J Korean Med Sci.
2024 Feb;39(7):e79. 10.3346/jkms.2024.39.e79.
Differences in Brain Metabolite Profiles Between Normothermia and Hypothermia
- Affiliations
-
- 1Department of Thoracic and Cardiovascular Surgery, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea
- 2Department of Clinical Pharmacology and Therapeutics, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea
- 3Department of Biomedical Sciences, Seoul National University College of Medicine, Seoul, Korea
- 4Department of Anesthesiology and Pain Medicine, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea
- KMID: 2553319
- DOI: http://doi.org/10.3346/jkms.2024.39.e79
Abstract
- Background
This study evaluated the difference in brain metabolite profiles between normothermia and hypothermia reaching 25°C in humans in vivo.
Methods
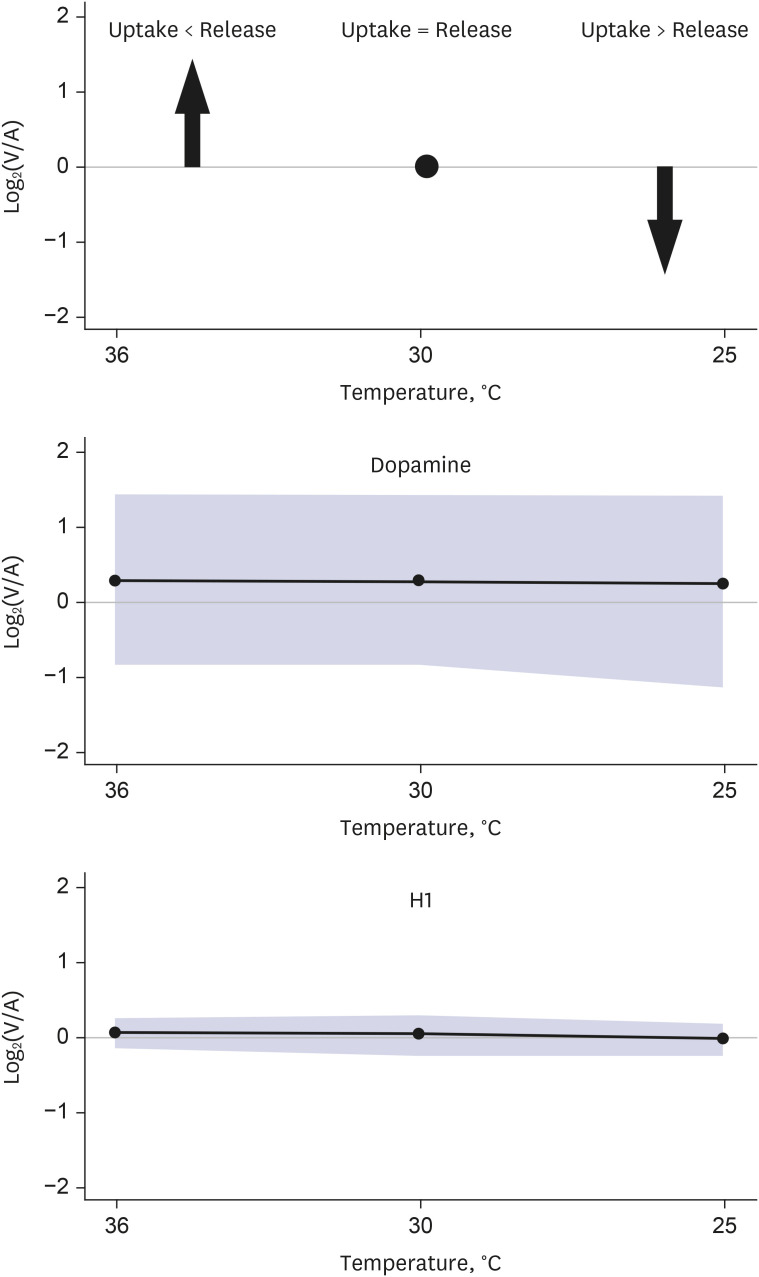
Thirteen patients who underwent thoracic aorta surgery under moderate hypothermia were prospectively enrolled. Plasma samples were collected simultaneously from the arteries and veins to estimate metabolite uptake or release. Targeted metabolomics based on liquid chromatographic mass spectrometry and direct flow injection were performed, and changes in the profiles of respective metabolites from normothermia to hypothermia were compared. The ratios of metabolite concentrations in venous blood samples to those in arterial blood samples (V/A ratios) were calculated, and log 2 transformation of the ratios [log2 (V/A)] was performed for comparison between the temperature groups.
Results
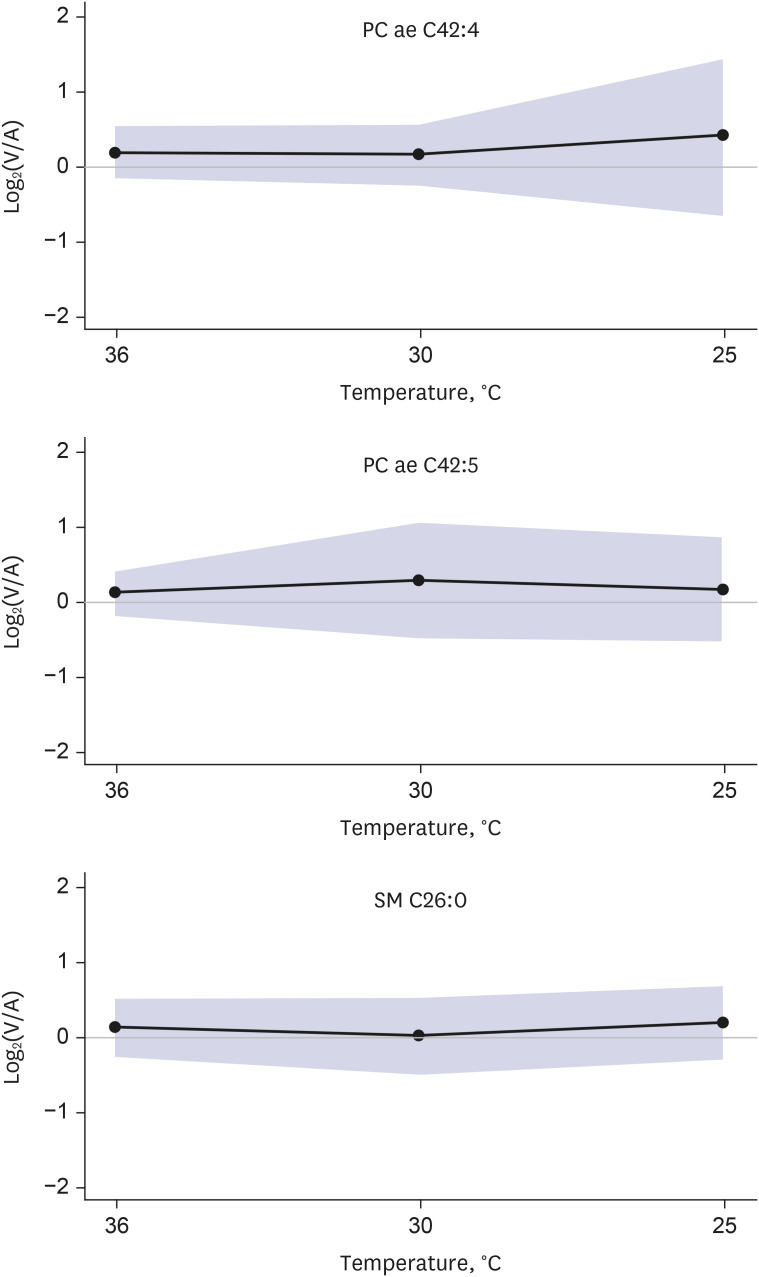
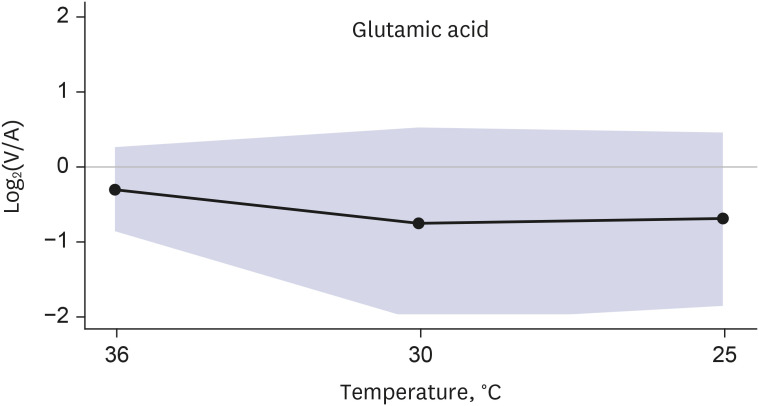
Targeted metabolomics were performed for 140 metabolites, including 20 amino acids, 13 biogenic amines, 10 acylcarnitines, 82 glycerophospholipids, 14 sphingomyelins, and 1 hexose. Of the 140 metabolites analyzed, 137 metabolites were released from the brain in normothermia, and the release of 132 of these 137 metabolites was decreased in hypothermia. Two metabolites (dopamine and hexose) showed constant release from the brain in hypothermia, and 3 metabolites (2 glycophospholipids and 1 sphingomyelin) showed conversion from release to uptake in hypothermia. Glutamic acid demonstrated a distinct brain metabolism in that it was taken up by the brain in normothermia, and the uptake was increased in hypothermia.
Conclusion
Targeted metabolomics demonstrated various degrees of changes in the release of metabolites by the hypothermic brain. The release of most metabolites was decreased in hypothermia, whereas glutamic acid showed a distinct brain metabolism.
Keyword
Figure
Reference
-
1. Liu BH, Pu J, Li ZQ, Zhang XR. The effects of hypothermia on glutamate and γ-aminobutyric acid metabolism during ischemia in monkeys: a repeated-measures ANOVA study. Sci Rep. 2022; 12(1):14470. PMID: 36008544.2. Mink JW, Blumenschine RJ, Adams DB. Ratio of central nervous system to body metabolism in vertebrates: its constancy and functional basis. Am J Physiol. 1981; 241(3):R203–R212. PMID: 7282965.3. Walker HK, Hall WD, Hurst JW. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd Edition. Boston, MA, USA: Butterworths;1990.4. Zhang HH, Wang L, Zhang W, Wan Z. The role of sphingomyelin metabolism in the protection of rat brain microvascular endothelial cells by mild hypothermia. Neurocrit Care. 2022; 36(2):546–559. PMID: 34508278.5. Marion DW, Penrod LE, Kelsey SF, Obrist WD, Kochanek PM, Palmer AM, et al. Treatment of traumatic brain injury with moderate hypothermia. N Engl J Med. 1997; 336(8):540–546. PMID: 9023090.6. Jiang J, Yu M, Zhu C. Effect of long-term mild hypothermia therapy in patients with severe traumatic brain injury: 1-year follow-up review of 87 cases. J Neurosurg. 2000; 93(4):546–549. PMID: 11014530.7. McIntyre LA, Fergusson DA, Hébert PC, Moher D, Hutchison JS. Prolonged therapeutic hypothermia after traumatic brain injury in adults: a systematic review. JAMA. 2003; 289(22):2992–2999. PMID: 12799408.8. Tokutomi T, Miyagi T, Takeuchi Y, Karukaya T, Katsuki H, Shigemori M. Effect of 35 degrees C hypothermia on intracranial pressure and clinical outcome in patients with severe traumatic brain injury. J Trauma. 2009; 66(1):166–173. PMID: 19131820.9. Hagl C, Ergin MA, Galla JD, Lansman SL, McCullough JN, Spielvogel D, et al. Neurologic outcome after ascending aorta-aortic arch operations: effect of brain protection technique in high-risk patients. J Thorac Cardiovasc Surg. 2001; 121(6):1107–1121. PMID: 11385378.10. Okita Y, Minatoya K, Tagusari O, Ando M, Nagatsuka K, Kitamura S. Prospective comparative study of brain protection in total aortic arch replacement: deep hypothermic circulatory arrest with retrograde cerebral perfusion or selective antegrade cerebral perfusion. Ann Thorac Surg. 2001; 72(1):72–79. PMID: 11465234.11. Griepp RB. Cerebral protection during aortic arch surgery. J Thorac Cardiovasc Surg. 2001; 121(3):425–427. PMID: 11241074.12. Jang C, Hui S, Zeng X, Cowan AJ, Wang L, Chen L, et al. Metabolite exchange between mammalian organs quantified in pigs. Cell Metab. 2019; 30(3):594–606.e3. PMID: 31257152.13. Xia J, Wishart DS. Using metaboanalyst 3.0 for comprehensive metabolomics data analysis. Curr Protoc Bioinformatics. 2016; 55:14.10.1–14.10.91.14. Patti GJ, Yanes O, Siuzdak G. Innovation: metabolomics: the apogee of the omics trilogy. Nat Rev Mol Cell Biol. 2012; 13(4):263–269. PMID: 22436749.15. Pacini D, Di Marco L, Leone A, Tonon C, Pettinato C, Fonti C, et al. Cerebral functions and metabolism after antegrade selective cerebral perfusion in aortic arch surgery. Eur J Cardiothorac Surg. 2010; 37(6):1322–1331. PMID: 20138537.16. Peng J, Zeng J, Cai B, Yang H, Cohen MJ, Chen W, et al. Establishment of quantitative severity evaluation model for spinal cord injury by metabolomic fingerprinting. PLoS One. 2014; 9(4):e93736. PMID: 24727691.17. Cohen MJ, Serkova NJ, Wiener-Kronish J, Pittet JF, Niemann CU. 1H-NMR-based metabolic signatures of clinical outcomes in trauma patients--beyond lactate and base deficit. J Trauma. 2010; 69(1):31–40. PMID: 20622576.18. Lexcen DR, Lusczek ER, Witowski NE, Mulier KE, Beilman GJ. Metabolomics classifies phase of care and identifies risk for mortality in a porcine model of multiple injuries and hemorrhagic shock. J Trauma Acute Care Surg. 2012; 73(2):Suppl 1. S147–S155. PMID: 22847086.19. Feng JZ, Wang WY, Zeng J, Zhou ZY, Peng J, Yang H, et al. Optimization of brain metabolism using metabolic-targeted therapeutic hypothermia can reduce mortality from traumatic brain injury. J Trauma Acute Care Surg. 2017; 83(2):296–304. PMID: 28452885.20. Cantarow A, Ricchiuti G. Epinephrine hyperglycemia: with particular reference to the arteriovenous blood sugar difference in hepatic disease. J Clin Invest. 1934; 13(3):461–478. PMID: 16694222.21. Hickam JB, Cargill WH, Golden A. Cardiovascular reactions to emotional stimuli. effect on the cardiac output, arteriovenous oxygen difference, arterial pressure, and peripheral resistance. J Clin Invest. 1948; 27(2):290–298. PMID: 16695554.22. Ivanisevic J, Elias D, Deguchi H, Averell PM, Kurczy M, Johnson CH, et al. Arteriovenous blood metabolomics: a readout of intra-tissue metabostasis. Sci Rep. 2015; 5(1):12757. PMID: 26244428.23. Desfeux A, El Ghazi F, Jégou S, Legros H, Marret S, Laudenbach V, et al. Dual effect of glutamate on GABAergic interneuron survival during cerebral cortex development in mice neonates. Cereb Cortex. 2010; 20(5):1092–1108. PMID: 19759125.24. Bak LK, Schousboe A, Waagepetersen HS. The glutamate/GABA-glutamine cycle: aspects of transport, neurotransmitter homeostasis and ammonia transfer. J Neurochem. 2006; 98(3):641–653. PMID: 16787421.25. Tiwari V, Ambadipudi S, Patel AB. Glutamatergic and GABAergic TCA cycle and neurotransmitter cycling fluxes in different regions of mouse brain. J Cereb Blood Flow Metab. 2013; 33(10):1523–1531. PMID: 23838829.26. Karki P, Hong P, Johnson J Jr, Pajarillo E, Son DS, Aschner M, et al. Arundic acid increases expression and function of astrocytic glutamate transporter EAAT1 via the ERK, Akt, and NF-κB pathways. Mol Neurobiol. 2018; 55(6):5031–5046. PMID: 28812276.27. Shigeri Y, Seal RP, Shimamoto K. Molecular pharmacology of glutamate transporters, EAATs and VGLUTs. Brain Res Brain Res Rev. 2004; 45(3):250–265. PMID: 15210307.28. Olney JW, Adamo NJ, Ratner A. Monosodium glutamate effects. Science. 1971; 172(3980):294. PMID: 5102256.29. Olney JW. Glutamate-induced neuronal necrosis in the infant mouse hypothalamus. An electron microscopic study. J Neuropathol Exp Neurol. 1971; 30(1):75–90. PMID: 5542543.30. Andiné P, Sandberg M, Bågenholm R, Lehmann A, Hagberg H. Intra- and extracellular changes of amino acids in the cerebral cortex of the neonatal rat during hypoxic-ischemia. Brain Res Dev Brain Res. 1991; 64(1-2):115–120. PMID: 1786636.31. Ma D, An Q, Zhang Z, Bian Q, Li Y, Li Y, et al. Head mild hypothermia exerts a neuroprotective role in ischemia-reperfusion injury by maintaining glial glutamate transporter 1. Ther Hypothermia Temp Manag. 2021; 11(3):155–163. PMID: 32985953.32. Wang D, Zhao Y, Zhang Y, Zhang T, Shang X, Wang J, et al. Hypothermia protects against oxygen-glucose deprivation-induced neuronal injury by down-regulating the reverse transport of glutamate by astrocytes as mediated by neurons. Neuroscience. 2013; 237:130–138. PMID: 23402854.33. Ghosh M, Yang Y, Rothstein JD, Robinson MB. Nuclear factor-κB contributes to neuron-dependent induction of glutamate transporter-1 expression in astrocytes. J Neurosci. 2011; 31(25):9159–9169. PMID: 21697367.34. Karnatovskaia LV, Wartenberg KE, Freeman WD. Therapeutic hypothermia for neuroprotection: history, mechanisms, risks, and clinical applications. Neurohospitalist. 2014; 4(3):153–163. PMID: 24982721.35. Yan TD, Bannon PG, Bavaria J, Coselli JS, Elefteriades JA, Griepp RB, et al. Consensus on hypothermia in aortic arch surgery. Ann Cardiothorac Surg. 2013; 2(2):163–168. PMID: 23977577.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Temperature Difference between Brain and Axilla according to Body Temperature in the Patient with Brain Injury
- The Effect of Regional Hypothermia of Neuromuscular Transmission
- Temperature Difference between the Brain and the Axilla according to the Patients Body Temperature
- Effect of Brain Hypothermia on Ischemic Injury and Matrix Metalloproteinase Activity in Focal Cerebral Ischemia
- Effect of Therapeutic Hypothermia on Mitogen-activated Protein Kinase Pathways in the Brain Tissue of a Swine Cardiac Arrest Model