linked hsp70 genes. Immunogenetics. 1990; 32:242–51.
Article20. Hunt C, Calderwood S. Characterization and sequence of a mouse hsp70 gene and its expression in mouse cell lines. Gene. 1990; 87:199–204.
Article21. Dix DJ, Garges JB, Hong RL. Inhibition of hsp70-1 and hsp70-3 expression disrupts preimplantation embryogenesis and heightens embryo sensitivity to Arsenic. Mol Reprod Dev. 1998; 51:373–80.22. Nakagawa T, Okano H, Furuichi T, et al. Novel subtypes of the mouse inositol 1,4,5-trisphosphate receptor are expressed in tissue- and developmentally specific manner. Proc Natl Acad Sci USA. 1991; 88:6244–8.23. Udono H, Srivastava PK. Heat shock protein 70 associated peptides elicit specific cancer immunity. J Exp Med. 1993; 178:1391–6.24. Tower J. Trasgenic method for increasing Dorsophilia life span. Mech Ageing Dev. 2000; 118:1–14.25. Rea IM, McNerlan S, Pockley AG. Serum heat shock protein and anti-heat shock protein antibody levels in aging. Exp Gerontol. 2001; 36:341–52.
Article26. Minois N, Khazaeli AA, Curtsinger JW. Locomotor activity as a function of age and life span in Drosophilia melanogaster overexpression hsp70. Exp Gerontol. 2001; 36:1137–53.27. Ding XZ, Tsokos GC, Kiang JG. Overexpression of hsp70 inhibits the phosphrylation of HSF1 by activating protein phosphatase and inhibiting protein kinase C activity. FASEB J. 1998; 12:451–9.28. Liossis SNC, Ding XZ, Kiang JG, Tsokos GC. Over-expression of the heat shock protein 70 enhances the TCR/CD3- and Fas/Apo-1/CD95-mediated apoptotic cell death in Jurkat cells. J Immunol. 1997; 158:5668–75.29. Futcher B, Latter GI, Monardo P, et al. A sampling of the yeast proteome. Mol Cell Biol. 1999; 19:7357–68.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of the Absence of Heat Shock Protein 70.1 (hsp70.1) on Retinal Photic Injury
- Expression of Heat Shock Protein 70 and Association with Activity of Superoxide Dismutase in Ischemic Preconditioning Model of Rabbit
- Expression of Heat Shock Protein 70 m-RNA in Rat Bladder Overdistended by Diuresis
- The Significance of Induction of Heat Shock Protein-70 and Glial Fibrillary Acidic Protein Messenger RNA by Delayed Postischemic Hyperthermia Following Transient Focal Ischemia
- The Protective Effect of Induced Heat Shock Protein in Human Corneal Epithelial Cells