J Korean Med Sci.
2024 Jan;39(1):e13. 10.3346/jkms.2024.39.e13.
S100 Calcium-Binding Protein A9, a Potential Novel Diagnostic Biomarker for Idiopathic Pulmonary Fibrosis
- Affiliations
-
- 1Department of Interdisciplinary Program in Biomedical Science Major, Soonchunhyang Graduate School, Bucheon, Korea
- 2Division of Allergy and Respiratory Disease, Soonchunhyang University Gumi Hospital, Gumi, Korea
- 3Department of Microbiology and BK21 Four Project, College of Medicine, Soonchunhyang University, Cheonan, Korea
- 4Genome Research Center and Division of Allergy and Respiratory Medicine, Soonchunhyang University Bucheon Hospital, Bucheon, Korea
- KMID: 2550787
- DOI: http://doi.org/10.3346/jkms.2024.39.e13
Abstract
- Background
Neutrophilic inflammation is a characteristic feature of idiopathic pulmonary fibrosis (IPF). S100 calcium-binding protein A9 (S100A9) is a neutrophil-derived protein involved in the development of neutrophil-related chronic inflammatory disorders. However, the role of S100A9 in IPF remains unclear.
Methods
We used enzyme-linked immunosorbent assays to measure S100A9 levels in bronchoalveolar lavage fluid (BALF) and serum obtained from healthy controls (HCs) and patients with IPF, non-specific interstitial pneumonia, hypersensitivity pneumonitis, and sarcoidosis.
Results
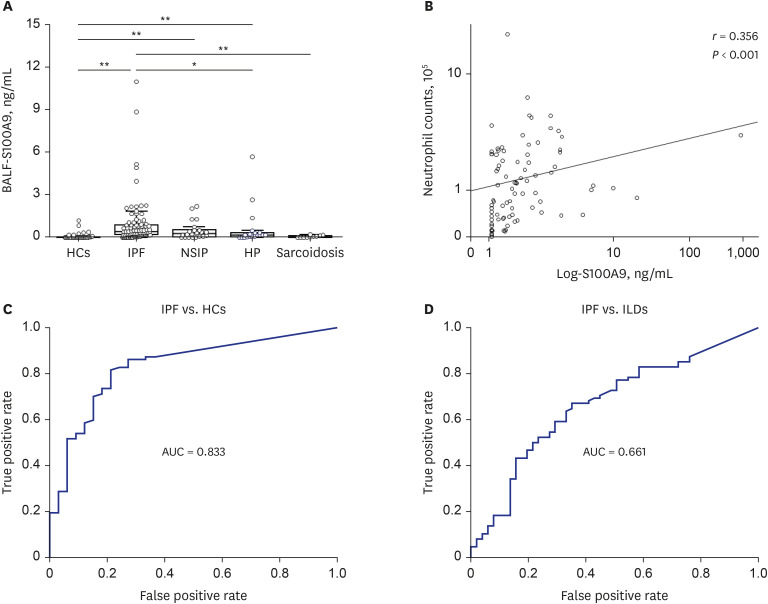
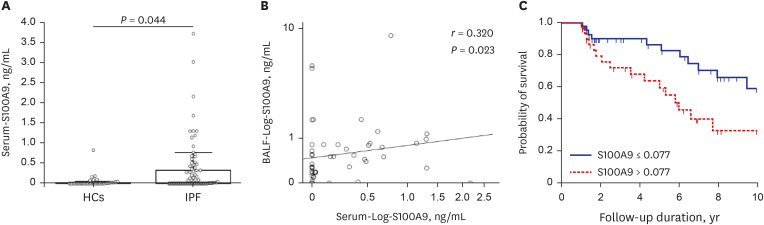
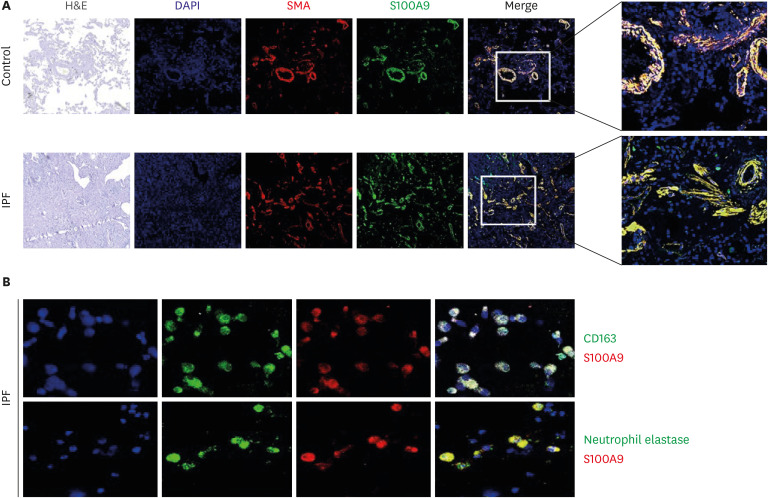
Compared with HCs, BALF S100A9 levels were significantly higher in IPF patients (P < 0.001), patients with hypersensitivity pneumonitis (P = 0.043), and patients with nonspecific interstitial pneumonia (P < 0.001). The S100A9 level in BALF of 0.093 ng/mL could distinguish IPF patients from HCs, with a specificity of 78.8% and a sensitivity of 81.6%. Similarly, the S100A9 level in BALF of 0.239 ng/mL had a specificity of 64.7% and a sensitivity of 66.7% for distinguishing IPF patients from patients with other interstitial lung diseases. Additionally, BALF S100A9 levels were significantly correlated with neutrophil counts (r = 0.356, P < 0.001) in BALF. IPF patients with S100A9 levels in BALF > 0.533 ng/ mL had lower survival rates, compared with patients who had levels ≤ 0.553 ng/mL (n = 49; hazard ratio [HR], 3.62; P = 0.021). Combination analysis revealed that IPF patients with S100A9 levels in BALF> 0.553 ng/mL or neutrophil percentages > 49.1% (n = 43) had significantly lower survival rates than patients with S100A9 levels in BALF ≤ 0.553 ng/mL and neutrophil percentages ≤ 49.1% (n = 41) (HR, 3.91; P = 0.014). Additionally, patients with serum S100A9 levels > 0.077 ng/mL (n = 29) had significantly lower survival rates than patients with levels ≤ 0.077 ng/mL (n = 53, HR, 2.52; P = 0.013). S100A9 was expressed on neutrophils and macrophages in BALF from IPF patients as well as α-smooth muscle actin positive cells in the lung tissues.
Conclusion
S100A9 is involved in the development and progression of IPF. Moreover, S100A9 levels in BALF and serum may be surrogate markers for IPF diagnosis and survival prediction, particularly when analyzed in combination with neutrophil percentages.
Figure
Reference
-
1. Chambers RC, Mercer PF. Mechanisms of alveolar epithelial injury, repair, and fibrosis. Ann Am Thorac Soc. 2015; 12(Suppl 1):S16–S20. PMID: 25830828.2. Richeldi L, Collard HR, Jones MG. Idiopathic pulmonary fibrosis. Lancet. 2017; 389(10082):1941–1952. PMID: 28365056.3. Moss BJ, Ryter SW, Rosas IO. Pathogenic mechanisms underlying idiopathic pulmonary fibrosis. Annu Rev Pathol. 2022; 17(1):515–546. PMID: 34813355.4. Desai O, Winkler J, Minasyan M, Herzog EL. The role of immune and inflammatory cells in idiopathic pulmonary fibrosis. Front Med (Lausanne). 2018; 5:43–43. PMID: 29616220.5. Giannandrea M, Parks WC. Diverse functions of matrix metalloproteinases during fibrosis. Dis Model Mech. 2014; 7(2):193–203. PMID: 24713275.6. Obayashi Y, Yamadori I, Fujita J, Yoshinouchi T, Ueda N, Takahara J. The role of neutrophils in the pathogenesis of idiopathic pulmonary fibrosis. Chest. 1997; 112(5):1338–1343. PMID: 9367478.7. Gregory AD, Kliment CR, Metz HE, Kim KH, Kargl J, Agostini BA, et al. Neutrophil elastase promotes myofibroblast differentiation in lung fibrosis. J Leukoc Biol. 2015; 98(2):143–152. PMID: 25743626.8. Inage M, Nakamura H, Kato S, Saito H, Abe S, Hino T, et al. Levels of cytokeratin 19 fragments in bronchoalveolar lavage fluid correlate to the intensity of neutrophil and eosinophil-alveolitis in patients with idiopathic pulmonary fibrosis. Respir Med. 2000; 94(2):155–160. PMID: 10714422.9. Lee JU, Choi JS, Kim MK, Min SA, Park JS, Park CS. Granulocyte colony-stimulating factor in bronchoalveolar lavage fluid is a potential biomarker for prognostic prediction of idiopathic pulmonary fibrosis. Korean J Intern Med. 2022; 37(5):979–988. PMID: 35730133.10. Jegal Y. The role of neutrophils in the pathogenesis of IPF. Korean J Intern Med. 2022; 37(5):945–946. PMID: 36068714.11. Kinder BW, Brown KK, Schwarz MI, Ix JH, Kervitsky A, King TE Jr. Baseline BAL neutrophilia predicts early mortality in idiopathic pulmonary fibrosis. Chest. 2008; 133(1):226–232. PMID: 18071016.12. Tabuena RP, Nagai S, Tsutsumi T, Handa T, Minoru T, Mikuniya T, et al. Cell profiles of bronchoalveolar lavage fluid as prognosticators of idiopathic pulmonary fibrosis/usual interstitial pneumonia among Japanese Patients. Respiration. 2005; 72(5):490–498. PMID: 16210888.13. Foell D, Wittkowski H, Vogl T, Roth J. S100 proteins expressed in phagocytes: a novel group of damage-associated molecular pattern molecules. J Leukoc Biol. 2007; 81(1):28–37. PMID: 16943388.14. Shibata F, Miyama K, Shinoda F, Mizumoto J, Takano K, Nakagawa H. Fibroblast growth-stimulating activity of S100A9 (MRP-14). Eur J Biochem. 2004; 271(11):2137–2143. PMID: 15153104.15. Xu X, Chen H, Zhu X, Ma Y, Liu Q, Xue Y, et al. S100A9 promotes human lung fibroblast cells activation through receptor for advanced glycation end-product-mediated extracellular-regulated kinase 1/2, mitogen-activated protein-kinase and nuclear factor-κB-dependent pathways. Clin Exp Immunol. 2013; 173(3):523–535. PMID: 23682982.16. Bargagli E, Olivieri C, Prasse A, Bianchi N, Magi B, Cianti R, et al. Calgranulin B (S100A9) levels in bronchoalveolar lavage fluid of patients with interstitial lung diseases. Inflammation. 2008; 31(5):351–354. PMID: 18784990.17. Bargagli E, Olivieri C, Cintorino M, Refini RM, Bianchi N, Prasse A, et al. Calgranulin B (S100A9/MRP14): a key molecule in idiopathic pulmonary fibrosis? Inflammation. 2011; 34(2):85–91. PMID: 20422274.18. Raghu G, Collard HR, Egan JJ, Martinez FJ, Behr J, Brown KK, et al. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med. 2011; 183(6):788–824. PMID: 21471066.19. Raghu G, Remy-Jardin M, Myers JL, Richeldi L, Ryerson CJ, Lederer DJ, et al. Diagnosis of idiopathic pulmonary fibrosis. an official ATS/ERS/JRS/ALAT clinical practice guideline. Am J Respir Crit Care Med. 2018; 198(5):e44–e68. PMID: 30168753.20. Travis WD, Costabel U, Hansell DM, King TE Jr, Lynch DA, Nicholson AG, et al. An official American Thoracic Society/European Respiratory Society statement: Update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am J Respir Crit Care Med. 2013; 188(6):733–748. PMID: 24032382.21. Erratum: diagnosis of hypersensitivity pneumonitis in adults: an official ATS/JRS/ALAT clinical practice guideline. Am J Respir Crit Care Med. 2021; 203(1):150–151.22. Crouser ED, Maier LA, Wilson KC, Bonham CA, Morgenthau AS, Patterson KC, et al. Diagnosis and detection of sarcoidosis. an official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020; 201(8):e26–e51. PMID: 32293205.23. Park SW, Ahn MH, Jang HK, Jang AS, Kim DJ, Koh ES, et al. Interleukin-13 and its receptors in idiopathic interstitial pneumonia: clinical implications for lung function. J Korean Med Sci. 2009; 24(4):614–620. PMID: 19654941.24. Budczies J, Klauschen F, Sinn BV, Győrffy B, Schmitt WD, Darb-Esfahani S, et al. Cutoff Finder: a comprehensive and straightforward Web application enabling rapid biomarker cutoff optimization. PLoS One. 2012; 7(12):e51862. PMID: 23251644.25. Hara A, Sakamoto N, Ishimatsu Y, Kakugawa T, Nakashima S, Hara S, et al. S100A9 in BALF is a candidate biomarker of idiopathic pulmonary fibrosis. Respir Med. 2012; 106(4):571–580. PMID: 22209187.26. Korthagen NM, Nagtegaal MM, van Moorsel CH, Kazemier KM, van den Bosch JM, Grutters JC. MRP14 is elevated in the bronchoalveolar lavage fluid of fibrosing interstitial lung diseases. Clin Exp Immunol. 2010; 161(2):342–347. PMID: 20550547.27. Bennett D, Salvini M, Fui A, Cillis G, Cameli P, Mazzei MA, et al. Calgranulin B and KL-6 in bronchoalveolar lavage of patients with IPF and NSIP. Inflammation. 2019; 42(2):463–470. PMID: 30680696.28. Ryckman C, McColl SR, Vandal K, de Médicis R, Lussier A, Poubelle PE, et al. Role of S100A8 and S100A9 in neutrophil recruitment in response to monosodium urate monohydrate crystals in the air-pouch model of acute gouty arthritis. Arthritis Rheum. 2003; 48(8):2310–2320. PMID: 12905486.29. Cesaro A, Anceriz N, Plante A, Pagé N, Tardif MR, Tessier PA. An inflammation loop orchestrated by S100A9 and calprotectin is critical for development of arthritis. PLoS One. 2012; 7(9):e45478. PMID: 23029038.30. Watters LC, Schwarz MI, Cherniack RM, Waldron JA, Dunn TL, Stanford RE, et al. Idiopathic pulmonary fibrosis. Pretreatment bronchoalveolar lavage cellular constituents and their relationships with lung histopathology and clinical response to therapy. Am Rev Respir Dis. 1987; 135(3):696–704. PMID: 3826895.31. Lee TH, Jang AS, Park JS, Kim TH, Choi YS, Shin HR, et al. Elevation of S100 calcium binding protein A9 in sputum of neutrophilic inflammation in severe uncontrolled asthma. Ann Allergy Asthma Immunol. 2013; 111(4):268–275.e1. PMID: 24054362.32. Lee TH, Chang HS, Bae DJ, Song HJ, Kim MS, Park JS, et al. Role of S100A9 in the development of neutrophilic inflammation in asthmatics and in a murine model. Clin Immunol. 2017; 183:158–166. PMID: 28847516.33. Pouwels SD, Nawijn MC, Bathoorn E, Riezebos-Brilman A, van Oosterhout AJ, Kerstjens HA, et al. Increased serum levels of LL37, HMGB1 and S100A9 during exacerbation in COPD patients. Eur Respir J. 2015; 45(5):1482–1485. PMID: 25931489.34. Quoc QL, Choi Y, Thi Bich TC, Yang EM, Shin YS, Park HS. S100A9 in adult asthmatic patients: a biomarker for neutrophilic asthma. Exp Mol Med. 2021; 53(7):1170–1179. PMID: 34285336.35. Wang CH, Punde TH, Huang CD, Chou PC, Huang TT, Wu WH, et al. Fibrocyte trafficking in patients with chronic obstructive asthma and during an acute asthma exacerbation. J Allergy Clin Immunol. 2015; 135(5):1154–62.e1. PMID: 25441632.36. Adams TS, Schupp JC, Poli S, Ayaub EA, Neumark N, Ahangari F, et al. Single-cell RNA-seq reveals ectopic and aberrant lung-resident cell populations in idiopathic pulmonary fibrosis. Sci Adv. 2020; 6(28):eaba1983. PMID: 32832599.37. Kyung SY, Byun KH, Yoon JY, Kim YJ, Lee SP, Park JW, et al. Advanced glycation end-products and receptor for advanced glycation end-products expression in patients with idiopathic pulmonary fibrosis and NSIP. Int J Clin Exp Pathol. 2013; 7(1):221–228. PMID: 24427342.38. Bhattacharyya S, Kelley K, Melichian DS, Tamaki Z, Fang F, Su Y, et al. Toll-like receptor 4 signaling augments transforming growth factor-β responses: a novel mechanism for maintaining and amplifying fibrosis in scleroderma. Am J Pathol. 2013; 182(1):192–205. PMID: 23141927.39. Chrysanthopoulou A, Mitroulis I, Apostolidou E, Arelaki S, Mikroulis D, Konstantinidis T, et al. Neutrophil extracellular traps promote differentiation and function of fibroblasts. J Pathol. 2014; 233(3):294–307. PMID: 24740698.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Two cases of idiopathic pulmonary fibrosis
- Pharmacological treatment of idiopathic pulmonary fibrosis and fibrosing interstitial lung diseases: current trends and future directions
- Diagnostic Efficacy of Serum Mac-2 Binding Protein Glycosylation Isomer and Other Markers for Liver Fibrosis in Non-Alcoholic Fatty Liver Diseases
- Idiopathic Pulmonary Fibrosis and Nonspecific Interstitial Pneumonia
- Identification of determinants that mediate binding between Tembusu virus and the cellular receptor heat shock protein A9