J Korean Med Sci.
2023 Dec;38(48):e361. 10.3346/jkms.2023.38.e361.
Susceptibility to Fosfomycin and Nitrofurantoin of ESBL-Positive Escherichia coli and Klebsiella pneumoniae Isolated From Urine of Pediatric Patients
- Affiliations
-
- 1Department of Clinical Research Design & Evaluation, Samsung Advanced Institute for Health Sciences and Technology (SAIHST), Sungkyunkwan University, Seoul, Korea
- 2Planning Team, SML Genetree, Seoul, Korea
- 3Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 4Asia Pacific Foundation for Infectious Diseases (APFID), Seoul, Korea
- 5Department of Pediatrics, Gyeongsang National University Changwon Hospital, Gyeongsang National University College of Medicine, Changwon, Korea
- 6Department of Pediatrics, Soonchunhyang University Bucheon Hospital, Soonchunhyang University College of Medicine, Bucheon, Korea
- 7Samsung Advanced Institute for Health Sciences and Technology (SAIHST), Sungkyunkwan University, Seoul, Korea
- KMID: 2549164
- DOI: http://doi.org/10.3346/jkms.2023.38.e361
Abstract
- Background
Pediatric urinary tract infection (UTI) caused by extended-spectrum β-lactamase (ESBL)-positive gram-negative bacilli (GNB) has limited options for oral antibiotic treatment. The purpose of this study was to investigate the susceptibility of ESBLpositive Escherichia coli and Klebsiella pneumoniae isolates from pediatric urine samples to two oral antibiotics (fosfomycin and nitrofurantoin).
Methods
From November 2020 to April 2022, ESBL-positive E. coli and K. pneumoniae isolates from urine samples were collected at Samsung Medical Center, Seoul, Korea. Patients over 18 years of age or with malignancy were excluded. For repeated isolates from the same patient, only the first isolate was tested. Minimum inhibitory concentrations (MICs) were measured using agar (fosfomycin) or broth (nitrofurantoin) dilution methods. MIC 50 and MIC 90 were measured for fosfomycin and nitrofurantoin in both E. coli and K. pneumoniae.
Results
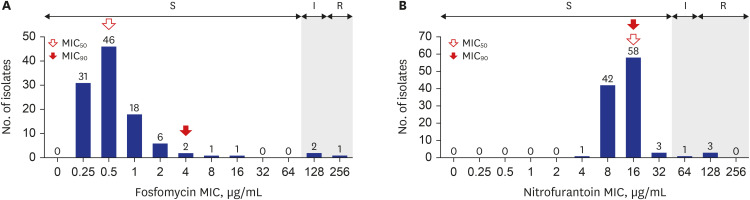
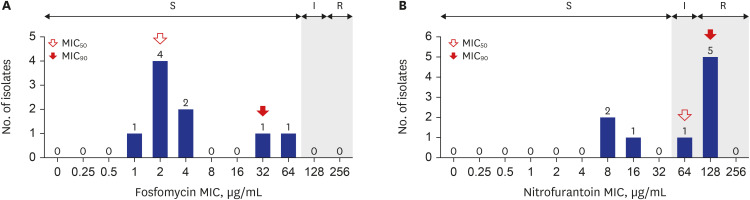
There were 117 isolates from 117 patients, with a median age of 7 months (range, 0.0–18.5 years). Among 117 isolates, 92.3% (108/117) were E. coli and 7.7% (9/117) were K. pneumoniae. Isolates from the pediatric intensive care unit (PICU) and general ward (GW) was 11.1% (13/117) and 88.9% (104/117), respectively. Among 108 E. coli isolates, MIC 50 and MIC 90 for fosfomycin were 0.5 μg/mL and 2 μg/mL, respectively. Fosfomycin susceptibility rate was 97.2% (105/108) with a breakpoint of 128 μg/mL. Fosfomycin susceptibility rate was significantly lower in PICU isolates than in GW isolates (81.8% vs. 99.0%, P = 0.027). For nitrofurantoin, both the MIC 50 and MIC 90 were 16 μg/mL. Nitrofurantoin susceptibility rate was 96.3% (104/108) with a breakpoint of 64 μg/mL based on Clinical and Laboratory Standards Institute guidelines. Among the nine K. pneumoniae isolates, the MIC 50 and MIC 90 for fosfomycin was 2 μg/mL and 32 μg/mL, respectively. MIC 50 and MIC 90 for nitrofurantoin were 64 μg/mL and 128 μg/mL, respectively.
Conclusion
For uncomplicated UTI caused by ESBL-positive GNB in Korean children, treatment with fosfomycin and nitrofurantoin for E. coli infections can be considered as an effective oral therapy option.
Keyword
Figure
Reference
-
1. Fan NC, Chen HH, Chen CL, Ou LS, Lin TY, Tsai MH, et al. Rise of community-onset urinary tract infection caused by extended-spectrum β-lactamase-producing Escherichia coli in children. J Microbiol Immunol Infect. 2014; 47(5):399–405. PMID: 23834784.
Article2. Dayan N, Dabbah H, Weissman I, Aga I, Even L, Glikman D. Urinary tract infections caused by community-acquired extended-spectrum β-lactamase-producing and nonproducing bacteria: a comparative study. J Pediatr. 2013; 163(5):1417–1421. PMID: 23919903.
Article3. Topaloglu R, Er I, Dogan BG, Bilginer Y, Ozaltin F, Besbas N, et al. Risk factors in community-acquired urinary tract infections caused by ESBL-producing bacteria in children. Pediatr Nephrol. 2010; 25(5):919–925. PMID: 20151161.
Article4. Saltoglu N, Karali R, Yemisen M, Ozaras R, Balkan II, Mete B, et al. Comparison of community-onset healthcare-associated and hospital-acquired urinary infections caused by extended-spectrum beta-lactamase-producing Escherichia coli and antimicrobial activities. Int J Clin Pract. 2015; 69(7):766–770. PMID: 25683907.
Article5. Ladhani S, Gransden W. Increasing antibiotic resistance among urinary tract isolates. Arch Dis Child. 2003; 88(5):444–445. PMID: 12716722.
Article6. Cheng MF, Chen WL, Huang IF, Chen JR, Chiou YH, Chen YS, et al. Urinary tract infection in infants caused by extended-spectrum beta-lactamase-producing Escherichia coli: comparison between urban and rural hospitals. Pediatr Nephrol. 2016; 31(8):1305–1312. PMID: 26975387.
Article7. Lee YK, Lee H, Kim JM, Kang JM, Lee ST, Lee NY, et al. The antibiotic resistance pattern of gram-negative bacteria in children younger than 24 months with a urinary tract infection: a retrospective single-center study over 15 consecutive years. Child Kidney Dis. 2015; 19(2):148–153.
Article8. Subcommittee on Urinary Tract Infection, Steering Committee on Quality Improvement and Management. Roberts KB. Urinary tract infection: clinical practice guideline for the diagnosis and management of the initial UTI in febrile infants and children 2 to 24 months. Pediatrics. 2011; 128(3):595–610. PMID: 21873693.
Article9. Doi A, Shimada T, Harada S, Iwata K, Kamiya T. The efficacy of cefmetazole against pyelonephritis caused by extended-spectrum beta-lactamase-producing Enterobacteriaceae. Int J Infect Dis. 2013; 17(3):e159–e163. PMID: 23140947.
Article10. Tasbakan MI, Pullukcu H, Sipahi OR, Yamazhan T, Ulusoy S. Nitrofurantoin in the treatment of extended-spectrum β-lactamase-producing Escherichia coli-related lower urinary tract infection. Int J Antimicrob Agents. 2012; 40(6):554–556. PMID: 23063743.
Article11. Neuner EA, Sekeres J, Hall GS, van Duin D. Experience with fosfomycin for treatment of urinary tract infections due to multidrug-resistant organisms. Antimicrob Agents Chemother. 2012; 56(11):5744–5748. PMID: 22926565.
Article12. European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID). EUCAST Definitive Document E.DEF 3.1, June 2000: Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by agar dilution. Clin Microbiol Infect. 2000; 6(9):509–515. PMID: 11168187.13. Vachvanichsanong P, McNeil EB, Dissaneewate P. Extended-spectrum beta-lactamase Escherichia coli and Klebsiella pneumoniae urinary tract infections. Epidemiol Infect. 2020; 149:e12. PMID: 33327984.
Article14. CLSI. Performance Standards for Antimicrobial Susceptibility Testing, 32nd Edition. CLSI Guideline M100. Wayne, PA, USA: Clinical and Laboratory Standards Institute;2022.15. Raphael E, Glymour MM, Chambers HF. Trends in prevalence of extended-spectrum beta-lactamase-producing Escherichia coli isolated from patients with community- and healthcare-associated bacteriuria: results from 2014 to 2020 in an urban safety-net healthcare system. Antimicrob Resist Infect Control. 2021; 10(1):118. PMID: 34380549.16. Seo MR, Kim SJ, Kim Y, Kim J, Choi TY, Kang JO, et al. Susceptibility of Escherichia coli from community-acquired urinary tract infection to fosfomycin, nitrofurantoin, and temocillin in Korea. J Korean Med Sci. 2014; 29(8):1178–1181. PMID: 25120333.
Article17. Karlowsky JA, Denisuik AJ, Lagacé-Wiens PR, Adam HJ, Baxter MR, Hoban DJ, et al. In vitro activity of fosfomycin against Escherichia coli isolated from patients with urinary tract infections in Canada as part of the CANWARD surveillance study. Antimicrob Agents Chemother. 2014; 58(2):1252–1256. PMID: 24323476.
Article18. Keepers TR, Gomez M, Celeri C, Krause KM, Biek D, Critchley I. Fosfomycin and comparator activity against select Enterobacteriaceae, Pseudomonas, and Enterococcus urinary tract infection isolates from the United States in 2012. Infect Dis Ther. 2017; 6(2):233–243. PMID: 28285420.
Article19. Raja NS. Oral treatment options for patients with urinary tract infections caused by extended spectrum βeta-lactamase (ESBL) producing Enterobacteriaceae. J Infect Public Health. 2019; 12(6):843–846. PMID: 31176606.
Article20. Jia P, Zhu Y, Li X, Kudinha T, Yang Y, Zhang G, et al. High prevalence of extended-spectrum beta-lactamases in Escherichia coli strains collected from strictly defined community-acquired urinary tract infections in adults in China: a multicenter prospective clinical microbiological and molecular study. Front Microbiol. 2021; 12:663033. PMID: 34305831.
Article21. Perween N, Rai S, Nandwani S, Kumar SK 2nd. Retrospective analysis of urinary tract infection in the pediatric population at a tertiary care centre. Cureus. 2022; 14(5):e24796. PMID: 35677001.
Article22. Ko KS, Suh JY, Peck KR, Lee MY, Oh WS, Kwon KT, et al. In vitro activity of fosfomycin against ciprofloxacin-resistant or extended-spectrum beta-lactamase-producing Escherichia coli isolated from urine and blood. Diagn Microbiol Infect Dis. 2007; 58(1):111–115. PMID: 17300900.23. Tamma PD, Aitken SL, Bonomo RA, Mathers AJ, van Duin D, Clancy CJ. Infectious Diseases Society of America 2022 guidance on the treatment of extended-spectrum β-lactamase producing Enterobacterales (ESBL-E), carbapenem-resistant Enterobacterales (CRE), and Pseudomonas aeruginosa with difficult-to-treat resistance (DTR-P. aeruginosa). Clin Infect Dis. 2022; 75(2):187–212. PMID: 35439291.
Article24. Elliott ZS, Barry KE, Cox HL, Stoesser N, Carroll J, Vegesana K, et al. The role of fosA in challenges with fosfomycin susceptibility testing of multispecies Klebsiella pneumoniae carbapenemase-producing clinical isolates. J Clin Microbiol. 2019; 57(10):e00634–e00619. PMID: 31340992.25. Huttner A, Kowalczyk A, Turjeman A, Babich T, Brossier C, Eliakim-Raz N, et al. Effect of 5-day nitrofurantoin vs single-dose fosfomycin on clinical resolution of uncomplicated lower urinary tract infection in women: a randomized clinical trial. JAMA. 2018; 319(17):1781–1789. PMID: 29710295.
Article26. Wald-Dickler N, Lee TC, Tangpraphaphorn S, Butler-Wu SM, Wang N, Degener T, et al. Fosfomycin vs ertapenem for outpatient treatment of complicated urinary tract infections: a multicenter, retrospective cohort study. Open Forum Infect Dis. 2021; 9(1):ofab620. PMID: 35036466.27. Hatlen TJ, Flor R, Nguyen MH, Lee GH, Miller LG. Oral fosfomycin use for pyelonephritis and complicated urinary tract infections: a 1 year review of outcomes and prescribing habits in a large municipal healthcare system. J Antimicrob Chemother. 2020; 75(7):1993–1997. PMID: 32303061.
Article28. Purcell R, Wang N, Gwee A. Can fosfomycin be used for the treatment of Gram-negative urinary tract infections in children? Arch Dis Child. 2021; 106(9):925–928. PMID: 33461959.
Article29. Kara A, Gurgoze MK. The use of nitrofurantoin for children with acute cystitis caused by extended-spectrum B-lactamase-producing Escherichia coli. J Pediatr Urol. 2019; 15(4):378.e1–378.e5.30. Murray TS, Peaper DR. The contribution of extended-spectrum β-lactamases to multidrug-resistant infections in children. Curr Opin Pediatr. 2015; 27(1):124–131. PMID: 25565574.
Article31. Purcell R, Yeoh D, Bowen A, Britton PN, Carr JP, Chen M, et al. A multicentre, retrospective audit of fosfomycin use for urinary tract infections in Australian children and adolescents. J Antimicrob Chemother. 2023; 78(7):1616–1621. PMID: 37190910.
Article32. Mayama T, Yokota M, Shimatani I, Ohyagi H. Analysis of oral fosfomycin calcium (Fosmicin) side-effects after marketing. Int J Clin Pharmacol Ther Toxicol. 1993; 31(2):77–82. PMID: 8458680.33. Mikkelsen LF, Rubak S. Reversible lung fibrosis in a 6-year-old girl after long term nitrofurantoin treatment. BMC Pulm Med. 2020; 20(1):313. PMID: 33243181.
Article34. Falagas ME, Vouloumanou EK, Samonis G, Vardakas KZ. Fosfomycin. Clin Microbiol Rev. 2016; 29(2):321–347. PMID: 26960938.
Article35. Yang TY, Lu PL, Tseng SP. Update on fosfomycin-modified genes in Enterobacteriaceae. J Microbiol Immunol Infect. 2019; 52(1):9–21. PMID: 29198952.
Article36. Cattoir V, Guérin F. How is fosfomycin resistance developed in Escherichia coli? Future Microbiol. 2018; 13:1693–1696. PMID: 30526061.37. Lee SY, Park YJ, Yu JK, Jung S, Kim Y, Jeong SH, et al. Prevalence of acquired fosfomycin resistance among extended-spectrum β-lactamase-producing Escherichia coli and Klebsiella pneumoniae clinical isolates in Korea and IS26-composite transposon surrounding fosA3. J Antimicrob Chemother. 2012; 67(12):2843–2847. PMID: 22893681.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- In Vitro Susceptibility of piperacillin/tazobactam Against extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae
- Evaluation of the Method to Screen Isolates of Extended-Spectrum -Lactamase-Producing Klebsiella pneumoniae and Escherichia coli Using Cefpodoxime Disk
- Comparison of Escherichia coli and Klebsiella pneumoniae Acute Pyelonephritis in Korean Patients
- Detection Rate of Extended-Spectrum beta-Lactamase Producers in Klebsiella pneumoniae and Escherichia coli Isolated at Yeungnam University Medical Center
- Antimicrobial Susceptibility of Escherichia coli and Klebsiella pneumoniae Blood Isolates over 5 years:Influence of Extended-Spectrum Beta-Lactamase-Producing Organisms