Endocrinol Metab.
2024 Oct;39(5):748-757. 10.3803/EnM.2024.2020.
Impact of Chronic Kidney Disease and Gout on End-Stage Renal Disease in Type 2 Diabetes: Population-Based Cohort Study
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Korea University Ansan Hospital, Ansan, Korea
- 2Division of Endocrinology and Metabolism, Department of Internal Medicine, Yeungnam University College of Medicine, Daegu, Korea
- 3Department of Statistics and Actuarial Science, Soongsil University, Seoul, Korea
- KMID: 2560283
- DOI: http://doi.org/10.3803/EnM.2024.2020
Abstract
- Background
We examined the impact of gout on the end-stage renal disease (ESRD) risk in patients with type 2 diabetes mellitus (T2DM) and determined whether this association differs according to chronic kidney disease (CKD) status.
Methods
Using the Korean National Health Insurance Service, this nationwide cohort study enrolled 847,884 patients with T2DM who underwent health checkups in 2009. Based on the presence of CKD (estimated glomerular filtration rate <60 mL/min/1.73 m2) and gout (two outpatient visits or one hospitalization within 5 years), patients were classified into four groups: CKD−Gout−, CKD− Gout+, CKD+Gout−, and CKD+Gout+. Patients with incident ESRD were followed up until December 2018.
Results
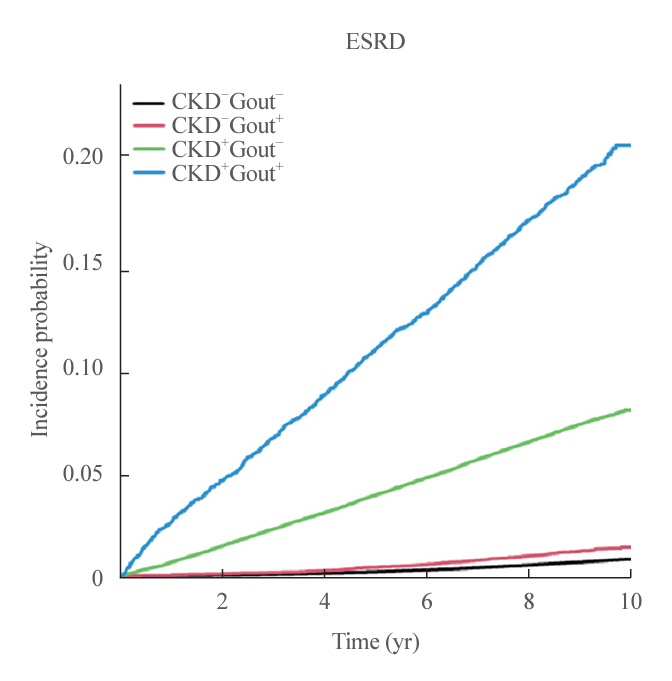
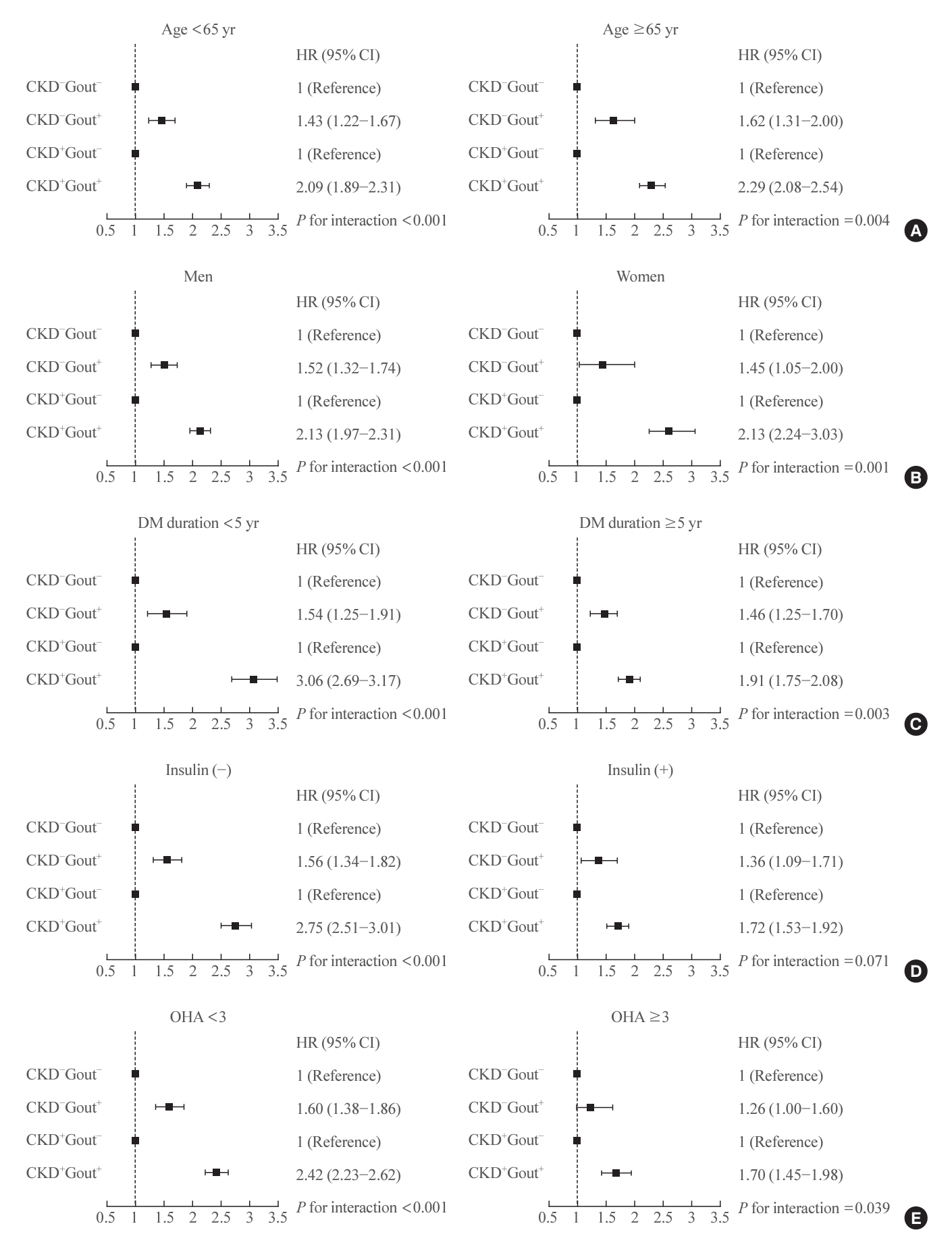
Among 847,884 patients, 11,825 (1.4%) experienced progression to ESRD. ESRD incidence increased in the following order: 0.77 per 1,000 person-years (PY) in the CKD−Gout− group, 1.34/1,000 PY in the CKD−Gout+ group, 8.20/1,000 PY in the CKD+Gout− group, and 23.06/1,000 PY in the CKD+Gout+ group. The presence of gout modified the ESRD risk in a status-dependent manner. Hazard ratios (HR) were 1.49 (95% confidence interval [CI], 1.32 to 1.69) and 2.24 (95% CI, 2.09 to 2.40) in patients without and with CKD, respectively, indicating a significant interaction (P<0.0001). The CKD+Gout+ group had a markedly higher risk of developing ESRD (HR, 18.9; 95% CI, 17.58 to 20.32) than the reference group (CKD−Gout−).
Conclusion
Gout substantially enhances the risk of ESRD, even in the absence of CKD. Concurrent CKD and gout synergistically increase the risk of ESRD. Therefore, physicians should carefully screen for hyperuricemia to prevent progression to ESRD.
Keyword
Figure
Reference
-
1. Jing J, Kielstein JT, Schultheiss UT, Sitter T, Titze SI, Schaeffner ES, et al. Prevalence and correlates of gout in a large cohort of patients with chronic kidney disease: the German Chronic Kidney Disease (GCKD) study. Nephrol Dial Transplant. 2015; 30:613–21.2. Koye DN, Magliano DJ, Nelson RG, Pavkov ME. The global epidemiology of diabetes and kidney disease. Adv Chronic Kidney Dis. 2018; 25:121–32.
Article3. Saran R, Robinson B, Abbott KC, Agodoa LY, Albertus P, Ayanian J, et al. US renal data system 2016 annual data report: epidemiology of kidney disease in the United States. Am J Kidney Dis. 2017; 69(3 Suppl 1):A7–8.4. Jha V, Al-Ghamdi SM, Li G, Wu MS, Stafylas P, Retat L, et al. Global economic burden associated with chronic kidney disease: a pragmatic review of medical costs for the inside CKD research programme. Adv Ther. 2023; 40:4405–20.
Article5. Singh JA, Cleveland JD. Gout is associated with a higher risk of chronic renal disease in older adults: a retrospective cohort study of U.S. Medicare population. BMC Nephrol. 2019; 20:93.
Article6. Yu KH, Kuo CF, Luo SF, See LC, Chou IJ, Chang HC, et al. Risk of end-stage renal disease associated with gout: a nationwide population study. Arthritis Res Ther. 2012; 14:R83.
Article7. Liu Q, Gamble G, Pickering K, Morton S, Dalbeth N. Prevalence and clinical factors associated with gout in patients with diabetes and prediabetes. Rheumatology (Oxford). 2012; 51:757–9.
Article8. Suppiah R, Dissanayake A, Dalbeth N. High prevalence of gout in patients with type 2 diabetes: male sex, renal impairment, and diuretic use are major risk factors. N Z Med J. 2008; 121:43–50.9. Song SO, Jung CH, Song YD, Park CY, Kwon HS, Cha BS, et al. Background and data configuration process of a nationwide population-based study using the Korean National Health Insurance System. Diabetes Metab J. 2014; 38:395–403.
Article10. Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999; 130:461–70.
Article11. Kuo CF, Grainge MJ, Zhang W, Doherty M. Global epidemiology of gout: prevalence, incidence and risk factors. Nat Rev Rheumatol. 2015; 11:649–62.
Article12. Dehlin M, Jacobsson L, Roddy E. Global epidemiology of gout: prevalence, incidence, treatment patterns and risk factors. Nat Rev Rheumatol. 2020; 16:380–90.
Article13. Tsai CW, Lin SY, Kuo CC, Huang CC. Serum uric acid and progression of kidney disease: a longitudinal analysis and mini-review. PLoS One. 2017; 12:e0170393.
Article14. Rincon-Choles H, Jolly SE, Arrigain S, Konig V, Schold JD, Nakhoul G, et al. Impact of uric acid levels on kidney disease progression. Am J Nephrol. 2017; 46:315–22.
Article15. Sellmayr M, Hernandez Petzsche MR, Ma Q, Kruger N, Liapis H, Brink A, et al. Only hyperuricemia with crystalluria, but not asymptomatic hyperuricemia, drives progression of chronic kidney disease. J Am Soc Nephrol. 2020; 31:2773–92.
Article16. Feig DI. Uric acid: a novel mediator and marker of risk in chronic kidney disease? Curr Opin Nephrol Hypertens. 2009; 18:526–30.
Article17. Mazzali M, Hughes J, Kim YG, Jefferson JA, Kang DH, Gordon KL, et al. Elevated uric acid increases blood pressure in the rat by a novel crystal-independent mechanism. Hypertension. 2001; 38:1101–6.
Article18. Kang DH, Nakagawa T, Feng L, Watanabe S, Han L, Mazzali M, et al. A role for uric acid in the progression of renal disease. J Am Soc Nephrol. 2002; 13:2888–97.
Article19. Zhou Y, Fang L, Jiang L, Wen P, Cao H, He W, et al. Uric acid induces renal inflammation via activating tubular NFκB signaling pathway. PLoS One. 2012; 7:e39738.
Article20. Xiao J, Zhang XL, Fu C, Han R, Chen W, Lu Y, et al. Soluble uric acid increases NALP3 inflammasome and interleukin1β expression in human primary renal proximal tubule epithelial cells through the Toll-like receptor 4-mediated pathway. Int J Mol Med. 2015; 35:1347–54.
Article21. Krishnan E, Pandya BJ, Chung L, Hariri A, Dabbous O. Hyperuricemia in young adults and risk of insulin resistance, prediabetes, and diabetes: a 15-year follow-up study. Am J Epidemiol. 2012; 176:108–16.
Article22. Li C, Hsieh MC, Chang SJ. Metabolic syndrome, diabetes, and hyperuricemia. Curr Opin Rheumatol. 2013; 25:210–6.
Article23. Hayden MR, Tyagi SC. Uric acid: a new look at an old risk marker for cardiovascular disease, metabolic syndrome, and type 2 diabetes mellitus: the urate redox shuttle. Nutr Metab (Lond). 2004; 1:10.
Article24. Perticone M, Maio R, Shehaj E, Gigliotti S, Caroleo B, Suraci E, et al. Sex-related differences for uric acid in the prediction of cardiovascular events in essential hypertension: a population prospective study. Cardiovasc Diabetol. 2023; 22:298.
Article25. Maloberti A, Maggioni S, Occhi L, Triglione N, Panzeri F, Nava S, et al. Sex-related relationships between uric acid and target organ damage in hypertension. J Clin Hypertens (Greenwich). 2018; 20:193–200.
Article26. Kolz M, Johnson T, Sanna S, Teumer A, Vitart V, Perola M, et al. Meta-analysis of 28,141 individuals identifies common variants within five new loci that influence uric acid concentrations. PLoS Genet. 2009; 5:e1000504.
Article27. Kim JY, Kim SJ, Nam CM, Moon KT, Park EC. Changes in prescription pattern, pharmaceutical expenditure and quality of care after introduction of reimbursement restriction in diabetes in Korea. Eur J Public Health. 2018; 28:209–14.
Article28. Baek JH, Yang YS, Ko SH, Han KD, Kim JH, Moon MK, et al. Real-world prescription patterns and barriers related to the use of sodium-glucose cotransporter 2 inhibitors among Korean patients with type 2 diabetes mellitus and cardiovascular disease. Diabetes Metab J. 2022; 46:701–12.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Familial Juvenile Hyperuricemic Nephropathy and Uromodulin Gene Mutation
- Transplantation Options in Type 1 Diabetes with End Stage Renal Disease
- Therapeutic Consideration for the Elderly Patients with End Stage Renal Disease
- Burden of End-Stage Kidney Disease by Type 2 Diabetes Mellitus Status in South Korea: A Nationwide Epidemiologic Study
- Diagnosis and test for diabetic kidney disease