J Stroke.
2024 May;26(2):131-163. 10.5853/jos.2023.02719.
Leukoaraiosis: Epidemiology, Imaging, Risk Factors, and Management of Age-Related Cerebral White Matter Hyperintensities
- Affiliations
-
- 1Department of Central Laboratory, Shanghai Children’s Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai, China
- 2Department of Neurology, The First Affiliated Hospital of Xiamen University, School of Medicine, Xiamen University, Xiamen, Fujian, China
- 3Xiamen Clinical Research Center for Neurological Diseases, The First Affiliated Hospital of Xiamen University, School of Medicine, Xiamen University, Xiamen, Fujian, China
- 4Fujian Provincial Clinical Research Center for Brain Diseases, The First Affiliated Hospital of Xiamen University, School of Medicine, Xiamen University, Xiamen, Fujian, China
- 5The Third Clinical College, Fujian Medical University, Fuzhou, Fujian, China
- 6Translational Medicine Research Center, School of Pharmaceutical Sciences, Xiamen University, Xiamen, Fujian, China
- KMID: 2556038
- DOI: http://doi.org/10.5853/jos.2023.02719
Abstract
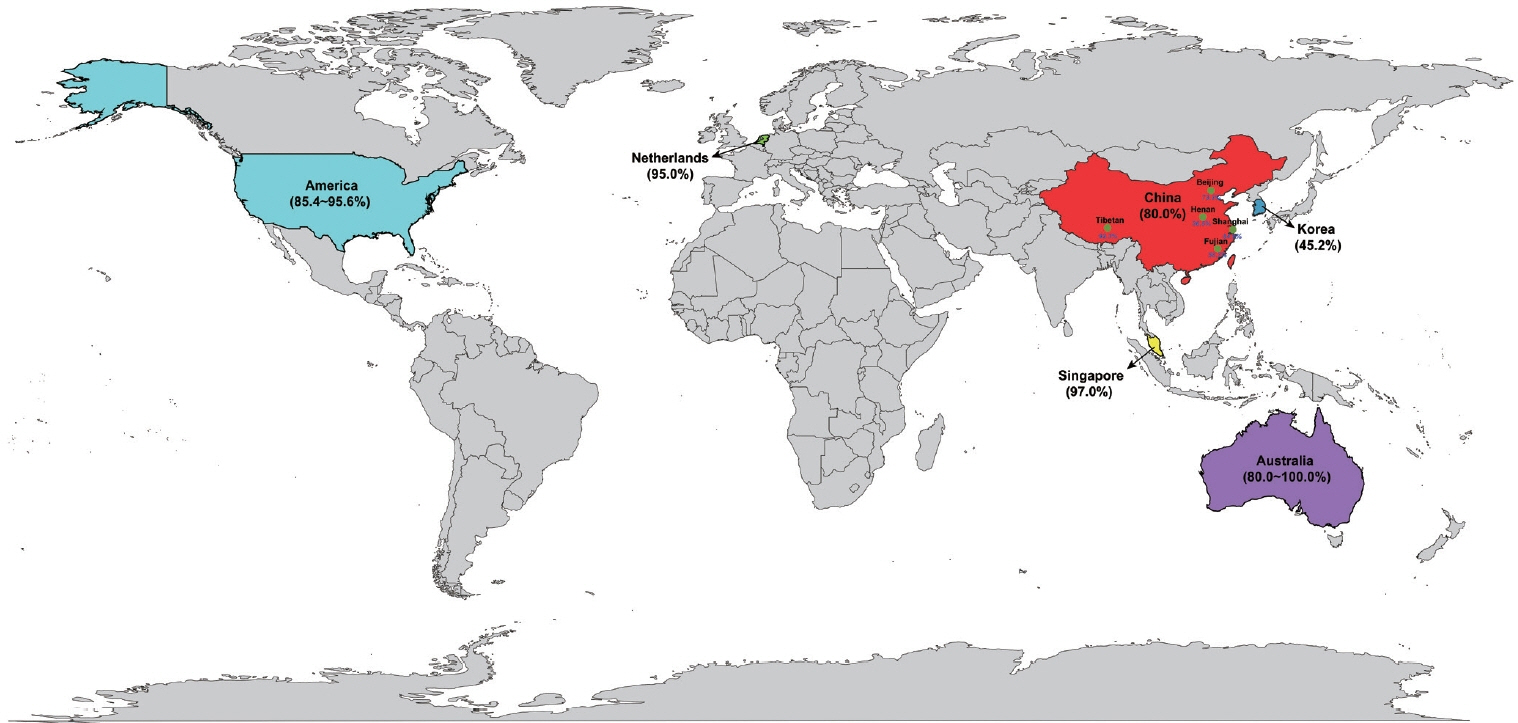
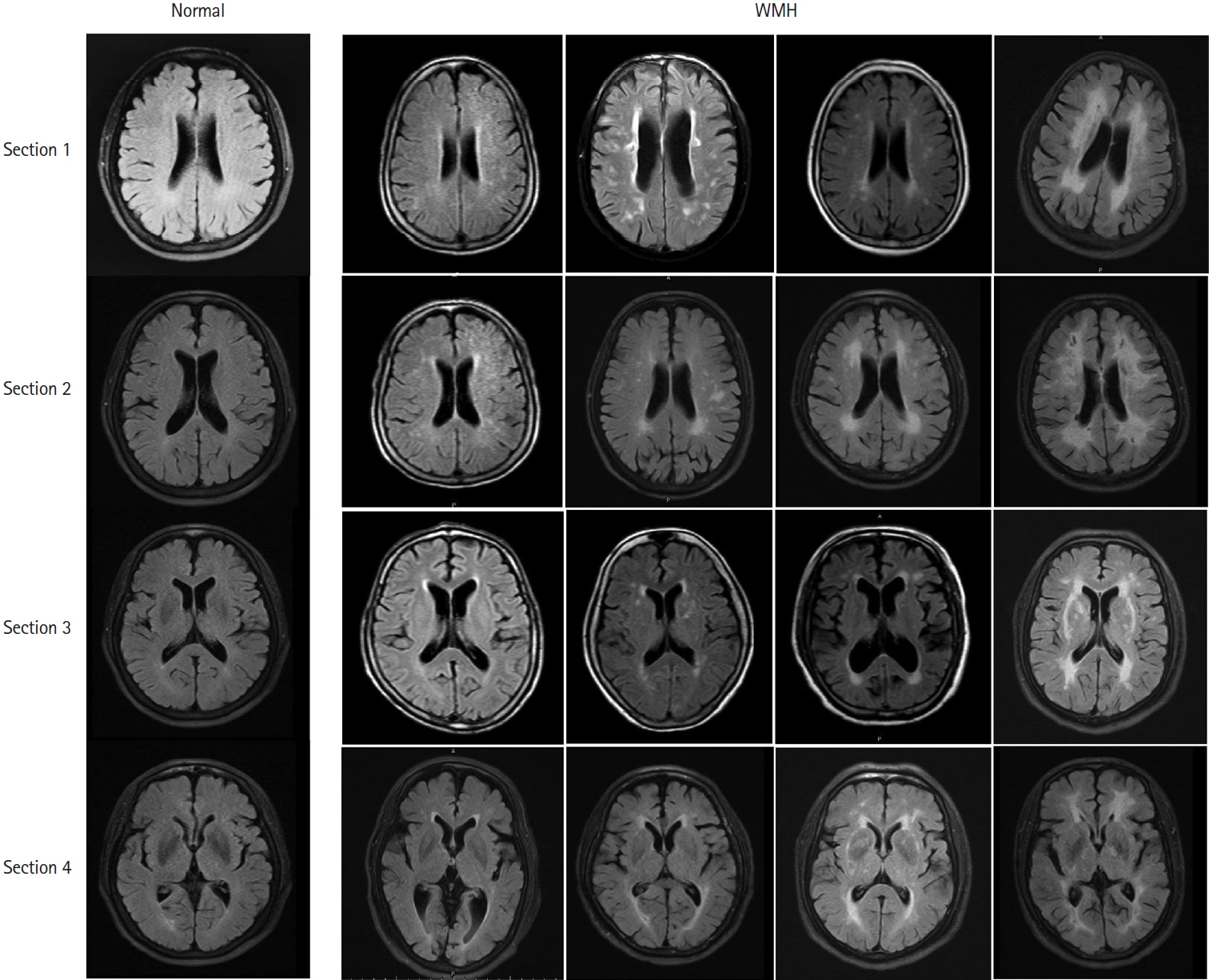
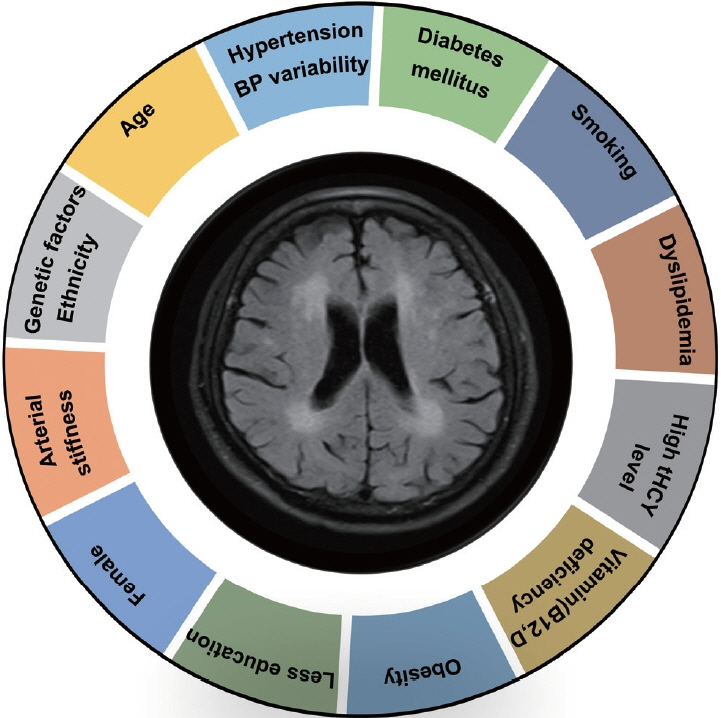
- Leukoaraiosis (LA) manifests as cerebral white matter hyperintensities on T2-weighted magnetic resonance imaging scans and corresponds to white matter lesions or abnormalities in brain tissue. Clinically, it is generally detected in the early 40s and is highly prevalent globally in individuals aged >60 years. From the imaging perspective, LA can present as several heterogeneous forms, including punctate and patchy lesions in deep or subcortical white matter; lesions with periventricular caps, a pencil-thin lining, and smooth halo; as well as irregular lesions, which are not always benign. Given its potential of having deleterious effects on normal brain function and the resulting increase in public health burden, considerable effort has been focused on investigating the associations between various risk factors and LA risk, and developing its associated clinical interventions. However, study results have been inconsistent, most likely due to potential differences in study designs, neuroimaging methods, and sample sizes as well as the inherent neuroimaging heterogeneity and multi-factorial nature of LA. In this article, we provided an overview of LA and summarized the current knowledge regarding its epidemiology, neuroimaging classification, pathological characteristics, risk factors, and potential intervention strategies.
Keyword
Figure
Reference
-
References
1. Hachinski VC, Potter P, Merskey H. Leuko-araiosis. Arch Neurol. 1987; 44:21–23.2. O’Sullivan M. Leukoaraiosis. Pract Neurol. 2008; 8:26–38.
Article3. Hachinski VC, Potter P, Merskey H. Leuko-araiosis: an ancient term for a new problem. Can J Neurol Sci. 1986; 13(4 Suppl):533–534.4. Longstreth WT Jr, Manolio TA, Arnold A, Burke GL, Bryan N, Jungreis CA, et al. Clinical correlates of white matter findings on cranial magnetic resonance imaging of 3301 elderly people. The cardiovascular health study. Stroke. 1996; 27:1274–1282.
Article5. Lin Q, Huang WQ, Ma QL, Lu CX, Tong SJ, Ye JH, et al. Incidence and risk factors of leukoaraiosis from 4683 hospitalized patients: a cross-sectional study. Medicine (Baltimore). 2017; 96:e7682.6. Wen W, Sachdev PS, Li JJ, Chen X, Anstey KJ. White matter hyperintensities in the forties: their prevalence and topography in an epidemiological sample aged 44-48. Hum Brain Mapp. 2009; 30:1155–1167.
Article7. Ylikoski A, Erkinjuntti T, Raininko R, Sarna S, Sulkava R, Tilvis R. White matter hyperintensities on MRI in the neurologically nondiseased elderly. Analysis of cohorts of consecutive subjects aged 55 to 85 years living at home. Stroke. 1995; 26:1171–1177.
Article8. Dufouil C, Godin O, Chalmers J, Coskun O, MacMahon S, Tzourio-Mazoyer N, et al. Severe cerebral white matter hyperintensities predict severe cognitive decline in patients with cerebrovascular disease history. Stroke. 2009; 40:2219–2221.
Article9. van den Heuvel DM, ten Dam VH, de Craen AJ, AdmiraalBehloul F, Olofsen H, Bollen EL, et al. Increase in periventricular white matter hyperintensities parallels decline in mental processing speed in a non-demented elderly population. J Neurol Neurosurg Psychiatry. 2006; 77:149–153.10. Zheng JJ, Delbaere K, Close JC, Sachdev PS, Lord SR. Impact of white matter lesions on physical functioning and fall risk in older people: a systematic review. Stroke. 2011; 42:2086–2090.
Article11. Moon SY, de Souto Barreto P, Rolland Y, Chupin M, Bouyahia A, Fillon L, et al. Prospective associations between white matter hyperintensities and lower extremity function. Neurology. 2018; 90:e1291–e1297.
Article12. Lee Y, Ko J, Choi YE, Oh JS, Kim JS, Sunwoo MK, et al. Areas of white matter hyperintensities and motor symptoms of Parkinson disease. Neurology. 2020; 95:e291–e298.
Article13. Clancy U, Gilmartin D, Jochems ACC, Knox L, Doubal FN, Wardlaw JM. Neuropsychiatric symptoms associated with cerebral small vessel disease: a systematic review and metaanalysis. Lancet Psychiatry. 2021; 8:225–236.
Article14. Tosto G, Zimmerman ME, Carmichael OT, Brickman AM; Alzheimer’s Disease Neuroimaging Initiative. Predicting aggressive decline in mild cognitive impairment: the importance of white matter hyperintensities. JAMA Neurol. 2014; 71:872–877.15. Kloppenborg RP, Nederkoorn PJ, Geerlings MI, van den Berg E. Presence and progression of white matter hyperintensities and cognition: a meta-analysis. Neurology. 2014; 82:2127–2138.16. Prins ND, Scheltens P. White matter hyperintensities, cognitive impairment and dementia: an update. Nat Rev Neurol. 2015; 11:157–165.17. Hu HY, Ou YN, Shen XN, Qu Y, Ma YH, Wang ZT, et al. White matter hyperintensities and risks of cognitive impairment and dementia: a systematic review and meta-analysis of 36 prospective studies. Neurosci Biobehav Rev. 2021; 120:16–27.18. Whitman GT, Tang Y, Lin A, Baloh RW. A prospective study of cerebral white matter abnormalities in older people with gait dysfunction. Neurology. 2001; 57:990–994.
Article19. Srikanth V, Beare R, Blizzard L, Phan T, Stapleton J, Chen J, et al. Cerebral white matter lesions, gait, and the risk of incident falls: a prospective population-based study. Stroke. 2009; 40:175–180.20. Su C, Yang X, Wei S, Zhao R. Association of cerebral small vessel disease with gait and balance disorders. Front Aging Neurosci. 2022; 14:834496.
Article21. Debette S, Markus HS. The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ. 2010; 341:c3666.
Article22. Ryu WS, Woo SH, Schellingerhout D, Jang MU, Park KJ, Hong KS, et al. Stroke outcomes are worse with larger leukoaraiosis volumes. Brain. 2017; 140:158–170.
Article23. Georgakis MK, Duering M, Wardlaw JM, Dichgans M. WMH and long-term outcomes in ischemic stroke: a systematic review and meta-analysis. Neurology. 2019; 92:e1298–e1308.24. Imaizumi T, Inamura S, Nomura T. The severities of white matter lesions possibly influence the recurrences of several stroke types. J Stroke Cerebrovasc Dis. 2014; 23:1897–1902.25. Rensma SP, van Sloten TT, Launer LJ, Stehouwer CDA. Cerebral small vessel disease and risk of incident stroke, dementia and depression, and all-cause mortality: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2018; 90:164–173.
Article26. Ghaznawi R, Geerlings MI, Jaarsma-Coes M, Hendrikse J, de Bresser J; UCC-Smart Study Group. Association of white matter hyperintensity markers on MRI and long-term risk of mortality and ischemic stroke: the SMART-MR study. Neurology. 2021; 96:e2172–e2183.27. Godin O, Dufouil C, Maillard P, Delcroix N, Mazoyer B, Crivello F, et al. White matter lesions as a predictor of depression in the elderly: the 3C-Dijon study. Biol Psychiatry. 2008; 63:663–669.
Article28. Herrmann LL, Le Masurier M, Ebmeier KP. White matter hyperintensities in late life depression: a systematic review. J Neurol Neurosurg Psychiatry. 2008; 79:619–624.
Article29. Wang L, Leonards CO, Sterzer P, Ebinger M. White matter lesions and depression: a systematic review and meta-analysis. J Psychiatr Res. 2014; 56:56–64.30. Fang Y, Qin T, Liu W, Ran L, Yang Y, Huang H, et al. Cerebral small-vessel disease and risk of incidence of depression: a meta-analysis of longitudinal cohort studies. J Am Heart Assoc. 2020; 9:e016512.
Article31. Launer LJ, Berger K, Breteler MM, Dufouil C, Fuhrer R, Giampaoli S, et al. Regional variability in the prevalence of cerebral white matter lesions: an MRI study in 9 European countries (CASCADE). Neuroepidemiology. 2006; 26:23–29.
Article32. Mok V, Srikanth V, Xiong Y, Phan TG, Moran C, Chu S, et al. Race-ethnicity and cerebral small vessel disease--comparison between Chinese and White populations. Int J Stroke. 2014; 9(Suppl A100):36–42.
Article33. de Leeuw FE, de Groot JC, Achten E, Oudkerk M, Ramos LM, Heijboer R, et al. Prevalence of cerebral white matter lesions in elderly people: a population based magnetic resonance imaging study. The Rotterdam scan study. J Neurol Neurosurg Psychiatry. 2001; 70:9–14.
Article34. Wen W, Sachdev P. The topography of white matter hyperintensities on brain MRI in healthy 60- to 64-year-old individuals. Neuroimage. 2004; 22:144–154.35. Liao D, Cooper L, Cai J, Toole J, Bryan N, Burke G, et al. The prevalence and severity of white matter lesions, their relationship with age, ethnicity, gender, and cardiovascular disease risk factors: the ARIC study. Neuroepidemiology. 1997; 16:149–162.
Article36. Lam BYK, Yiu B, Ampil E, Chen CL, Dikot Y, Dominguez JC, et al. High burden of cerebral white matter lesion in 9 Asian cities. Sci Rep. 2021; 11:11587.
Article37. Zhang S, Kang X. Investigation of the risk factors for leukoaraiosis (LA). Asia Pac J Public Health. 2013; 25(4 Suppl):64S71S.
Article38. Han F, Zhai FF, Wang Q, Zhou LX, Ni J, Yao M, et al. Prevalence and risk factors of cerebral small vessel disease in a Chinese population-based sample. J Stroke. 2018; 20:239–246.
Article39. Jin H, Ding Z, Lian S, Zhao Y, He S, Zhou L, et al. Prevalence and risk factors of white matter lesions in Tibetan patients without acute stroke. Stroke. 2020; 51:149–153.
Article40. Verny M, Duyckaerts C, Pierot L, Hauw JJ. Leuko-araiosis. Dev Neurosci. 1991; 13:245–250.41. Wahlund LO, Barkhof F, Fazekas F, Bronge L, Augustin M, Sjögren M, et al. A new rating scale for age-related white matter changes applicable to MRI and CT. Stroke. 2001; 32:1318–1322.
Article42. Xiong YY, Mok V. Age-related white matter changes. J Aging Res. 2011; 2011:617927.
Article43. Fazekas F, Barkhof F, Wahlund LO, Pantoni L, Erkinjuntti T, Scheltens P, et al. CT and MRI rating of white matter lesions. Cerebrovasc Dis. 2002; 13(Suppl 2):31–36.
Article44. Kates R, Atkinson D, Brant-Zawadzki M. Fluid-attenuated inversion recovery (FLAIR): clinical prospectus of current and future applications. Top Magn Reson Imaging. 1996; 8:389–396.45. Caligiuri ME, Perrotta P, Augimeri A, Rocca F, Quattrone A, Cherubini A. Automatic detection of white matter hyperintensities in healthy aging and pathology using magnetic resonance imaging: a review. Neuroinformatics. 2015; 13:261–276.
Article46. Tomura N, Kato K, Takahashi S, Sashi R, Sakuma I, Narita K, et al. Comparison of multishot echo-planar fluid-attenuated inversion-recovery imaging with fast spin-echo fluid-attenuated inversion-recovery and T2-weighted imaging in depiction of white matter lesions. J Comput Assist Tomogr. 2002; 26:810–814.
Article47. Piguet O, Ridley LJ, Grayson DA, Bennett HP, Creasey H, Lye TC, et al. Comparing white matter lesions on T2 and FLAIR MRI in the Sydney older persons study. Eur J Neurol. 2005; 12:399–402.
Article48. Barkhof F, Scheltens P. Imaging of white matter lesions. Cerebrovasc Dis. 2002; 13(Suppl 2):21–30.
Article49. Grueter BE, Schulz UG. Age-related cerebral white matter disease (leukoaraiosis): a review. Postgrad Med J. 2012; 88:79–87.
Article50. Taylor WD, Hsu E, Krishnan KR, MacFall JR. Diffusion tensor imaging: background, potential, and utility in psychiatric research. Biol Psychiatry. 2004; 55:201–207.
Article51. Assaf Y, Pasternak O. Diffusion tensor imaging (DTI)-based white matter mapping in brain research: a review. J Mol Neurosci. 2008; 34:51–61.
Article52. Jones DK, Lythgoe D, Horsfield MA, Simmons A, Williams SC, Markus HS. Characterization of white matter damage in ischemic leukoaraiosis with diffusion tensor MRI. Stroke. 1999; 30:393–397.
Article53. Haacke EM, Ayaz M, Khan A, Manova ES, Krishnamurthy B, Gollapalli L, et al. Establishing a baseline phase behavior in magnetic resonance imaging to determine normal vs. abnormal iron content in the brain. J Magn Reson Imaging. 2007; 26:256–264.54. Kraft E, Trenkwalder C, Auer DP. T2*-weighted MRI differentiates multiple system atrophy from Parkinson’s disease. Neurology. 2002; 59:1265–1267.
Article55. Yates PA, Villemagne VL, Ellis KA, Desmond PM, Masters CL, Rowe CC. Cerebral microbleeds: a review of clinical, genetic, and neuroimaging associations. Front Neurol. 2014; 4:205.
Article56. Huang WQ, Lin HN, Lin Q, Tzeng CM. Susceptibility weighted imaging (SWI) recommended as a regular magnetic resonance diagnosis for vascular dementia to identify independent idiopathic normal pressure hydrocephalus before ventriculo-peritoneal (V-P) shunt treatment: a case study. Front Neurol. 2019; 10:262.
Article57. Fazekas F, Chawluk JB, Alavi A, Hurtig HI, Zimmerman RA. MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR Am J Roentgenol. 1987; 149:351–356.
Article58. Schmidt R, Schmidt H, Haybaeck J, Loitfelder M, Weis S, Cavalieri M, et al. Heterogeneity in age-related white matter changes. Acta Neuropathol. 2011; 122:171–185.
Article59. Kim KW, MacFall JR, Payne ME. Classification of white matter lesions on magnetic resonance imaging in elderly persons. Biol Psychiatry. 2008; 64:273–280.
Article60. Payne ME, Fetzer DL, MacFall JR, Provenzale JM, Byrum CE, Krishnan KR. Development of a semi-automated method for quantification of MRI gray and white matter lesions in geriatric subjects. Psychiatry Res. 2002; 115:63–77.
Article61. Scheltens P, Erkinjunti T, Leys D, Wahlund LO, Inzitari D, del Ser T, et al. White matter changes on CT and MRI: an overview of visual rating scales. Eur Neurol. 1998; 39:80–89.
Article62. Mäntylä R, Erkinjuntti T, Salonen O, Aronen HJ, Peltonen T, Pohjasvaara T, et al. Variable agreement between visual rating scales for white matter hyperintensities on MRI. Comparison of 13 rating scales in a poststroke cohort. Stroke. 1997; 28:1614–1623.
Article63. Kapeller P, Barber R, Vermeulen RJ, Adèr H, Scheltens P, Freidl W, et al. Visual rating of age-related white matter changes on magnetic resonance imaging: scale comparison, interrater agreement, and correlations with quantitative measurements. Stroke. 2003; 34:441–445.64. Admiraal-Behloul F, van den Heuvel DM, Olofsen H, van Osch MJ, van der Grond J, van Buchem MA, et al. Fully automatic segmentation of white matter hyperintensities in MR images of the elderly. Neuroimage. 2005; 28:607–617.
Article65. Scheltens P, Barkhof F, Leys D, Pruvo JP, Nauta JJ, Vermersch P, et al. A semiquantative rating scale for the assessment of signal hyperintensities on magnetic resonance imaging. J Neurol Sci. 1993; 114:7–12.
Article66. Andere A, Jindal G, Molino J, Collins S, Merck D, Burton T, et al. Volumetric white matter hyperintensity ranges correspond to Fazekas scores on brain MRI. J Stroke Cerebrovasc Dis. 2022; 31:106333.
Article67. Lin Q, Huang WQ, Tzeng CM. Genetic associations of leukoaraiosis indicate pathophysiological mechanisms in white matter lesions etiology. Rev Neurosci. 2015; 26:343–358.
Article68. Huang WQ, Yi KH, Li Z, Wang H, Li ML, Cai LL, et al. DNA methylation profiling reveals the change of inflammation-associated ZC3H12D in leukoaraiosis. Front Aging Neurosci. 2018; 10:143.
Article69. Griffanti L, Jenkinson M, Suri S, Zsoldos E, Mahmood A, Filippini N, et al. Classification and characterization of periventricular and deep white matter hyperintensities on MRI: a study in older adults. Neuroimage. 2018; 170:174–181.
Article70. de Groot JC, de Leeuw FE, Oudkerk M, Hofman A, Jolles J, Breteler MM. Cerebral white matter lesions and depressive symptoms in elderly adults. Arch Gen Psychiatry. 2000; 57:1071–1076.
Article71. Krishnan MS, O’Brien JT, Firbank MJ, Pantoni L, Carlucci G, Erkinjuntti T, et al. Relationship between periventricular and deep white matter lesions and depressive symptoms in older people. The LADIS study. Int J Geriatr Psychiatry. 2006; 21:983–989.
Article72. Fazekas F, Kleinert R, Offenbacher H, Schmidt R, Kleinert G, Payer F, et al. Pathologic correlates of incidental MRI white matter signal hyperintensities. Neurology. 1993; 43:1683–1689.
Article73. Prins ND, van Dijk EJ, den Heijer T, Vermeer SE, Koudstaal PJ, Oudkerk M, et al. Cerebral white matter lesions and the risk of dementia. Arch Neurol. 2004; 61:1531–1534.74. de Groot JC, de Leeuw FE, Oudkerk M, van Gijn J, Hofman A, Jolles J, et al. Cerebral white matter lesions and cognitive function: the Rotterdam scan study. Ann Neurol. 2000; 47:145–151.
Article75. De Groot JC, De Leeuw FE, Oudkerk M, Van Gijn J, Hofman A, Jolles J, et al. Periventricular cerebral white matter lesions predict rate of cognitive decline. Ann Neurol. 2002; 52:335–341.
Article76. Sze G, De Armond SJ, Brant-Zawadzki M, Davis RL, Norman D, Newton TH. Foci of MRI signal (pseudo lesions) anterior to the frontal horns: histologic correlations of a normal finding. AJR Am J Roentgenol. 1986; 147:331–337.
Article77. Fazekas F, Schmidt R, Scheltens P. Pathophysiologic mechanisms in the development of age-related white matter changes of the brain. Dement Geriatr Cogn Disord. 1998; 9(Suppl 1):2–5.
Article78. Thomas AJ, O’Brien JT, Barber R, McMeekin W, Perry R. A neuropathological study of periventricular white matter hyperintensities in major depression. J Affect Disord. 2003; 76:49–54.79. van Swieten JC, van den Hout JH, van Ketel BA, Hijdra A, Wokke JH, van Gijn J. Periventricular lesions in the white matter on magnetic resonance imaging in the elderly: a morphometric correlation with arteriolosclerosis and dilated perivascular spaces. Brain. 1991; 114(Pt 2):761–774.
Article80. Wu X, Ya J, Zhou D, Ding Y, Ji X, Meng R. Pathogeneses and imaging features of cerebral white matter lesions of vascular origins. Aging Dis. 2021; 12:2031–2051.
Article81. Mayer PL, Kier EL. The controversy of the periventricular white matter circulation: a review of the anatomic literature. AJNR Am J Neuroradiol. 1991; 12:223–228.82. Moody DM, Bell MA, Challa VR. Features of the cerebral vascular pattern that predict vulnerability to perfusion or oxygenation deficiency: an anatomic study. AJNR Am J Neuroradiol. 1990; 11:431–439.83. Beyer JL, Young R, Kuchibhatla M, Krishnan KR. Hyperintense MRI lesions in bipolar disorder: a meta-analysis and review. Int Rev Psychiatry. 2009; 21:394–409.
Article84. ten Dam VH, van den Heuvel DM, de Craen AJ, Bollen EL, Murray HM, Westendorp RG, et al. Decline in total cerebral blood flow is linked with increase in periventricular but not deep white matter hyperintensities. Radiology. 2007; 243:198–203.
Article85. Stewart PA, Magliocco M, Hayakawa K, Farrell CL, Del Maestro RF, Girvin J, et al. A quantitative analysis of blood-brain barrier ultrastructure in the aging human. Microvasc Res. 1987; 33:270–282.
Article86. Pantoni L, Garcia JH. Pathogenesis of leukoaraiosis: a review. Stroke. 1997; 28:652–659.87. Pantoni L. Pathophysiology of age-related cerebral white matter changes. Cerebrovasc Dis. 2002; 13(Suppl 2):7–10.
Article88. Jung KH, Stephens KA, Yochim KM, Riphagen JM, Kim CM, Buckner RL, et al. Heterogeneity of cerebral white matter lesions and clinical correlates in older adults. Stroke. 2021; 52:620–630.89. Schmidt R, Fazekas F, Kapeller P, Schmidt H, Hartung HP. MRI white matter hyperintensities: three-year follow-up of the Austrian stroke prevention study. Neurology. 1999; 53:132–139.90. Schmidt R, Enzinger C, Ropele S, Schmidt H, Fazekas F; Austrian Stroke Prevention Study. Progression of cerebral white matter lesions: 6-year results of the Austrian stroke prevention study. Lancet. 2003; 361:2046–2048.
Article91. Longstreth WT Jr, Arnold AM, Beauchamp NJ Jr, Manolio TA, Lefkowitz D, Jungreis C, et al. Incidence, manifestations, and predictors of worsening white matter on serial cranial magnetic resonance imaging in the elderly: the cardiovascular health study. Stroke. 2005; 36:56–61.
Article92. Taylor WD, MacFall JR, Provenzale JM, Payne ME, McQuoid DR, Steffens DC, et al. Serial MR imaging of volumes of hyperintense white matter lesions in elderly patients: correlation with vascular risk factors. AJR Am J Roentgenol. 2003; 181:571–576.
Article93. Masana Y, Motozaki T. Emergence and progress of white matter lesion in brain check-up. Acta Neurol Scand. 2003; 107:187–194.94. van Dijk EJ, Prins ND, Vrooman HA, Hofman A, Koudstaal PJ, Breteler MM. Progression of cerebral small vessel disease in relation to risk factors and cognitive consequences: Rotterdam scan study. Stroke. 2008; 39:2712–2719.
Article95. Sachdev P, Wen W, Chen X, Brodaty H. Progression of white matter hyperintensities in elderly individuals over 3 years. Neurology. 2007; 68:214–222.
Article96. Gyanwali B, Shaik MA, Tan BY, Venketasubramanian N, Chen C, Hilal S. Risk factors for and clinical relevance of incident and progression of cerebral small vessel disease markers in an Asian memory clinic population. J Alzheimers Dis. 2019; 67:1209–1219.
Article97. Sachdev P, Chen X, Wen W. White matter hyperintensities in mid-adult life. Curr Opin Psychiatry. 2008; 21:268–274.
Article98. van den Heuvel DM, Admiraal-Behloul F, ten Dam VH, Olofsen H, Bollen EL, Murray HM, et al. Different progression rates for deep white matter hyperintensities in elderly men and women. Neurology. 2004; 63:1699–1701.
Article99. Liao D, Cooper L, Cai J, Toole JF, Bryan NR, Hutchinson RG, et al. Presence and severity of cerebral white matter lesions and hypertension, its treatment, and its control: the ARIC study. Stroke. 1996; 27:2262–2270.
Article100. de Leeuw FE, de Groot JC, Oudkerk M, Witteman JC, Hofman A, van Gijn J, et al. A follow-up study of blood pressure and cerebral white matter lesions. Ann Neurol. 1999; 46:827–833.
Article101. de Leeuw FE, de Groot JC, Oudkerk M, Witteman JC, Hofman A, van Gijn J, et al. Hypertension and cerebral white matter lesions in a prospective cohort study. Brain. 2002; 125(Pt 4):765–772.
Article102. van Dijk EJ, Breteler MM, Schmidt R, Berger K, Nilsson LG, Oudkerk M, et al. The association between blood pressure, hypertension, and cerebral white matter lesions: cardiovascular determinants of dementia study. Hypertension. 2004; 44:625–630.
Article103. Zhao Y, Ke Z, He W, Cai Z. Volume of white matter hyperintensities increases with blood pressure in patients with hypertension. J Int Med Res. 2019; 47:3681–3689.104. Basile AM, Pantoni L, Pracucci G, Asplund K, Chabriat H, Erkinjuntti T, et al. Age, hypertension, and lacunar stroke are the major determinants of the severity of age-related white matter changes: the LADIS (leukoaraiosis and disability in the elderly) study. Cerebrovasc Dis. 2006; 21:315–322.
Article105. Vuorinen M, Solomon A, Rovio S, Nieminen L, Kåreholt I, Tuomilehto J, et al. Changes in vascular risk factors from midlife to late life and white matter lesions: a 20-year follow-up study. Dement Geriatr Cogn Disord. 2011; 31:119–125.
Article106. Habes M, Erus G, Toledo JB, Zhang T, Bryan N, Launer LJ, et al. White matter hyperintensities and imaging patterns of brain ageing in the general population. Brain. 2016; 139(Pt 4):1164–1179.
Article107. Guo X, Pantoni L, Simoni M, Bengtsson C, Björkelund C, Lissner L, et al. Blood pressure components and changes in relation to white matter lesions: a 32-year prospective population study. Hypertension. 2009; 54:57–62.
Article108. Lane CA, Barnes J, Nicholas JM, Sudre CH, Cash DM, Parker TD, et al. Associations between blood pressure across adulthood and late-life brain structure and pathology in the neuroscience substudy of the 1946 British birth cohort (Insight 46): an epidemiological study. Lancet Neurol. 2019; 18:942–952.109. Wartolowska KA, Webb AJS. Midlife blood pressure is associated with the severity of white matter hyperintensities: analysis of the UK Biobank cohort study. Eur Heart J. 2021; 42:750–757.
Article110. Sargurupremraj M, Suzuki H, Jian X, Sarnowski C, Evans TE, Bis JC, et al. Cerebral small vessel disease genomics and its implications across the lifespan. Nat Commun. 2020; 11:6285.111. Wilkinson I, Webb AJS. Consistency of associations of systolic and diastolic blood pressure with white matter hyperintensities: a meta-analysis. Int J Stroke. 2022; 17:291–298.
Article112. Marcus J, Gardener H, Rundek T, Elkind MS, Sacco RL, Decarli C, et al. Baseline and longitudinal increases in diastolic blood pressure are associated with greater white matter hyperintensity volume: the northern Manhattan study. Stroke. 2011; 42:2639–2641.
Article113. Shokouhi M, Qiu D, Samman Tahhan A, Quyyumi AA, Hajjar I. Differential associations of diastolic and systolic pressures with cerebral measures in older individuals with mild cognitive impairment. Am J Hypertens. 2018; 31:1268–1277.
Article114. Sudre CH, Smith L, Atkinson D, Chaturvedi N, Ourselin S, Barkhof F, et al. Cardiovascular risk factors and white matter hyperintensities: difference in susceptibility in South Asians compared with Europeans. J Am Heart Assoc. 2018; 7:e010533.
Article115. Caunca MR, Simonetto M, Cheung YK, Alperin N, Lee SH, Elkind MSV, et al. Diastolic blood pressure is associated with regional white matter lesion load: the northern Manhattan study. Stroke. 2020; 51:372–378.
Article116. Verhaaren BF, Vernooij MW, de Boer R, Hofman A, Niessen WJ, van der Lugt A, et al. High blood pressure and cerebral white matter lesion progression in the general population. Hypertension. 2013; 61:1354–1359.
Article117. Dufouil C, de Kersaint-Gilly A, Besançon V, Levy C, Auffray E, Brunnereau L, et al. Longitudinal study of blood pressure and white matter hyperintensities: the EVA MRI cohort. Neurology. 2001; 56:921–926.
Article118. Pantoni L. Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010; 9:689–701.119. Sierra C. Essential hypertension, cerebral white matter pathology and ischemic stroke. Curr Med Chem. 2014; 21:2156–2164.
Article120. Dufouil C, Chalmers J, Coskun O, Besançon V, Bousser MG, Guillon P, et al. Effects of blood pressure lowering on cerebral white matter hyperintensities in patients with stroke: the PROGRESS (perindopril protection against recurrent stroke study) magnetic resonance imaging substudy. Circulation. 2005; 112:1644–1650.
Article121. Firbank MJ, Wiseman RM, Burton EJ, Saxby BK, O’Brien JT, Ford GA. Brain atrophy and white matter hyperintensity change in older adults and relationship to blood pressure. Brain atrophy, WMH change and blood pressure. J Neurol. 2007; 254:713–721.
Article122. van Middelaar T, Argillander TE, Schreuder FHBM, Deinum J, Richard E, Klijn CJM. Effect of antihypertensive medication on cerebral small vessel disease: a systematic review and metaanalysis. Stroke. 2018; 49:1531–1533.
Article123. Kjeldsen SE, Narkiewicz K, Burnier M, Oparil S. Intensive blood pressure lowering prevents mild cognitive impairment and possible dementia and slows development of white matter lesions in brain: the SPRINT memory and cognition in decreased hypertension (SPRINT MIND) study. Blood Press. 2018; 27:247–248.
Article124. SPRINT MIND Investigators for the SPRINT Research Group. Association of intensive vs standard blood pressure control with cerebral white matter lesions. JAMA. 2019; 322:524–534.125. Cox SR, Lyall DM, Ritchie SJ, Bastin ME, Harris MA, Buchanan CR, et al. Associations between vascular risk factors and brain MRI indices in UK Biobank. Eur Heart J. 2019; 40:2290–2300.
Article126. Veldink JH, Scheltens P, Jonker C, Launer LJ. Progression of cerebral white matter hyperintensities on MRI is related to diastolic blood pressure. Neurology. 1998; 51:319–320.
Article127. Zhang D, Tang Y, Ge J, Liu Y, Jin J, He M. Age and diastolic blood pressure play an important role in the progression of white matter lesions: a meta-analysis. Eur Neurol. 2020; 83:351–359.
Article128. Pantoni L, Garcia JH. The significance of cerebral white matter abnormalities 100 years after Binswanger’s report. A review. Stroke. 1995; 26:1293–1301.
Article129. Topakian R, Barrick TR, Howe FA, Markus HS. Blood-brain barrier permeability is increased in normal-appearing white matter in patients with lacunar stroke and leucoaraiosis. J Neurol Neurosurg Psychiatry. 2010; 81:192–197.130. Oishi E, Ohara T, Sakata S, Fukuhara M, Hata J, Yoshida D, et al. Day-to-day blood pressure variability and risk of dementia in a general Japanese elderly population: the Hisayama study. Circulation. 2017; 136:516–525.
Article131. Stevens SL, Wood S, Koshiaris C, Law K, Glasziou P, Stevens RJ, et al. Blood pressure variability and cardiovascular disease: systematic review and meta-analysis. BMJ. 2016; 354:i4098.
Article132. Alpérovitch A, Blachier M, Soumaré A, Ritchie K, Dartigues JF, Richard-Harston S, et al. Blood pressure variability and risk of dementia in an elderly cohort, the three-city study. Alzheimers Dement. 2014; 10(5 Suppl):S330–S337.
Article133. Gunstad J, Cohen RA, Tate DF, Paul RH, Poppas A, Hoth K, et al. Blood pressure variability and white matter hyperintensities in older adults with cardiovascular disease. Blood Press. 2005; 14:353–358.
Article134. Brickman AM, Reitz C, Luchsinger JA, Manly JJ, Schupf N, Muraskin J, et al. Long-term blood pressure fluctuation and cerebrovascular disease in an elderly cohort. Arch Neurol. 2010; 67:564–569.135. Liu Z, Zhao Y, Zhang H, Chai Q, Cui Y, Diao Y, et al. Excessive variability in systolic blood pressure that is self-measured at home exacerbates the progression of brain white matter lesions and cognitive impairment in the oldest old. Hypertens Res. 2016; 39:245–253.
Article136. Filomena J, Riba-Llena I, Vinyoles E, Tovar JL, Mundet X, Castañé X, et al. Short-term blood pressure variability relates to the presence of subclinical brain small vessel disease in primary hypertension. Hypertension. 2015; 66:634–640. discussion 445.
Article137. Yang S, Yuan J, Qin W, Yang L, Fan H, Li Y, et al. Twenty-four-hour ambulatory blood pressure variability is associated with total magnetic resonance imaging burden of cerebral small-vessel disease. Clin Interv Aging. 2018; 13:1419–1427.138. Chen X, Zhu Y, Geng S, Li Q, Jiang H. Association of blood pressure variability and intima-media thickness with white matter hyperintensities in hypertensive patients. Front Aging Neurosci. 2019; 11:192.
Article139. van Middelaar T, Richard E, Moll van Charante EP, van Gool WA, van Dalen JW. Visit-to-visit blood pressure variability and progression of white matter hyperintensities among older people with hypertension. J Am Med Dir Assoc. 2019; 20:1175–1177.e1.140. Shen J, Yang L, Xu Z, Wei W. Association between twenty-four-hour ambulatory blood pressure variability and cerebral small vessel disease burden in acute ischemic stroke. Behav Neurol. 2022; 2022:3769577.
Article141. Zhang B, Huo Y, Yang Z, Lv H, Wang Y, Feng J, et al. Day to day blood pressure variability associated with cerebral arterial dilation and white matter hyperintensity. Hypertension. 2022; 79:1455–1465.
Article142. Tully PJ, Yano Y, Launer LJ, Kario K, Nagai M, Mooijaart SP, et al. Association between blood pressure variability and cerebral small-vessel disease: a systematic review and meta-analysis. J Am Heart Assoc. 2020; 9:e013841.143. Havlik RJ, Foley DJ, Sayer B, Masaki K, White L, Launer LJ. Variability in midlife systolic blood pressure is related to latelife brain white matter lesions: the Honolulu-Asia aging study. Stroke. 2002; 33:26–30.
Article144. Zhou TL, Rensma SP, van der Heide FCT, Henry RMA, Kroon AA, Houben AJHM, et al. Blood pressure variability and microvascular dysfunction: the Maastricht study. J Hypertens. 2020; 38:1541–1550.145. Jiang X, Guo Y, Zhao Y, Gao X, Peng D, Zhang H, et al. Multiscale dynamics of blood pressure fluctuation is associated with white matter lesion burden in older adults with and without hypertension: observations from a pilot study. Front Cardiovasc Med. 2021; 8:636702.
Article146. Starmans NL, Wolters FJ, Leeuwis AE, Bron EE, Brunner La Rocca HP, Staals J, et al. Twenty-four hour blood pressure variability and the prevalence and the progression of cerebral white matter hyperintensities. J Cereb Blood Flow Metab. 2023; 43:801–811.
Article147. Ma Y, Song A, Viswanathan A, Blacker D, Vernooij MW, Hofman A, et al. Blood pressure variability and cerebral small vessel disease: a systematic review and meta-analysis of population-based cohorts. Stroke. 2020; 51:82–89.
Article148. Jeerakathil T, Wolf PA, Beiser A, Massaro J, Seshadri S, D’Agostino RB, et al. Stroke risk profile predicts white matter hyperintensity volume: the Framingham study. Stroke. 2004; 35:1857–1861.
Article149. Jongen C, van der Grond J, Kappelle LJ, Biessels GJ, Viergever MA, Pluim JP; Utrecht Diabetic Encephalopathy Study Group. Automated measurement of brain and white matter lesion volume in type 2 diabetes mellitus. Diabetologia. 2007; 50:1509–1516.150. King KS, Vintimilla RM, Braskie MN, Wei K, Hall JR, Borzage M, et al. Vascular risk profile and white matter hyperintensity volume among Mexican Americans and non-Hispanic Whites: the HABLE study. Alzheimers Dement (Amst). 2022; 14:e12263.
Article151. Guan J, Yan C, Gao Q, Li J, Wang L, Hong M, et al. Analysis of risk factors in patients with leukoaraiosis. Medicine (Baltimore). 2017; 96:e6153.
Article152. Hilal S, Mok V, Youn YC, Wong A, Ikram MK, Chen CL. Prevalence, risk factors and consequences of cerebral small vessel diseases: data from three Asian countries. J Neurol Neurosurg Psychiatry. 2017; 88:669–674.
Article153. Debette S, Seshadri S, Beiser A, Au R, Himali JJ, Palumbo C, et al. Midlife vascular risk factor exposure accelerates structural brain aging and cognitive decline. Neurology. 2011; 77:461–468.
Article154. Power MC, Deal JA, Sharrett AR, Jack CR Jr, Knopman D, Mosley TH, et al. Smoking and white matter hyperintensity progression: the ARIC-MRI study. Neurology. 2015; 84:841–848.155. Das AS, Regenhardt RW, Vernooij MW, Blacker D, Charidimou A, Viswanathan A. Asymptomatic cerebral small vessel disease: insights from population-based studies. J Stroke. 2019; 21:121–138.
Article156. Jimenez-Conde J, Biffi A, Rahman R, Kanakis A, Butler C, Sonni S, et al. Hyperlipidemia and reduced white matter hyperintensity volume in patients with ischemic stroke. Stroke. 2010; 41:437–442.
Article157. Ohwaki K, Yano E, Tamura A, Inoue T, Saito I. Hypercholesterolemia is associated with a lower risk of cerebral ischemic small vessel disease detected on brain checkups. Clin Neurol Neurosurg. 2013; 115:669–672.
Article158. Ke D, Zhou F, Liang H, Xu Y, Lou H. Hypertriglyceridemia is associated with reduced leukoaraiosis severity in patients with a small vessel stroke. Behav Neurol. 2018; 2018:1361780.
Article159. Wang Z, Chen Q, Chen J, Yang N, Zheng K. Risk factors of cerebral small vessel disease: a systematic review and metaanalysis. Medicine (Baltimore). 2021; 100:e28229.160. Okamura T, Hashimoto Y, Hamaguchi M, Ohbora A, Kojima T, Fukui M. Metabolically healthy obesity and risk of leukoaraiosis; a population based cross-sectional study. Endocr J. 2018; 65:669–675.
Article161. Coutinho T, Turner ST, Kullo IJ. Aortic pulse wave velocity is associated with measures of subclinical target organ damage. JACC Cardiovasc Imaging. 2011; 4:754–761.
Article162. Mitchell GF, van Buchem MA, Sigurdsson S, Gotal JD, Jonsdottir MK, Kjartansson Ó, et al. Arterial stiffness, pressure and flow pulsatility and brain structure and function: the age, gene/environment susceptibility--Reykjavik study. Brain. 2011; 134(Pt 11):3398–3407.
Article163. Poels MM, Zaccai K, Verwoert GC, Vernooij MW, Hofman A, van der Lugt A, et al. Arterial stiffness and cerebral small vessel disease: the Rotterdam scan study. Stroke. 2012; 43:2637–2642.
Article164. Saji N, Shimizu H, Kawarai T, Tadano M, Kita Y, Yokono K. Increased brachial-ankle pulse wave velocity is independently associated with white matter hyperintensities. Neuroepidemiology. 2011; 36:252–257.
Article165. Tsao CW, Seshadri S, Beiser AS, Westwood AJ, Decarli C, Au R, et al. Relations of arterial stiffness and endothelial function to brain aging in the community. Neurology. 2013; 81:984–991.
Article166. Singer J, Trollor JN, Baune BT, Sachdev PS, Smith E. Arterial stiffness, the brain and cognition: a systematic review. Ageing Res Rev. 2014; 15:16–27.
Article167. van Sloten TT, Protogerou AD, Henry RM, Schram MT, Launer LJ, Stehouwer CD. Association between arterial stiffness, cerebral small vessel disease and cognitive impairment: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2015; 53:121–130.
Article168. Haidegger M, Lindenbeck S, Hofer E, Rodler C, Zweiker R, Perl S, et al. Arterial stiffness and its influence on cerebral morphology and cognitive function. Ther Adv Neurol Disord. 2023; 16:17562864231180715.
Article169. Caughey MC, Qiao Y, Meyer ML, Palta P, Matsushita K, Tanaka H, et al. Relationship between central artery stiffness, brain arterial dilation, and white matter hyperintensities in older adults: the ARIC study—brief report. Arterioscler Thromb Vasc Biol. 2021; 41:2109–2116.170. Robert C, Ling LH, Tan ESJ, Gyanwali B, Venketasubramanian N, Lim SL, et al. Effects of carotid artery stiffness on cerebral small-vessel disease and cognition. J Am Heart Assoc. 2022; 11:e027295.
Article171. Miyagi T, Ishida A, Shinzato T, Ohya Y. Arterial stiffness is associated with small vessel disease irrespective of blood pressure in stroke-free individuals. Stroke. 2023; 54:2814–2821.
Article172. Tomoto T, Tarumi T, Zhang R. Central arterial stiffness, brain white matter hyperintensity and total brain volume across the adult lifespan. J Hypertens. 2023; 41:819–829.
Article173. Brandts A, van Elderen SG, Westenberg JJ, van der Grond J, van Buchem MA, Huisman MV, et al. Association of aortic arch pulse wave velocity with left ventricular mass and lacunar brain infarcts in hypertensive patients: assessment with MR imaging. Radiology. 2009; 253:681–688.
Article174. van Elderen SG, Brandts A, Westenberg JJ, van der Grond J, Tamsma JT, van Buchem MA, et al. Aortic stiffness is associated with cardiac function and cerebral small vessel disease in patients with type 1 diabetes mellitus: assessment by magnetic resonance imaging. Eur Radiol. 2010; 20:1132–1138.
Article175. Ohmine T, Miwa Y, Yao H, Yuzuriha T, Takashima Y, Uchino A, et al. Association between arterial stiffness and cerebral white matter lesions in community-dwelling elderly subjects. Hypertens Res. 2008; 31:75–81.
Article176. Hannawi Y, Vaidya D, Yanek LR, Johansen MC, Kral BG, Becker LC, et al. Association of vascular properties with the brain white matter hyperintensity in middle-aged population. J Am Heart Assoc. 2022; 11:e024606.
Article177. Funck KL, Laugesen E, Høyem P, Stausbøl-Grøn B, Kim WY, Østergaard L, et al. Arterial stiffness and progression of cerebral white matter hyperintensities in patients with type 2 diabetes and matched controls: a 5-year cohort study. Diabetol Metab Syndr. 2021; 13:71.
Article178. Del Brutto OH, Mera RM, Costa AF, Recalde BY, Rumbea DA, Sedler MJ. Arterial stiffness and progression of white matter hyperintensities of presumed vascular origin in communitydwelling older adults of Amerindian ancestry: the Atahualpa project cohort. Clin Neurol Neurosurg. 2022; 221:107411.
Article179. Allison EY, Al-Khazraji BK. Association of arterial stiffness index and brain structure in the UK Biobank: a 10-year retrospective analysis. Aging Dis. 2023 Jun 8 [Epub]. https://doi.org/10.14336/AD.2023.0419.
Article180. Scheuermann BC, Parr SK, Schulze KM, Kunkel ON, Turpin VG, Liang J, et al. Associations of cerebrovascular regulation and arterial stiffness with cerebral small vessel disease: a systematic review and meta-analysis. J Am Heart Assoc. 2023; 12:e032616.
Article181. Hassan A, Hunt BJ, O’Sullivan M, Bell R, D’Souza R, Jeffery S, et al. Homocysteine is a risk factor for cerebral small vessel disease, acting via endothelial dysfunction. Brain. 2004; 127(Pt 1):212–219.
Article182. Welch GN, Loscalzo J. Homocysteine and atherothrombosis. N Engl J Med. 1998; 338:1042–1050.
Article183. Kamath AF, Chauhan AK, Kisucka J, Dole VS, Loscalzo J, Handy DE, et al. Elevated levels of homocysteine compromise blood-brain barrier integrity in mice. Blood. 2006; 107:591–593.
Article184. Wald DS, Law M, Morris JK. Homocysteine and cardiovascular disease: evidence on causality from a meta-analysis. BMJ. 2002; 325:1202.
Article185. Homocysteine Studies Collaboration. Homocysteine and risk of ischemic heart disease and stroke: a meta-analysis. JAMA. 2002; 288:2015–2022.186. Boushey CJ, Beresford SA, Omenn GS, Motulsky AG. A quantitative assessment of plasma homocysteine as a risk factor for vascular disease. Probable benefits of increasing folic acid intakes. JAMA. 1995; 274:1049–1057.
Article187. Dufouil C, Alpérovitch A, Ducros V, Tzourio C. Homocysteine, white matter hyperintensities, and cognition in healthy elderly people. Ann Neurol. 2003; 53:214–221.
Article188. Longstreth WT Jr, Katz R, Olson J, Bernick C, Carr JJ, Malinow MR, et al. Plasma total homocysteine levels and cranial magnetic resonance imaging findings in elderly persons: the cardiovascular health study. Arch Neurol. 2004; 61:67–72.
Article189. Seshadri S, Wolf PA, Beiser AS, Selhub J, Au R, Jacques PF, et al. Association of plasma total homocysteine levels with subclinical brain injury: cerebral volumes, white matter hyperintensity, and silent brain infarcts at volumetric magnetic resonance imaging in the Framingham Offspring Study. Arch Neurol. 2008; 65:642–649.190. Hogervorst E, Ribeiro HM, Molyneux A, Budge M, Smith AD. Plasma homocysteine levels, cerebrovascular risk factors, and cerebral white matter changes (leukoaraiosis) in patients with Alzheimer disease. Arch Neurol. 2002; 59:787–793.191. Vermeer SE, van Dijk EJ, Koudstaal PJ, Oudkerk M, Hofman A, Clarke R, et al. Homocysteine, silent brain infarcts, and white matter lesions: the Rotterdam scan study. Ann Neurol. 2002; 51:285–289.
Article192. Wright CB, Paik MC, Brown TR, Stabler SP, Allen RH, Sacco RL, et al. Total homocysteine is associated with white matter hyperintensity volume: the northern Manhattan study. Stroke. 2005; 36:1207–1211.
Article193. Naka H, Nomura E, Takahashi T, Wakabayashi S, Kajikawa H, Kohriyama T, et al. Plasma total homocysteine levels are associated with advanced leukoaraiosis but not with asymptomatic microbleeds on T2*-weighted MRI in patients with stroke. Eur J Neurol. 2006; 13:261–265.
Article194. Shimomura T, Anan F, Umeno Y, Eshima N, Saikawa T, Yoshimatsu H, et al. Hyperhomocysteinaemia is a significant risk factor for white matter lesions in Japanese type 2 diabetic patients. Eur J Neurol. 2008; 15:289–294.
Article195. Anan F, Masaki T, Tatsukawa H, Nagano S, Oribe M, Eshima N, et al. The role of homocysteine as a significant risk factor for white matter lesions in Japanese women with rheumatoid arthritis. Metabolism. 2009; 58:69–73.
Article196. Tseng YL, Chang YY, Liu JS, Su CS, Lai SL, Lan MY. Association of plasma homocysteine concentration with cerebral white matter hyperintensity on magnetic resonance images in stroke patients. J Neurol Sci. 2009; 284:36–39.
Article197. Pavlovic AM, Pekmezovic T, Obrenovic R, Novakovic I, Tomic G, Mijajlovic M, et al. Increased total homocysteine level is associated with clinical status and severity of white matter changes in symptomatic patients with subcortical small vessel disease. Clin Neurol Neurosurg. 2011; 113:711–715.
Article198. Raz N, Yang Y, Dahle CL, Land S. Volume of white matter hyperintensities in healthy adults: contribution of age, vascular risk factors, and inflammation-related genetic variants. Biochim Biophys Acta. 2012; 1822:361–369.
Article199. Cloonan L, Fitzpatrick KM, Kanakis AS, Furie KL, Rosand J, Rost NS. Metabolic determinants of white matter hyperintensity burden in patients with ischemic stroke. Atherosclerosis. 2015; 240:149–153.
Article200. Shan Y, Tan S, Wang Y, Li K, Zhang L, Liao S, et al. Risk factors and clinical manifestations of juxtacortical small lesions: a neuroimaging study. Front Neurol. 2017; 8:497.
Article201. Piao X, Wu G, Yang P, Shen J, De A, Wu J, et al. Association between homocysteine and cerebral small vessel disease: a meta-analysis. J Stroke Cerebrovasc Dis. 2018; 27:2423–2430.
Article202. Shen Y, Dong ZF, Pan PL, Xu G, Huang JY, Liu CF. Association of homocysteine, folate, and white matter hyperintensities in Parkinson’s patients with different motor phenotypes. Neurol Sci. 2019; 40:1855–1863.
Article203. Nam KW, Kwon HM, Jeong HY, Park JH, Kwon H, Jeong SM. Serum homocysteine level is related to cerebral small vessel disease in a healthy population. Neurology. 2019; 92:e317–e325.
Article204. Wang X, Yin H, Ji X, Sang S, Shao S, Wang G, et al. Association between homocysteine and white matter hyperintensities in rural-dwelling Chinese people with asymptomatic intracranial arterial stenosis: a population-based study. Brain Behav. 2021; 11:e02205.
Article205. Kloppenborg RP, Geerlings MI, Visseren FL, Mali WP, Vermeulen M, van der Graaf Y, et al. Homocysteine and progression of generalized small-vessel disease: the SMART-MR study. Neurology. 2014; 82:777–783.
Article206. Hooshmand B, Mangialasche F, Kalpouzos G, Solomon A, Kåreholt I, Smith AD, et al. Association of vitamin B12, folate, and sulfur amino acids with brain magnetic resonance imaging measures in older adults: a longitudinal population-based study. JAMA Psychiatry. 2016; 73:606–613.
Article207. Sachdev P, Parslow R, Salonikas C, Lux O, Wen W, Kumar R, et al. Homocysteine and the brain in midadult life: evidence for an increased risk of leukoaraiosis in men. Arch Neurol. 2004; 61:1369–1376.208. Gao Y, Wei S, Song B, Qin J, Fang H, Ji Y, et al. Homocysteine level is associated with white matter hyperintensity locations in patients with acute ischemic stroke. PLoS One. 2015; 10:e0144431.
Article209. Lee KO, Woo MH, Chung D, Choi JW, Kim NK, Kim OJ, et al. Differential impact of plasma homocysteine levels on the periventricular and subcortical white matter hyperintensities on the brain. Front Neurol. 2019; 10:1174.
Article210. Refsum H, Smith AD, Ueland PM, Nexo E, Clarke R, McPartlin J, et al. Facts and recommendations about total homocysteine determinations: an expert opinion. Clin Chem. 2004; 50:3–32.211. Selhub J, Jacques PF, Wilson PW, Rush D, Rosenberg IH. Vitamin status and intake as primary determinants of homocysteinemia in an elderly population. JAMA. 1993; 270:2693–2698.
Article212. Clarke R, Smith AD, Jobst KA, Refsum H, Sutton L, Ueland PM. Folate, vitamin B12, and serum total homocysteine levels in confirmed Alzheimer disease. Arch Neurol. 1998; 55:1449–1455.
Article213. Stabler SP. Vitamin B12 deficiency. N Engl J Med. 2013; 368:149–160.214. Selhub J, Bagley LC, Miller J, Rosenberg IH. B vitamins, homocysteine, and neurocognitive function in the elderly. Am J Clin Nutr. 2000; 71:614S–620S.
Article215. Kado DM, Karlamangla AS, Huang MH, Troen A, Rowe JW, Selhub J, et al. Homocysteine versus the vitamins folate, B6, and B12 as predictors of cognitive function and decline in older high-functioning adults: MacArthur studies of successful aging. Am J Med. 2005; 118:161–167.
Article216. Pieters B, Staals J, Knottnerus I, Rouhl R, Menheere P, Kessels A, et al. Periventricular white matter lucencies relate to low vitamin B12 levels in patients with small vessel stroke. Stroke. 2009; 40:1623–1626.
Article217. de Lau LM, Smith AD, Refsum H, Johnston C, Breteler MM. Plasma vitamin B12 status and cerebral white-matter lesions. J Neurol Neurosurg Psychiatry. 2009; 80:149–157.
Article218. van Overbeek EC, Staals J, van Oostenbrugge RJ. Vitamin B12 and progression of white matter lesions. A 2-year follow-up study in first-ever lacunar stroke patients. PLoS One. 2013; 8:e78100.
Article219. Iosifescu DV, Papakostas GI, Lyoo IK, Lee HK, Renshaw PF, Alpert JE, et al. Brain MRI white matter hyperintensities and one-carbon cycle metabolism in non-geriatric outpatients with major depressive disorder (part I). Psychiatry Res. 2005; 140:291–299.
Article220. Scott TM, Tucker KL, Bhadelia A, Benjamin B, Patz S, Bhadelia R, et al. Homocysteine and B vitamins relate to brain volume and white-matter changes in geriatric patients with psychiatric disorders. Am J Geriatr Psychiatry. 2004; 12:631–638.221. Hickie I, Naismith S, Ward PB, Scott E, Mitchell P, Wilhelm K, et al. Vascular risk and low serum B12 predict white matter lesions in patients with major depression. J Affect Disord. 2005; 85:327–332.
Article222. Tangney CC, Aggarwal NT, Li H, Wilson RS, Decarli C, Evans DA, et al. Vitamin B12, cognition, and brain MRI measures: a cross-sectional examination. Neurology. 2011; 77:1276–1282.
Article223. Narayan SK, Firbank MJ, Saxby BK, Stansby G, Hansrani M, O’Brien JT, et al. Elevated plasma homocysteine is associated with increased brain atrophy rates in older subjects with mild hypertension. Dement Geriatr Cogn Disord. 2011; 31:341–348.
Article224. Sponne IE, Gaire D, Stabler SP, Droesch S, Barbé FM, Allen RH, et al. Inhibition of vitamin B12 metabolism by OH-cobalamin c-lactam in rat oligodendrocytes in culture: a model for studying neuropathy due to vitamin B12 deficiency. Neurosci Lett. 2000; 288:191–194.
Article225. Kim S, Lim IK, Park GH, Paik WK. Biological methylation of myelin basic protein: enzymology and biological significance. Int J Biochem Cell Biol. 1997; 29:743–751.
Article226. Annweiler C, Allali G, Allain P, Bridenbaugh S, Schott AM, Kressig RW, et al. Vitamin D and cognitive performance in adults: a systematic review. Eur J Neurol. 2009; 16:1083–1089.
Article227. Sultan S. Neuroimaging changes associated with vitamin D deficiency–a narrative review. Nutr Neurosci. 2022; 25:1650–1658.
Article228. Zhao Y, Xu J, Feng Z, Wang J. Impact of 25-hydroxy vitamin D on white matter hyperintensity in elderly patients: a systematic review and meta-analysis. Front Neurol. 2022; 12:721427.
Article229. Buell JS, Dawson-Hughes B, Scott TM, Weiner DE, Dallal GE, Qui WQ, et al. 25-hydroxyvitamin D, dementia, and cerebrovascular pathology in elders receiving home services. Neurology. 2010; 74:18–26.
Article230. Prager JM, Thomas C, Ankenbrandt WJ, Meyer JR, Gao Y, Ragin A, et al. Association of white matter hyperintensities with low serum 25-hydroxyvitamin D levels. AJNR Am J Neuroradiol. 2014; 35:1145–1149.231. Chung PW, Park KY, Kim JM, Shin DW, Park MS, Chung YJ, et al. 25-hydroxyvitamin D status is associated with chronic cerebral small vessel disease. Stroke. 2015; 46:248–251.
Article232. Feng C, Tang N, Huang H, Zhang G, Qi X, Shi F. 25-Hydroxy vitamin D level is associated with total MRI burden of cerebral small vessel disease in ischemic stroke patients. Int J Neurosci. 2019; 129:49–54.
Article233. Annweiler C, Bartha R, Karras SN, Gautier J, Roche F, Beauchet O. Vitamin D and white matter abnormalities in older adults: a quantitative volumetric analysis of brain MRI. Exp Gerontol. 2015; 63:41–47.
Article234. Wang L, Zhao XM, Yuan XZ, Wang FY, Shen J, Wang Y. Association between serum 25-hydroxyvitamin D level and cognitive impairment in patients with white matter lesions: a cross-sectional study. Med Princ Pract. 2020; 29:451–457.
Article235. Sakurai T, Ogama N, Toba K. Lower vitamin D is associated with white matter hyperintensity in elderly women with Alzheimer’s disease and amnestic mild cognitive impairment. J Am Geriatr Soc. 2014; 62:1993–1994.
Article236. Schramm S, Schliephake L, Himpfen H, Caspers S, Erbel R, Jöckel KH, et al. Vitamin D and white matter hyperintensities: results of the population-based Heinz Nixdorf Recall Study and 1000BRAINS. Eur J Neurol. 2021; 28:1849–1858.
Article237. Annweiler C, Annweiler T, Bartha R, Herrmann FR, Camicioli R, Beauchet O. Vitamin D and white matter abnormalities in older adults: a cross-sectional neuroimaging study. Eur J Neurol. 2014; 21:1436–e95.
Article238. Michos ED, Carson KA, Schneider AL, Lutsey PL, Xing L, Sharrett AR, et al. Vitamin D and subclinical cerebrovascular disease: the atherosclerosis risk in communities brain magnetic resonance imaging study. JAMA Neurol. 2014; 71:863–871.239. Littlejohns TJ, Kos K, Henley WE, Lang IA, Annweiler C, Beauchet O, et al. Vitamin D and risk of neuroimaging abnormalities. PLoS One. 2016; 11:e0154896.
Article240. Karakis I, Pase MP, Beiser A, Booth SL, Jacques PF, Rogers G, et al. Association of serum vitamin D with the risk of incident dementia and subclinical indices of brain aging: the Framingham heart study. J Alzheimers Dis. 2016; 51:451–461.
Article241. Bowman GL, Silbert LC, Howieson D, Dodge HH, Traber MG, Frei B, et al. Nutrient biomarker patterns, cognitive function, and MRI measures of brain aging. Neurology. 2012; 78:241–249.
Article242. Brouwer-Brolsma EM, van der Zwaluw NL, van Wijngaarden JP, Dhonukshe-Rutten RA, in’t Veld PH, Feskens EJ, et al. Higher serum 25-hydroxyvitamin D and lower plasma glucose are associated with larger gray matter volume but not with white matter or total brain volume in Dutch community-dwelling older adults. J Nutr. 2015; 145:1817–1823.
Article243. Putaala J, Kurkinen M, Tarvos V, Salonen O, Kaste M, Tatlisumak T. Silent brain infarcts and leukoaraiosis in young adults with first-ever ischemic stroke. Neurology. 2009; 72:1823–1829.
Article244. Kim KW, Seo H, Kwak MS, Kim D. Visceral obesity is associated with white matter hyperintensity and lacunar infarct. Int J Obes (Lond). 2017; 41:683–688.
Article245. Lampe L, Zhang R, Beyer F, Huhn S, Kharabian Masouleh S, Preusser S, et al. Visceral obesity relates to deep white matter hyperintensities via inflammation. Ann Neurol. 2019; 85:194–203.
Article246. Alqarni A, Jiang J, Crawford JD, Koch F, Brodaty H, Sachdev P, et al. Sex differences in risk factors for white matter hyperintensities in non-demented older individuals. Neurobiol Aging. 2021; 98:197–204.247. Seixas AA, Turner AD, Bubu OM, Jean-Louis G, de Leon MJ, Osorio RS, et al. Obesity and race may explain differential burden of white matter hyperintensity load. Clin Interv Aging. 2021; 16:1563–1571.
Article248. Murray AD, McNeil CJ, Salarirad S, Whalley LJ, Staff RT. Early life socioeconomic circumstance and late life brain hyperintensities--a population based cohort study. PLoS One. 2014; 9:e88969.249. Mortamais M, Portet F, Brickman AM, Provenzano FA, Muraskin J, Akbaraly TN, et al. Education modulates the impact of white matter lesions on the risk of mild cognitive impairment and dementia. Am J Geriatr Psychiatry. 2014; 22:1336–1345.
Article250. Schretlen DJ, Inscore AB, Vannorsdall TD, Kraut M, Pearlson GD, Gordon B, et al. Serum uric acid and brain ischemia in normal elderly adults. Neurology. 2007; 69:1418–1423.
Article251. Maniwa K, Yano S, Sheikh AM, Onoda K, Mitaki S, Isomura M, et al. Association between cystatin C gene polymorphism and the prevalence of white matter lesion in elderly healthy subjects. Sci Rep. 2020; 10:4688.
Article252. Backhouse EV, McHutchison CA, Cvoro V, Shenkin SD, Wardlaw JM. Early life risk factors for cerebrovascular disease: a systematic review and meta-analysis. Neurology. 2017; 88:976–984.
Article253. Christensen H, Batterham PJ, Mackinnon AJ, Anstey KJ, Wen W, Sachdev PS. Education, atrophy, and cognitive change in an epidemiological sample in early old age. Am J Geriatr Psychiatry. 2009; 17:218–226.
Article254. Elkins JS, Longstreth WT Jr, Manolio TA, Newman AB, Bhadelia RA, Johnston SC. Education and the cognitive decline associated with MRI-defined brain infarct. Neurology. 2006; 67:435–440.
Article255. Breteler MM, van Swieten JC, Bots ML, Grobbee DE, Claus JJ, van den Hout JH, et al. Cerebral white matter lesions, vascular risk factors, and cognitive function in a population-based study: the Rotterdam study. Neurology. 1994; 44:1246–1252.
Article256. Habes M, Sotiras A, Erus G, Toledo JB, Janowitz D, Wolk DA, et al. White matter lesions: spatial heterogeneity, links to risk factors, cognition, genetics, and atrophy. Neurology. 2018; 91:e964–e975.257. Sachdev PS, Parslow R, Wen W, Anstey KJ, Easteal S. Sex differences in the causes and consequences of white matter hyperintensities. Neurobiol Aging. 2009; 30:946–956.
Article258. Sullivan P, Pary R, Telang F, Rifai AH, Zubenko GS. Risk factors for white matter changes detected by magnetic resonance imaging in the elderly. Stroke. 1990; 21:1424–1428.
Article259. Schmidt R, Fazekas F, Kleinert G, Offenbacher H, Gindl K, Payer F, et al. Magnetic resonance imaging signal hyperintensities in the deep and subcortical white matter. A comparative study between stroke patients and normal volunteers. Arch Neurol. 1992; 49:825–827.
Article260. Assareh AA, Mather KA, Crawford JD, Wen W, Anstey KJ, Easteal S, et al. Renin-angiotensin system genetic polymorphisms and brain white matter lesions in older Australians. Am J Hypertens. 2014; 27:1191–1198.
Article261. Geerlings MI, Appelman AP, Vincken KL, Algra A, Witkamp TD, Mali WP, et al. Brain volumes and cerebrovascular lesions on MRI in patients with atherosclerotic disease. The SMARTMR study. Atherosclerosis. 2010; 210:130–136.
Article262. Brickman AM, Schupf N, Manly JJ, Luchsinger JA, Andrews H, Tang MX, et al. Brain morphology in older African Americans, Caribbean Hispanics, and whites from northern Manhattan. Arch Neurol. 2008; 65:1053–1061.
Article263. Nyquist PA, Bilgel MS, Gottesman R, Yanek LR, Moy TF, Becker LC, et al. Extreme deep white matter hyperintensity volumes are associated with African American race. Cerebrovasc Dis. 2014; 37:244–250.
Article264. Gottesman RF, Coresh J, Catellier DJ, Sharrett AR, Rose KM, Coker LH, et al. Blood pressure and white-matter disease progression in a biethnic cohort: atherosclerosis risk in communities (ARIC) study. Stroke. 2010; 41:3–8.
Article265. Turner ST, Jack CR, Fornage M, Mosley TH, Boerwinkle E, de Andrade M. Heritability of leukoaraiosis in hypertensive sibships. Hypertension. 2004; 43:483–487.
Article266. Atwood LD, Wolf PA, Heard-Costa NL, Massaro JM, Beiser A, D’Agostino RB, et al. Genetic variation in white matter hyperintensity volume in the Framingham study. Stroke. 2004; 35:1609–1613.
Article267. Sachdev PS, Thalamuthu A, Mather KA, Ames D, Wright MJ, Wen W; OATS Collaborative Research Team. White matter hyperintensities are under strong genetic influence. Stroke. 2016; 47:1422–1428.
Article268. Paternoster L, Chen W, Sudlow CL. Genetic determinants of white matter hyperintensities on brain scans: a systematic assessment of 19 candidate gene polymorphisms in 46 studies in 19,000 subjects. Stroke. 2009; 40:2020–2026.
Article269. Lopez LM, Hill WD, Harris SE, Valdes Hernandez M, Munoz Maniega S, Bastin ME, et al. Genes from a translational analysis support a multifactorial nature of white matter hyperintensities. Stroke. 2015; 46:341–347.
Article270. Jian X, Satizabal CL, Smith AV, Wittfeld K, Bis JC, Smith JA, et al. Exome chip analysis identifies low-frequency and rare variants in MRPL38 for white matter hyperintensities on brain magnetic resonance imaging. Stroke. 2018; 49:1812–1819.
Article271. Yadav BK, Shin BS. Single-nucleotide polymorphisms of tight junction component claudin-1 associated with leukoaraiosis. J Stroke Cerebrovasc Dis. 2015; 24:1662–1670.
Article272. Oliveira-Filho J, Ornellas AC, Zhang CR, Oliveira LM, AraújoSantos T, Borges VM, et al. COX-2 rs20417 polymorphism is associated with stroke and white matter disease. J Stroke Cerebrovasc Dis. 2015; 24:1817–1822.
Article273. Zhang M, Zhu W, Yun W, Wang Q, Cheng M, Zhang Z, et al. Correlation of matrix metalloproteinase-2 single nucleotide polymorphisms with the risk of small vessel disease (SVD). J Neurol Sci. 2015; 356:61–64.
Article274. Yadav BK, Oh SY, Kim NK, Shin BS. Association of rs2075575 and rs9951307 polymorphisms of AQP-4 gene with leukoaraiosis. J Stroke Cerebrovasc Dis. 2014; 23:1199–1206.
Article275. Huang WQ, Ye HM, Li FF, Yi KH, Zhang Y, Cai LL, et al. Analysis of genetic polymorphisms associated with leukoaraiosis in the southern Chinese population: a case-control study. Medicine (Baltimore). 2016; 95:e3857.276. Huang WQ, Ye HM, Cai LL, Ma QL, Lu CX, Tong SJ, et al. The associations of PMF1, ICAM1, AGT, TRIM65, FBF1, and ACOX1 variants with leukoaraiosis in Chinese population. Front Genet. 2019; 10:615.
Article277. Yadav BK, Shin BS. Single-nucleotide polymorphisms of the adherent junction component cadherin gene are associated with leukoaraiosis. Gene. 2018; 676:65–72.
Article278. Yadav BK, Yadav R, Kang HG, Kim KW, Lee CH, Shin BS. Association of genetic variation in a Wnt signaling pathway gene (β-catenin) with susceptibility to leukoaraiosis. Genet Test Mol Biomarkers. 2020; 24:708–716.
Article279. Li J, Abedi V, Zand R, Griessenauer CJ. Replication of top loci from COL4A1/2 associated with white matter hyperintensity burden in patients with ischemic stroke. Stroke. 2020; 51:3751–3755.
Article280. Parikh NS, Dueker N, Varela D, Del Brutto VJ, Rundek T, Wright CB, et al. Association between PNPLA3 rs738409 G variant and MRI cerebrovascular disease biomarkers. J Neurol Sci. 2020; 416:116981.281. Davis CM, Bah TM, Zhang WH, Nelson JW, Golgotiu K, Nie X, et al. GPR39 localization in the aging human brain and correlation of expression and polymorphism with vascular cognitive impairment. Alzheimers Dement (N Y). 2021; 7:e12214.
Article282. Liu JY, Yao M, Dai Y, Han F, Zhai FF, Zhang DD, et al. Rare NOTCH3 variants in a Chinese population-based cohort and its relationship with cerebral small vessel disease. Stroke. 2021; 52:3918–3925.
Article283. Ferroni P, Palmirotta R, Egeo G, Aurilia C, Valente MG, Spila A, et al. Association of LTA and SOD gene polymorphisms with cerebral white matter hyperintensities in migraine patients. Int J Mol Sci. 2022; 23:13781.
Article284. Gao Y, Su B, Luo Y, Tian Y, Hong S, Gao S, et al. HLA-C*07:01 and HLA-DQB1*02:01 protect against white matter hyperintensities and deterioration of cognitive function: a population-based cohort study. Brain Behav Immun. 2024; 115:250–257.
Article285. Fornage M, Debette S, Bis JC, Schmidt H, Ikram MA, Dufouil C, et al. Genome-wide association studies of cerebral white matter lesion burden: the CHARGE consortium. Ann Neurol. 2011; 69:928–939.286. Verhaaren BF, Debette S, Bis JC, Smith JA, Ikram MK, Adams HH, et al. Multiethnic genome-wide association study of cerebral white matter hyperintensities on MRI. Circ Cardiovasc Genet. 2015; 8:398–409.287. Malik R, Beaufort N, Frerich S, Gesierich B, Georgakis MK, Rannikmäe K, et al. Whole-exome sequencing reveals a role of HTRA1 and EGFL8 in brain white matter hyperintensities. Brain. 2021; 144:2670–2682.
Article288. Traylor M, Tozer DJ, Croall ID, Lisiecka-Ford DM, Olorunda AO, Boncoraglio G, et al. Genetic variation in PLEKHG1 is associated with white matter hyperintensities (n = 11,226). Neurology. 2019; 92:e749–e757.289. Persyn E, Hanscombe KB, Howson JMM, Lewis CM, Traylor M, Markus HS. Genome-wide association study of MRI markers of cerebral small vessel disease in 42,310 participants. Nat Commun. 2020; 11:2175.
Article290. Rutten-Jacobs LCA, Tozer DJ, Duering M, Malik R, Dichgans M, Markus HS, et al. Genetic study of white matter integrity in UK Biobank (n=8448) and the overlap with stroke, depression, and dementia. Stroke. 2018; 49:1340–1347.
Article291. Armstrong NJ, Mather KA, Sargurupremraj M, Knol MJ, Malik R, Satizabal CL, et al. Common genetic variation indicates separate causes for periventricular and deep white matter hyperintensities. Stroke. 2020; 51:2111–2121.
Article292. Simpson JE, Hosny O, Wharton SB, Heath PR, Holden H, Fernando MS, et al. Microarray RNA expression analysis of cerebral white matter lesions reveals changes in multiple functional pathways. Stroke. 2009; 40:369–375.
Article293. Xu H, Stamova B, Jickling G, Tian Y, Zhan X, Ander BP, et al. Distinctive RNA expression profiles in blood associated with white matter hyperintensities in brain. Stroke. 2010; 41:2744–2749.
Article294. Lin H, Satizabal C, Xie Z, Yang Q, Huan T, Joehanes R, et al. Whole blood gene expression and white matter hyperintensities. Mol Neurodegener. 2017; 12:67.
Article295. Jickling GC, Ander BP, Zhan X, Stamova B, Hull H, DeCarli C, et al. Progression of cerebral white matter hyperintensities is related to leucocyte gene expression. Brain. 2022; 145:3179–3186.296. Hou XH, Bi YL, Tan MS, Xu W, Li JQ, Shen XN, et al. Genomewide association study identifies Alzheimer’s risk variant in MS4A6A influencing cerebrospinal fluid sTREM2 levels. Neurobiol Aging. 2019; 84:241.e13–241.e20.
Article297. Peters XQ, Malinga TH, Agoni C, Olotu FA, Soliman MES. Zoning in on Tankyrases: a brief review on the past, present and prospective studies. Anticancer Agents Med Chem. 2019; 19:1920–1934.
Article298. Yang HY, Shen JX, Wang Y, Liu Y, Shen DY, Quan S. Tankyrase promotes aerobic glycolysis and proliferation of ovarian cancer through activation of Wnt/β-catenin signaling. Biomed Res Int. 2019; 2019:2686340.299. Iwaya T, Maesawa C, Kimura T, Ogasawara S, Ikeda K, Kimura Y, et al. Infrequent mutation of the human envoplakin gene is closely linked to the tylosis oesophageal cancer locus in sporadic oesophageal squamous cell carcinomas. Oncol Rep. 2005; 13:703–707.
Article300. Weber R, Weimar C, Blatchford J, Hermansson K, Wanke I, Möller-Hartmann C, et al. Telmisartan on top of antihypertensive treatment does not prevent progression of cerebral white matter lesions in the prevention regimen for effectively avoiding second strokes (PRoFESS) MRI substudy. Stroke. 2012; 43:2336–2342.
Article301. Peng J, Lu F, Wang Z, Zhong M, Sun L, Hu N, et al. Excessive lowering of blood pressure is not beneficial for progression of brain white matter hyperintensive and cognitive impairment in elderly hypertensive patients: 4-year follow-up study. J Am Med Dir Assoc. 2014; 15:904–910.
Article302. Murray AM, Hsu FC, Williamson JD, Bryan RN, Gerstein HC, Sullivan MD, et al. ACCORDION MIND: results of the observational extension of the ACCORD MIND randomised trial. Diabetologia. 2017; 60:69–80.
Article303. Godin O, Tzourio C, Maillard P, Mazoyer B, Dufouil C. Antihypertensive treatment and change in blood pressure are associated with the progression of white matter lesion volumes: the three-city (3C)-Dijon magnetic resonance imaging study. Circulation. 2011; 123:266–273.
Article304. Schiffrin EL. Blood pressure lowering in PROGRESS (perindopril protection against recurrent stroke study) and white matter hyperintensities: should this progress matter to patients? Circulation. 2005; 112:1525–1526.
Article305. van Dalen JW, Moll van Charante EP, Caan MWA, Scheltens P, Majoie CBLM, Nederveen AJ, et al. Effect of long-term vascular care on progression of cerebrovascular lesions: magnetic resonance imaging substudy of the PreDIVA trial (prevention of dementia by intensive vascular care). Stroke. 2017; 48:1842–1848.
Article306. de Havenon A, Majersik JJ, Tirschwell DL, McNally JS, Stoddard G, Rost NS. Blood pressure, glycemic control, and white matter hyperintensity progression in type 2 diabetics. Neurology. 2019; 92:e1168–e1175.
Article307. White WB, Wakefield DB, Moscufo N, Guttmann CRG, Kaplan RF, Bohannon RW, et al. Effects of intensive versus standard ambulatory blood pressure control on cerebrovascular outcomes in older people (INFINITY). Circulation. 2019; 140:1626–1635.
Article308. Lai Y, Jiang C, Du X, Sang C, Guo X, Bai R, et al. Effect of intensive blood pressure control on the prevention of white matter hyperintensity: systematic review and meta-analysis of randomized trials. J Clin Hypertens (Greenwich). 2020; 22:1968–1973.
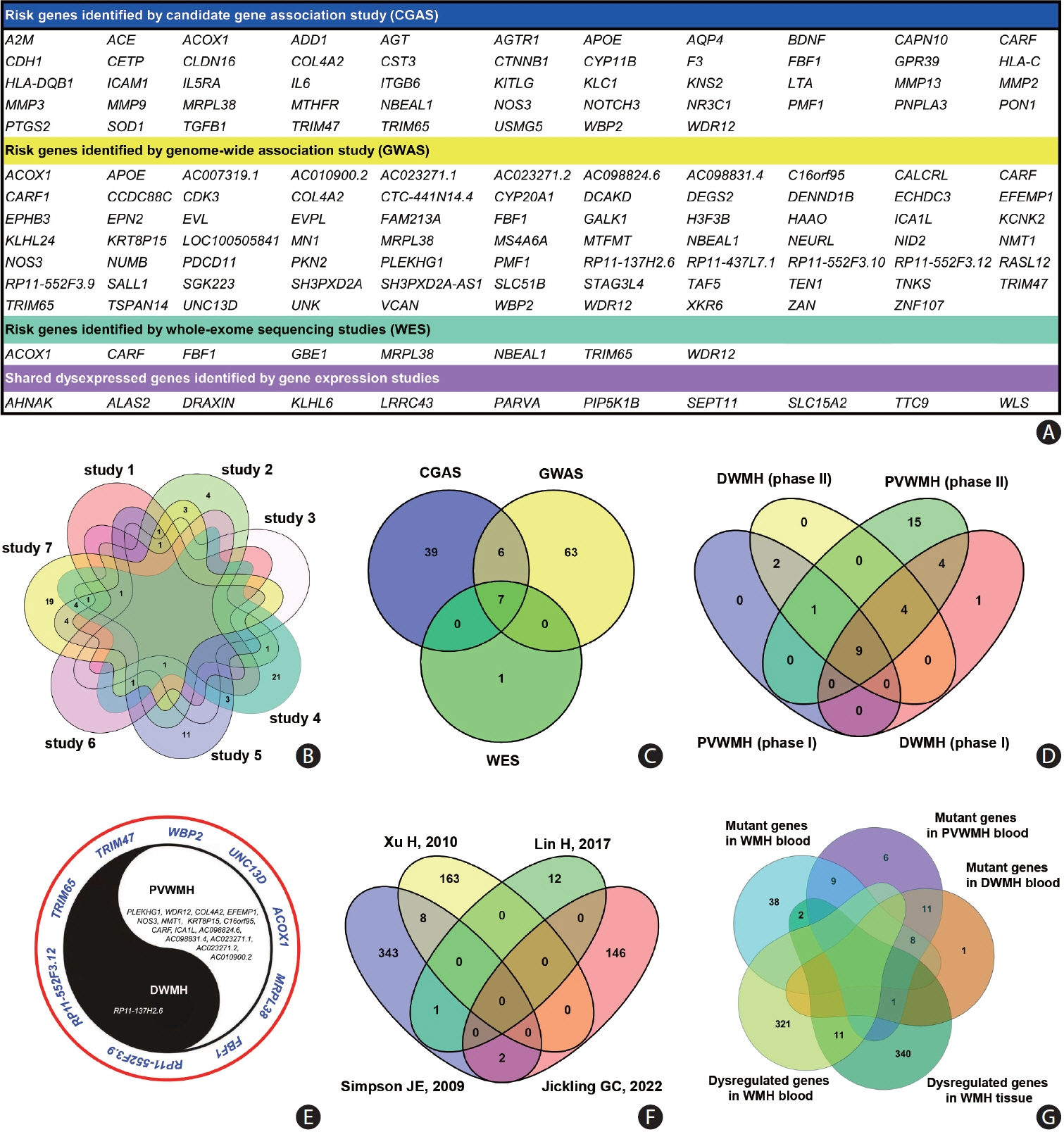
Article309. Su C, Wu H, Yang X, Zhao B, Zhao R. The relation between antihypertensive treatment and progression of cerebral small vessel disease: a systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore). 2021; 100:e26749.310. Heutz RA, Weijs RW, de Heus RA, Claassen JA. Antihypertensives in dementia: good or bad for the brain? J Cereb Blood Flow Metab. 2023; 43:1796–1799.311. Willmot M, Ghadami A, Whysall B, Clarke W, Wardlaw J, Bath PM. Transdermal glyceryl trinitrate lowers blood pressure and maintains cerebral blood flow in recent stroke. Hypertension. 2006; 47:1209–1215.
Article312. Sare GM, Gray LJ, Bath PM. Effect of antihypertensive agents on cerebral blood flow and flow velocity in acute ischaemic stroke: systematic review of controlled studies. J Hypertens. 2008; 26:1058–1064.
Article313. Kate M, Asdaghi N, Gioia LC, Buck B, Majumdar SR, Jeerakathil T, et al. Blood pressure reduction in hypertensive acute ischemic stroke patients does not affect cerebral blood flow. J Cereb Blood Flow Metab. 2019; 39:1878–1887.
Article314. Croall ID, Tozer DJ, Moynihan B, Khan U, O’Brien JT, Morris RG, et al. Effect of standard vs intensive blood pressure control on cerebral blood flow in small vessel disease: the PRESERVE randomized clinical trial. JAMA Neurol. 2018; 75:720–727.
Article315. de Heus RAA, de Jong DLK, Lawlor BL, Claassen JAHR; NILVAD Study Group. Longitudinal changes in the control mechanisms for blood pressure and cerebral blood flow in Alzheimer’s disease: secondary results of a randomized controlled trial. Cereb Circ Cogn Behav. 2021; 2:100024.
Article316. van Rijssel AE, Stins BC, Beishon LC, Sanders ML, Quinn TJ, Claassen JAHR, et al. Effect of antihypertensive treatment on cerebral blood flow in older adults: a systematic review and meta-analysis. Hypertension. 2022; 79:1067–1078.
Article317. Efimova NY, Chernov VI, Efimova IY, Lishmanov YB. Influence of antihypertensive therapy on cerebral perfusion in patients with metabolic syndrome: relationship with cognitive function and 24-h arterial blood pressure monitoring. Cardiovasc Ther. 2015; 33:209–215.
Article318. Dolui S, Detre JA, Gaussoin SA, Herrick JS, Wang DJJ, Tamura MK, et al. Association of intensive vs standard blood pressure control with cerebral blood flow: secondary analysis of the SPRINT MIND randomized clinical trial. JAMA Neurol. 2022; 79:380–389.
Article319. Ikeme JC, Pergola PE, Scherzer R, Shlipak MG, Catanese L, McClure LA, et al. Cerebral white matter hyperintensities, kidney function decline, and recurrent stroke after intensive blood pressure lowering: results from the secondary prevention of small subcortical strokes (SPS3) trial. J Am Heart Assoc. 2019; 8:e010091.
Article320. Williamson JD, Launer LJ, Bryan RN, Coker LH, Lazar RM, Gerstein HC, et al. Cognitive function and brain structure in persons with type 2 diabetes mellitus after intensive lowering of blood pressure and lipid levels: a randomized clinical trial. JAMA Intern Med. 2014; 174:324–333.
Article321. Verghese J, Lipton RB, Hall CB, Kuslansky G, Katz MJ. Low blood pressure and the risk of dementia in very old individuals. Neurology. 2003; 61:1667–1672.
Article322. McGrath ER, Beiser AS, DeCarli C, Plourde KL, Vasan RS, Greenberg SM, et al. Blood pressure from mid- to late life and risk of incident dementia. Neurology. 2017; 89:2447–2454.
Article323. Yeung A, Kiss A, Gallagher D. Intensive control of hypertension and risk of Alzheimer’s dementia in older adults with depression. Int J Geriatr Psychiatry. 2020; 35:888–896.
Article324. Jiang C, Lai Y, Du X, Wang Y, Li S, He L, et al. Effects of intensive blood pressure control on cardiovascular and cognitive outcomes in patients with atrial fibrillation: insights from the SPRINT trial. Europace. 2022; 24:1560–1568.
Article325. Poon IO. Effects of antihypertensive drug treatment on the risk of dementia and cognitive impairment. Pharmacotherapy. 2008; 28:366–375.
Article326. Hughes D, Judge C, Murphy R, Loughlin E, Costello M, Whiteley W, et al. Association of blood pressure lowering with incident dementia or cognitive impairment: a systematic review and meta-analysis. JAMA. 2029; 323:1934–1944.
Article327. Peters R, Xu Y, Fitzgerald O, Aung HL, Beckett N, Bulpitt C, et al. Blood pressure lowering and prevention of dementia: an individual patient data meta-analysis. Eur Heart J. 2022; 43:4980–4990.
Article328. Ding J, Davis-Plourde KL, Sedaghat S, Tully PJ, Wang W, Phillips C, et al. Antihypertensive medications and risk for incident dementia and Alzheimer’s disease: a meta-analysis of individual participant data from prospective cohort studies. Lancet Neurol. 2020; 19:61–70.
Article329. Lee CJ, Hwang J, Kang CY, Kim HC, Ryu DR, Ihm SH, et al. Protective effect of controlled blood pressure on risk of dementia in low-risk, grade 1 hypertension. J Hypertens. 2021; 39:1662–1669.
Article330. Canavan M, O’Donnell MJ. Hypertension and cognitive impairment: a review of mechanisms and key concepts. Front Neurol. 2022; 13:821135.331. Tully PJ, Dartigues JF, Debette S, Helmer C, Artero S, Tzourio C. Dementia risk with antihypertensive use and blood pressure variability: a cohort study. Neurology. 2016; 87:601–608.
Article332. Lennon MJ, Lam BCP, Lipnicki DM, Crawford JD, Peters R, Schutte AE, et al. Use of antihypertensives, blood pressure, and estimated risk of dementia in late life: an individual participant data meta-analysis. JAMA Netw Open. 2023; 6:e2333353.333. Jiang C, Li S, Wang Y, Lai Y, Bai Y, Zhao M, et al. Diastolic blood pressure and intensive blood pressure control on cognitive outcomes: insights from the SPRINT MIND trial. Hypertension. 2023; 80:580–589.
Article334. Peters R, Beckett N, Forette F, Tuomilehto J, Clarke R, Ritchie C, et al. Incident dementia and blood pressure lowering in the hypertension in the very elderly trial cognitive function assessment (HYVET-COG): a double-blind, placebo controlled trial. Lancet Neurol. 2008; 7:683–689.
Article335. van Middelaar T, van Vught LA, van Gool WA, Simons EMF, van den Born BH, Moll van Charante EP, et al. Blood pressurelowering interventions to prevent dementia: a systematic review and meta-analysis. J Hypertens. 2018; 36:1780–1787.336. SPRINT MIND Investigators for the SPRINT Research Group. Effect of intensive vs standard blood pressure control on probable dementia: a randomized clinical trial. JAMA. 2019; 321:553–561.337. Rojas-Saunero LP, Hilal S, Murray EJ, Logan RW, Ikram MA, Swanson SA. Hypothetical blood-pressure-lowering interventions and risk of stroke and dementia. Eur J Epidemiol. 2021; 36:69–79.
Article338. Kellar D, Lockhart SN, Aisen P, Raman R, Rissman RA, Brewer J, et al. Intranasal insulin reduces white matter hyperintensity progression in association with improvements in cognition and CSF biomarker profiles in mild cognitive impairment and Alzheimer’s disease. J Prev Alzheimers Dis. 2021; 8:240–248.
Article339. Inkeri J, Adeshara K, Harjutsalo V, Forsblom C, Liebkind R, Tatlisumak T, et al. Glycemic control is not related to cerebral small vessel disease in neurologically asymptomatic individuals with type 1 diabetes. Acta Diabetol. 2022; 59:481–490.
Article340. Livny A, Ravona-Springer R, Heymann A, Priess R, Kushnir T, Tsarfaty G, et al. Long-term variability in glycemic control is associated with white matter hyperintensities in APOE4 genotype carriers with type 2 diabetes. Diabetes Care. 2016; 39:1056–1059.
Article341. Launer LJ, Miller ME, Williamson JD, Lazar RM, Gerstein HC, Murray AM, et al. Effects of intensive glucose lowering on brain structure and function in people with type 2 diabetes (ACCORD MIND): a randomised open-label substudy. Lancet Neurol. 2011; 10:969–977.
Article342. Wardlaw JM, Debette S, Jokinen H, De Leeuw FE, Pantoni L, Chabriat H, et al. ESO guideline on covert cerebral small vessel disease. Eur Stroke J. 2021; 6:CXI–CLXII.
Article343. Mortensen MB, Falk E. Primary prevention with statins in the elderly. J Am Coll Cardiol. 2018; 71:85–94.344. Cholesterol Treatment Trialists’ Collaboration. Efficacy and safety of statin therapy in older people: a meta-analysis of individual participant data from 28 randomised controlled trials. Lancet. 2019; 393:407–415.345. Martínez-Sánchez P, Rivera-Ordóñez C, Fuentes B, Ortega-Casarrubios MA, Idrovo L, Díez-Tejedor E. The beneficial effect of statins treatment by stroke subtype. Eur J Neurol. 2009; 16:127–133.
Article346. Bernick C, Katz R, Smith NL, Rapp S, Bhadelia R, Carlson M, et al. Statins and cognitive function in the elderly: the cardiovascular health study. Neurology. 2005; 65:1388–1394.347. ten Dam VH, van den Heuvel DM, van Buchem MA, Westendorp RG, Bollen EL, Ford I, et al. Effect of pravastatin on cerebral infarcts and white matter lesions. Neurology. 2005; 64:1807–1809.
Article348. Ramanan VK, Przybelski SA, Graff-Radford J, Castillo AM, Lowe VJ, Mielke MM, et al. Statins and brain health: Alzheimer’s disease and cerebrovascular disease biomarkers in older adults. J Alzheimers Dis. 2018; 65:1345–1352.
Article349. Vogt NM, Hunt JFV, Ma Y, Van Hulle CA, Adluru N, Chappell RJ, et al. Effects of simvastatin on white matter integrity in healthy middle-aged adults. Ann Clin Transl Neurol. 2021; 8:1656–1667.
Article350. Mok VC, Lam WW, Fan YH, Wong A, Ng PW, Tsoi TH, et al. Effects of statins on the progression of cerebral white matter lesion: post hoc analysis of the ROCAS (regression of cerebral artery stenosis) study. J Neurol. 2009; 256:750–757.
Article351. Ji T, Zhao Y, Wang J, Cui Y, Duan D, Chai Q, et al. Effect of low-dose statins and apolipoprotein E genotype on cerebral small vessel disease in older hypertensive patients: a subgroup analysis of a randomized clinical trial. J Am Med Dir Assoc. 2018; 19:995–1002.e4.
Article352. Zhang H, Cui Y, Zhao Y, Dong Y, Duan D, Wang J, et al. Effects of sartans and low-dose statins on cerebral white matter hyperintensities and cognitive function in older patients with hypertension: a randomized, double-blind and placebocontrolled clinical trial. Hypertens Res. 2019; 42:717–729.
Article353. Guo Y, Li Y, Liu X, Cui Y, Zhao Y, Sun S, et al. Assessing the effectiveness of statin therapy for alleviating cerebral small vessel disease progression in people ≥75years of age. BMC Geriatr. 2020; 20:292.
Article354. Xue J, Wu Z, Gong S, Qin S, Gu A. High-dose atorvastatin improves vascular endothelial function in patients with leukoaraiosis. J Clin Lab Anal. 2020; 34:e23081.
Article355. Sterzer P, Meintzschel F, Rösler A, Lanfermann H, Steinmetz H, Sitzer M. Pravastatin improves cerebral vasomotor reactivity in patients with subcortical small-vessel disease. Stroke. 2001; 32:2817–2820.
Article356. Ii M, Losordo DW. Statins and the endothelium. Vascul Pharmacol. 2007; 46:1–9.
Article357. Liu Z, Zhao Y, Wei F, Ye L, Lu F, Zhang H, et al. Treatment with telmisartan/rosuvastatin combination has a beneficial synergistic effect on ameliorating Th17/Treg functional imbalance in hypertensive patients with carotid atherosclerosis. Atherosclerosis. 2014; 233:291–299.
Article358. Janic´ M, Lunder M, Šabovicˇ M. A low-dose combination of fluvastatin and valsartan: a new “drug” and a new approach for decreasing the arterial age. Biomed Res Int. 2015; 2015:235709.359. Rizos CV, Liberopoulos EN, Tellis CC, Tselepis AD, Elisaf MS. The effect of combining rosuvastatin with sartans of different peroxisome proliferator receptor-γ activating capacity on plasma 8-isoprostane prostaglandin F2a levels. Arch Med Sci. 2013; 9:172–176.360. Kim JS. Role of blood lipid levels and lipid-lowering therapy in stroke patients with different levels of cerebral artery diseases: reconsidering recent stroke guidelines. J Stroke. 2021; 23:149–161.
Article361. Collins R, Armitage J, Parish S, Sleight P, Peto R; Heart Protection Study Collaborative Group. Effects of cholesterollowering with simvastatin on stroke and other major vascular events in 20536 people with cerebrovascular disease or other high-risk conditions. Lancet. 2004; 363:757–767.
Article362. Goldstein LB, Amarenco P, Szarek M, Callahan A 3rd, Hennerici M, Sillesen H, et al. Hemorrhagic stroke in the stroke prevention by aggressive reduction in cholesterol levels study. Neurology. 2008; 70(24 Pt 2):2364–2370.363. Hosomi N, Nagai Y, Kohriyama T, Ohtsuki T, Aoki S, Nezu T, et al. The Japan statin treatment against recurrent stroke (JSTARS): a multicenter, randomized, open-label, parallel-group study. EBioMedicine. 2015; 2:1071–1078.
Article364. Hosomi N, Kitagawa K, Nagai Y, Nakagawa Y, Aoki S, Nezu T, et al. Different influences of statin treatment in preventing at-risk stroke subtypes: a post hoc analysis of J-STARS. J Atheroscler Thromb. 2020; 27:449–460.
Article365. Boxer AL, Kramer JH, Johnston K, Goldman J, Finley R, Miller BL. Executive dysfunction in hyperhomocystinemia responds to homocysteine-lowering treatment. Neurology. 2005; 64:1431–1434.
Article366. Cavalieri M, Schmidt R, Chen C, Mok V, de Freitas GR, Song S, et al. B vitamins and magnetic resonance imaging-detected ischemic brain lesions in patients with recent transient ischemic attack or stroke: the VITAmins TO Prevent Stroke (VITATOPS) MRI-substudy. Stroke. 2012; 43:3266–3270.
Article367. Fassbender K, Mielke O, Bertsch T, Nafe B, Fröschen S, Hennerici M. Homocysteine in cerebral macroangiography and microangiopathy. Lancet. 1999; 353:1586–1587.368. Weir DG, Scott JM. Brain function in the elderly: role of vitamin B12 and folate. Br Med Bull. 1999; 55:669–682.
Article369. Park HK, Kob SB, Jung KH, Jang MU, Kim DH, Kim JT, et al. 2022 Update of the Korean clinical practice guidelines for stroke: antithrombotic therapy for patients with acute ischemic stroke or transient ischemic attack. J Stroke. 2022; 24:166–175.
Article370. Ishida K, Messé SR. Antiplatelet strategies for secondary prevention of stroke and TIA. Curr Atheroscler Rep. 2014; 16:449.
Article371. Ter Telgte A, van Leijsen EMC, Wiegertjes K, Klijn CJM, Tuladhar AM, de Leeuw FE. Cerebral small vessel disease: from a focal to a global perspective. Nat Rev Neurol. 2018; 14:387–398.
Article372. Huang N, Chen D, Wu X, Chen X, Zhang X, Niu J, et al. Aspirin promotes oligodendroglial differentiation through inhibition of Wnt signaling pathway. Mol Neurobiol. 2016; 53:3258–3266.373. Chen J, Zuo S, Wang J, Huang J, Zhang X, Liu Y, et al. Aspirin promotes oligodendrocyte precursor cell proliferation and differentiation after white matter lesion. Front Aging Neurosci. 2014; 6:7.
Article374. Holcombe A, Ammann E, Espeland MA, Kelley BJ, Manson JE, Wallace R, et al. Chronic use of aspirin and total white matter lesion volume: results from the women’s health initiative memory study of magnetic resonance imaging study. J Stroke Cerebrovasc Dis. 2017; 26:2128–2136.
Article375. Ward SA, Raniga P, Ferris NJ, Woods RL, Storey E, Bailey MJ, et al. ASPREE-NEURO study protocol: a randomized controlled trial to determine the effect of low-dose aspirin on cerebral microbleeds, white matter hyperintensities, cognition, and stroke in the healthy elderly. Int J Stroke. 2017; 12:108–113.
Article376. Zheng SL, Roddick AJ. Association of aspirin use for primary prevention with cardiovascular events and bleeding events: a systematic review and meta-analysis. JAMA. 2019; 321:277–287.
Article377. Shinohara Y, Katayama Y, Uchiyama S, Yamaguchi T, Handa S, Matsuoka K, et al. Cilostazol for prevention of secondary stroke (CSPS 2): an aspirin-controlled, double-blind, randomised noninferiority trial. Lancet Neurol. 2010; 9:959–968.
Article378. Lin MP, Meschia JF, Gopal N, Barrett KM, Ross OA, Ertekin-Taner N, et al. Cilostazol versus aspirin for secondary stroke prevention: systematic review and meta-analysis. J Stroke Cerebrovasc Dis. 2021; 30:105581.
Article379. Han HJ, Kim BC, Youn YC, Jeong JH, Kim JH, Lee JH, et al. A comparison study of cilostazol and aspirin on changes in volume of cerebral small vessel disease white matter changes: protocol of a multicenter, randomized controlled trial. Dement Neurocogn Disord. 2019; 18:138–148.
Article380. Kim BC, Youn YC, Jeong JH, Han HJ, Kim JH, Lee JH, et al. Cilostazol versus aspirin on white matter changes in cerebral small vessel disease: a randomized controlled trial. Stroke. 2022; 53:698–709.
Article381. Ip BYM, Lam BYK, Hui VMH, Au LWC, Liu MWT, Shi L, et al. Efficacy and safety of cilostazol in decreasing progression of cerebral white matter hyperintensities—a randomized controlled trial. Alzheimers Dement (N Y). 2022; 8:e12369.
Article382. Gons RA, van Norden AG, de Laat KF, van Oudheusden LJ, van Uden IW, Zwiers MP, et al. Cigarette smoking is associated with reduced microstructural integrity of cerebral white matter. Brain. 2011; 134(Pt 7):2116–2124.383. Akbar Z, Fituri S, Ouagueni A, Alalwani J, Sukik A, Al-Jayyousi GF, et al. Associations of the MIND diet with cardiometabolic diseases and their risk factors: a systematic review. Diabetes Metab Syndr Obes. 2023; 16:3353–3371.
Article384. Makin SDJ, Mubki GF, Doubal FN, Shuler K, Staals J, Dennis MS, et al. Small vessel disease and dietary salt intake: cross-sectional study and systematic review. J Stroke Cerebrovasc Dis. 2017; 26:3020–3028.
Article385. Strazzullo P, D’Elia L, Kandala NB, Cappuccio FP. Salt intake, stroke, and cardiovascular disease: meta-analysis of prospective studies. BMJ. 2009; 339:b4567.
Article386. Del Brutto OH, Recalde BY, Mera RM. Dietary oily fish intake is inversely associated with severity of white matter hyperintensities of presumed vascular origin. A population-based study in frequent fish consumers of Amerindian ancestry. J Stroke Cerebrovasc Dis. 2021; 30:105778.
Article387. Song S, Gaynor AM, Cruz E, Lee S, Gazes Y, Habeck C, et al. Mediterranean diet and white matter hyperintensity change over time in cognitively intact adults. Nutrients. 2022; 14:3664.
Article388. Barnes LL, Dhana K, Liu X, Carey VJ, Ventrelle J, Johnson K, et al. Trial of the MIND diet for prevention of cognitive decline in older persons. N Engl J Med. 2023; 389:602–611.
Article389. Venkatraman VK, Sanderson A, Cox KL, Ellis KA, Steward C, Phal PM, et al. Effect of a 24-month physical activity program on brain changes in older adults at risk of Alzheimer’s disease: the AIBL active trial. Neurobiol Aging. 2020; 89:132–141.
Article390. Ho AJ, Raji CA, Becker JT, Lopez OL, Kuller LH, Hua X, et al. The effects of physical activity, education, and body mass index on the aging brain. Hum Brain Mapp. 2011; 32:1371–1382.
Article391. Rosano C, Venkatraman VK, Guralnik J, Newman AB, Glynn NW, Launer L, et al. Psychomotor speed and functional brain MRI 2 years after completing a physical activity treatment. J Gerontol A Biol Sci Med Sci. 2010; 65:639–647.
Article392. Willey JZ, Moon YP, Paik MC, Yoshita M, Decarli C, Sacco RL, et al. Lower prevalence of silent brain infarcts in the physically active: the northern Manhattan study. Neurology. 2011; 76:2112–2118.
Article393. Smith JA, Turner ST, Sun YV, Fornage M, Kelly RJ, Mosley TH, et al. Complexity in the genetic architecture of leukoaraiosis in hypertensive sibships from the GENOA study. BMC Med Genomics. 2009; 2:16.
Article394. Sen A, Gider P, Cavalieri M, Freudenberger P, Farzi A, Schallert M, et al. Association of cardiorespiratory fitness and morphological brain changes in the elderly: results of the Austrian stroke prevention study. Neurodegener Dis. 2012; 10:135–137.
Article395. Tseng BY, Gundapuneedi T, Khan MA, Diaz-Arrastia R, Levine BD, Lu H, et al. White matter integrity in physically fit older adults. Neuroimage. 2013; 82:510–516.
Article396. Gow AJ, Bastin ME, Muñoz Maniega S, Valdés Hernández MC, Morris Z, Murray C, et al. Neuroprotective lifestyles and the aging brain: activity, atrophy, and white matter integrity. Neurology. 2012; 79:1802–1808.397. Torres ER, Strack EF, Fernandez CE, Tumey TA, Hitchcock ME. Physical activity and white matter hyperintensities: a systematic review of quantitative studies. Prev Med Rep. 2015; 2:319–325.
Article398. Podewils LJ, Guallar E, Beauchamp N, Lyketsos CG, Kuller LH, Scheltens P. Physical activity and white matter lesion progression: assessment using MRI. Neurology. 2007; 68:1223–1226.
Article399. Pan Y, Shen J, Cai X, Chen H, Zong G, Zhu W, et al. Adherence to a healthy lifestyle and brain structural imaging markers. Eur J Epidemiol. 2023; 38:657–668.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Leukoaraiosis on the Severity and Course of Delirium
- Comparison of White Matter Lesions on the Magnetic Resonance Imaging in Major Depression and Vascular Depression
- Silent Microbleeds and Old Hematomas in Spontaneous Cerebral Hemorrhages
- Asymptomatic Cerebral Small Vessel Disease: Insights from Population-Based Studies
- Cerebral White Matter Lesions and Apolipoprotein E polymorphism