Brain Tumor Res Treat.
2023 Oct;11(4):223-231. 10.14791/btrt.2023.0026.
FLASH Radiotherapy: A FLASHing Idea to Preserve Neurocognitive Function
- Affiliations
-
- 1College of Veterinary Medicine, Seoul National University, Seoul, Korea
- 2College of Medicine, Seoul National University, Seoul, Korea
- KMID: 2547410
- DOI: http://doi.org/10.14791/btrt.2023.0026
Abstract
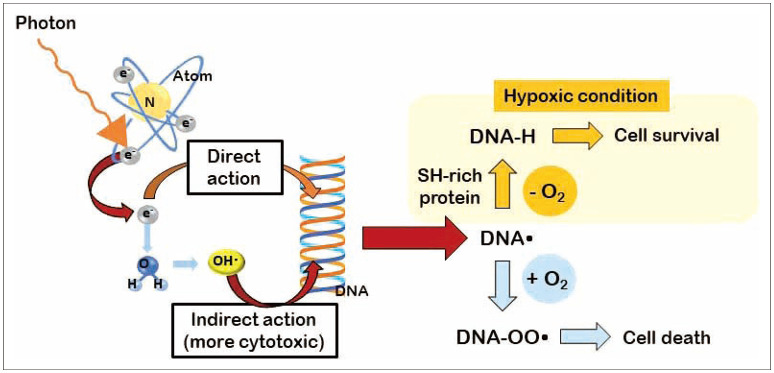
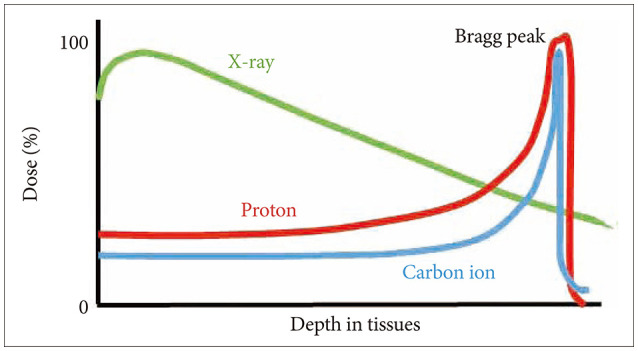
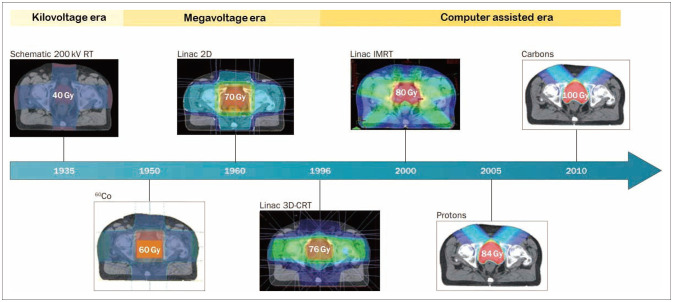
- FLASH radiotherapy (FLASH RT) is a technique to deliver ultra-high dose rate in a fraction of a second. Evidence from experimental animal models suggest that FLASH RT spares various normal tissues including the lung, gastrointestinal track, and brain from radiation-induced toxicity (a phenomenon known as FLASH effect), which is otherwise commonly observed with conventional dose rate RT. However, it is not simply the ultra-high dose rate alone that brings the FLASH effect. Multiple parameters such as instantaneous dose rate, pulse size, pulse repetition frequency, and the total duration of exposure all need to be carefully optimized simultaneously. Furthermore it is critical to validate FLASH effects in an in vivo experimental model system. The exact molecular mechanism responsible for this FLASH effect is not yet understood although a number of hypotheses have been proposed including oxygen depletion and less reactive oxygen species (ROS) production by FLASH RT, and enhanced ability of normal tissues to handle ROS and labile iron pool compared to tumors. In this review, we briefly overview the process of ionization event and history of radiotherapy and fractionation of ionizing radiation. We also highlight some of the latest FLASH RT reviews and results with a special interest to neurocognitive protection in rodent model with whole brain irradiation. Lastly we discuss some of the issues remain to be answered with FLASH RT including undefined molecular mechanism, lack of standardized parameters, low penetration depth for electron beam, and tumor hypoxia still being a major hurdle for local control. Nevertheless, researchers are close to having all answers to the issues that we have raised, hence we believe that advancement of FLASH RT will be made more quickly than one can anticipate.
Figure
Reference
-
1. Podgorsak EB. Treatment machines for external beam radiotherapy. Podgorsak EB, editor. Radiation oncology physics: a handbook for teachers and students. Vienna: International Atomic Energy Agency;2005. p. 123–160.2. Hall EJ, Giaccia AJ. Physics and chemistry of radiation absorption. Radiobiology for the radiologist. 8th ed. Alphen aan den Rijn: Wolters Kluwer;2019. p. 2–10.3. Reisz JA, Bansal N, Qian J, Zhao W, Furdui CM. Effects of ionizing radiation on biological molecules--mechanisms of damage and emerging methods of detection. Antioxid Redox Signal. 2014; 21:260–292. PMID: 24382094.
Article4. Brown JM, Wilson WR. Exploiting tumour hypoxia in cancer treatment. Nat Rev Cancer. 2004; 4:437–447. PMID: 15170446.
Article5. Ahn GO, Brown M. Targeting tumors with hypoxia-activated cytotoxins. Front Biosci. 2007; 12:3483–3501. PMID: 17485316.
Article6. Hall EJ, Giaccia AJ. Linear energy transfer and relative biological effectiveness. Radiobiology for the radiologist. 8th ed. Alphen aan den Rijn: Wolters Kluwer;2019. p. 101–110.7. Hall EJ, Giaccia AJ. Oxygen effect and reoxygenation. Radiobiology for the radiologist. 8th ed. Alphen aan den Rijn: Wolters Kluwer;2019. p. 82–100.8. Bussière MR, Adams JA. Treatment planning for conformal proton radiation therapy. Technol Cancer Res Treat. 2003; 2:389–399. PMID: 14529304.
Article9. Thariat J, Hannoun-Levi JM, Sun Myint A, Vuong T, Gérard JP. Past, present, and future of radiotherapy for the benefit of patients. Nat Rev Clin Oncol. 2013; 10:52–60. PMID: 23183635.
Article10. Regaud C, Ferroux R. Discordance des effects de rayons X, d’une part dans le testicile, par le peau, d’autre parts dans le fractionment de la dose. Compt Rend Soc Biol. 1927; 97:431–434.11. Bernier J, Hall EJ, Giaccia A. Radiation oncology: a century of achievements. Nat Rev Cancer. 2004; 4:737–747. PMID: 15343280.
Article12. Hall EJ, Giaccia AJ. Time, dose, and fractionation in radiotherapy. Radiobiology for the radiologist. 8th ed. Alphen aan den Rijn: Wolters Kluwer;2019. p. 417–436.13. Liao G, Zhao Z, Yang H, Li X. Efficacy and safety of hypofractionated radiotherapy for the treatment of newly diagnosed glioblastoma multiforme: a systematic review and meta-analysis. Front Oncol. 2019; 9:1017. PMID: 31681570.
Article14. Buchberger DS, Videtic GMM. Stereotactic body radiotherapy for the management of early-stage non-small-cell lung cancer: a clinical overview. JCO Oncol Pract. 2023; 19:239–249. PMID: 36800644.
Article15. Dong B, Chen R, Zhu X, Wu Q, Jin J, Wang W, et al. Comparison of stereotactic body radiation therapy versus surgery for multiple primary lung cancers after prior radical resection: a multicenter retrospective study. Clin Transl Radiat Oncol. 2023; 40:100601. PMID: 36936471.
Article16. Ajithkumar T, Price S, Horan G, Burke A, Jefferies S. Prevention of radiotherapy-induced neurocognitive dysfunction in survivors of paediatric brain tumours: the potential role of modern imaging and radiotherapy techniques. Lancet Oncol. 2017; 18:e91–e100. PMID: 28214420.
Article17. Parsons MW, Peters KB, Floyd SR, Brown P, Wefel JS. Preservation of neurocognitive function in the treatment of brain metastases. Neurooncol Adv. 2021; 3(Suppl 5):v96–v107. PMID: 34859237.
Article18. Monje M, Dietrich J. Cognitive side effects of cancer therapy demonstrate a functional role for adult neurogenesis. Behav Brain Res. 2012; 227:376–379. PMID: 21621557.
Article19. Pazzaglia S, Briganti G, Mancuso M, Saran A. Neurocognitive decline following radiotherapy: mechanisms and therapeutic implications. Cancers (Basel). 2020; 12:146. PMID: 31936195.
Article20. Schüler E, Trovati S, King G, Lartey F, Rafat M, Villegas M, et al. Experimental platform for ultra-high dose rate FLASH irradiation of small animals using a clinical linear accelerator. Int J Radiat Oncol Biol Phys. 2017; 97:195–203. PMID: 27816362.
Article21. Favaudon V, Caplier L, Monceau V, Pouzoulet F, Sayarath M, Fouillade C, et al. Ultrahigh dose-rate FLASH irradiation increases the differential response between normal and tumor tissue in mice. Sci Transl Med. 2014; 6:245ra93.
Article22. Webb S. Motion effects in (intensity modulated) radiation therapy: a review. Phys Med Biol. 2006; 51:R403–R425. PMID: 16790915.
Article23. No HJ, Wu YF, Dworkin ML, Manjappa R, Skinner L, Ashraf MR, et al. Clinical linear accelerator-based electron FLASH: pathway for practical translation to FLASH clinical trials. Int J Radiat Oncol Biol Phys. 2023; 117:482–492. PMID: 37105403.
Article24. Vozenin MC, Montay-Gruel P, Limoli C, Germond JF. All irradiations that are ultra-high dose rate may not be FLASH: the critical importance of beam parameter characterization and in vivo validation of the FLASH effect. Radiat Res. 2020; 194:571–572. PMID: 32853355.
Article25. Montay-Gruel P, Acharya MM, Gonçalves Jorge P, Petit B, Petridis IG, Fuchs P, et al. Hypofractionated FLASH-RT as an effective treatment against glioblastoma that reduces neurocognitive side effects in mice. Clin Cancer Res. 2021; 27:775–784. PMID: 33060122.
Article26. Wilson JD, Hammond EM, Higgins GS, Petersson K. Ultra-high dose rate (FLASH) radiotherapy: silver bullet or fool’s gold? Front Oncol. 2020; 9:1563. PMID: 32010633.
Article27. Levy K, Natarajan S, Wang J, Chow S, Eggold JT, Loo PE, et al. Abdominal FLASH irradiation reduces radiation-induced gastrointestinal toxicity for the treatment of ovarian cancer in mice. Sci Rep. 2020; 10:21600. PMID: 33303827.
Article28. Montay-Gruel P, Acharya MM, Petersson K, Alikhani L, Yakkala C, Allen BD, et al. Long-term neurocognitive benefits of FLASH radiotherapy driven by reduced reactive oxygen species. Proc Natl Acad Sci U S A. 2019; 116:10943–10951. PMID: 31097580.
Article29. Barcellos-Hoff MH. Latency and activation in the control of TGF-beta. J Mammary Gland Biol Neoplasia. 1996; 1:353–363. PMID: 10887509.30. Simmons DA, Lartey FM, Schüler E, Rafat M, King G, Kim A, et al. Reduced cognitive deficits after FLASH irradiation of whole mouse brain are associated with less hippocampal dendritic spine loss and neuroinflammation. Radiother Oncol. 2019; 139:4–10. PMID: 31253467.
Article31. Dewey DL, Boag JW. Modification of the oxygen effect when bacteria are given large pulses of radiation. Nature. 1959; 183:1450–1451. PMID: 13657161.
Article32. El Khatib M, Van Slyke AL, Velalopoulou A, Kim MM, Shoniyozov K, Allu SR, et al. Ultrafast tracking of oxygen dynamics during proton FLASH. Int J Radiat Oncol Biol Phys. 2022; 113:624–634. PMID: 35314293.
Article33. Vozenin MC, Hendry JH, Limoli CL. Biological benefits of ultra-high dose rate FLASH radiotherapy: sleeping beauty awoken. Clin Oncol (R Coll Radiol). 2019; 31:407–415. PMID: 31010708.
Article34. Berry RJ, Hall EJ, Forster DW, Storr TH, Goodman MJ. Survival of mammalian cells exposed to X rays at ultra-high dose-rates. Br J Radiol. 1969; 42:102–107. PMID: 4975207.
Article35. Adrian G, Konradsson E, Lempart M, Bäck S, Ceberg C, Petersson K. The FLASH effect depends on oxygen concentration. Br J Radiol. 2020; 93:20190702. PMID: 31825653.
Article36. Beyreuther E, Brand M, Hans S, Hideghéty K, Karsch L, Leßmann E, et al. Feasibility of proton FLASH effect tested by zebrafish embryo irradiation. Radiother Oncol. 2019; 139:46–50. PMID: 31266652.
Article37. Venkatesulu BP, Sharma A, Pollard-Larkin JM, Sadagopan R, Symons J, Neri S, et al. Ultra high dose rate (35 Gy/sec) radiation does not spare the normal tissue in cardiac and splenic models of lymphopenia and gastrointestinal syndrome. Sci Rep. 2019; 9:17180. PMID: 31748640.
Article38. Montay-Gruel P, Petersson K, Jaccard M, Boivin G, Germond JF, Petit B, et al. Irradiation in a flash: unique sparing of memory in mice after whole brain irradiation with dose rates above 100Gy/s. Radiother Oncol. 2017; 124:365–369. PMID: 28545957.
Article39. Pena LA, Fuks Z, Kolesnick RN. Radiation-induced apoptosis of endothelial cells in the murine central nervous system: protection by fibroblast growth factor and sphingomyelinase deficiency. Cancer Res. 2000; 60:321–327. PMID: 10667583.40. Barazzuol L, Coppes RP, van Luijk P. Prevention and treatment of radiotherapy-induced side effects. Mol Oncol. 2020; 14:1538–1554. PMID: 32521079.
Article41. Barazzuol L, Hopkins SR, Ju L, Jeggo PA. Distinct response of adult neural stem cells to low versus high dose ionising radiation. DNA Repair (Amst). 2019; 76:70–75. PMID: 30822688.
Article42. Barazzuol L, Ju L, Jeggo PA. A coordinated DNA damage response promotes adult quiescent neural stem cell activation. PLoS Biol. 2017; 15:e2001264. PMID: 28489848.
Article43. Monje ML, Mizumatsu S, Fike JR, Palmer TD. Irradiation induces neural precursor-cell dysfunction. Nat Med. 2002; 8:955–962. PMID: 12161748.
Article44. Allen BD, Acharya MM, Montay-Gruel P, Jorge PG, Bailat C, Petit B, et al. Maintenance of tight junction integrity in the absence of vascular dilation in the brain of mice exposed to ultra-high-dose-rate FLASH irradiation. Radiat Res. 2020; 194:625–635. PMID: 33348373.
Article45. Alaghband Y, Cheeks SN, Allen BD, Montay-Gruel P, Doan NL, Petit B, et al. Neuroprotection of radiosensitive juvenile mice by ultra-high dose rate FLASH irradiation. Cancers (Basel). 2020; 12:1671. PMID: 32599789.
Article46. Paolicelli RC, Bolasco G, Pagani F, Maggi L, Scianni M, Panzanelli P, et al. Synaptic pruning by microglia is necessary for normal brain development. Science. 2011; 333:1456–1458. PMID: 21778362.
Article47. Ding X, Wang J, Huang M, Chen Z, Liu J, Zhang Q, et al. Loss of microglial SIRPα promotes synaptic pruning in preclinical models of neurodegeneration. Nat Commun. 2021; 12:2030. PMID: 33795678.
Article48. Vozenin MC, De Fornel P, Petersson K, Favaudon V, Jaccard M, Germond JF, et al. The advantage of FLASH radiotherapy confirmed in mini-pig and cat-cancer patients. Clin Cancer Res. 2019; 25:35–42. PMID: 29875213.
Article49. Bourhis J, Sozzi WJ, Jorge PG, Gaide O, Bailat C, Duclos F, et al. Treatment of a first patient with FLASH-radiotherapy. Radiother Oncol. 2019; 139:18–22. PMID: 31303340.
Article50. Mascia AE, Daugherty EC, Zhang Y, Lee E, Xiao Z, Sertorio M, et al. Proton FLASH radiotherapy for the treatment of symptomatic bone metastases: the FAST-01 nonrandomized trial. JAMA Oncol. 2023; 9:62–69. PMID: 36273324.
Article51. Spitz DR, Buettner GR, Petronek MS, St-Aubin JJ, Flynn RT, Waldron TJ, et al. An integrated physico-chemical approach for explaining the differential impact of FLASH versus conventional dose rate irradiation on cancer and normal tissue responses. Radiother Oncol. 2019; 139:23–27. PMID: 31010709.
Article52. Eggold JT, Chow S, Melemenidis S, Wang J, Natarajan S, Loo PE, et al. Abdominopelvic FLASH irradiation improves PD-1 immune checkpoint inhibition in preclinical models of ovarian cancer. Mol Cancer Ther. 2022; 21:371–381. PMID: 34866044.
Article53. Shi X, Yang Y, Zhang W, Wang J, Xiao D, Ren H, et al. FLASH X-ray spares intestinal crypts from pyroptosis initiated by cGAS-STING activation upon radioimmunotherapy. Proc Natl Acad Sci U S A. 2022; 119:e2208506119. PMID: 36256824.
Article54. Jorge PG, Melemenidis S, Grilj V, Buchillier T, Manjappa R, Viswanathan V, et al. Design and validation of a dosimetric comparison scheme tailored for ultra-high dose-rate electron beams to support multicenter FLASH preclinical studies. Radiother Oncol. 2022; 175:203–209. PMID: 36030934.
Article55. Skaggs LS. Depth dose of electrons from the betatron. Radiology. 1949; 53:868–874. PMID: 15398652.
Article56. Velalopoulou A, Karagounis IV, Cramer GM, Kim MM, Skoufos G, Goia D, et al. FLASH proton radiotherapy spares normal epithelial and mesenchymal tissues while preserving sarcoma response. Cancer Res. 2021; 81:4808–4821. PMID: 34321243.
Article57. Diffenderfer ES, Sørensen BS, Mazal A, Carlson DJ. The current status of preclinical proton FLASH radiation and future directions. Med Phys. 2022; 49:2039–2054. PMID: 34644403.
Article58. Diffenderfer ES, Verginadis II, Kim MM, Shoniyozov K, Velalopoulou A, Goia D, et al. Design, implementation, and in vivo validation of a novel proton FLASH radiation therapy system. Int J Radiat Oncol Biol Phys. 2020; 106:440–448. PMID: 31928642.
Article59. Maxim PG, Tantawi SG, Loo BW Jr. PHASER: a platform for clinical translation of FLASH cancer radiotherapy. Radiother Oncol. 2019; 139:28–33. PMID: 31178058.
Article60. Ahn GO, Ware DC, Denny WA, Wilson WR. Optimization of the auxiliary ligand shell of cobalt(III)(8-hydroxyquinoline) complexes as model hypoxia-selective radiation-activated prodrugs. Radiat Res. 2004; 162:315–325. PMID: 15333003.
Article61. Leavitt RJ, Almeida A, Grilj V, Montay-Gruel P, Godfroid C, Petit B, et al. Hypoxic tumors are sensitive to FLASH radiotherapy. bioRxiv [Preprint]. 2023; cited 2023 August 23. Available at: . DOI: 10.1101/2022.11.27.518083.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Intraoperative monitoring of flash visual evoked potential under general anesthesia
- Neurocognitive Dysfunction Related with Sleep Factors in Patients with Obstructive Sleep Apnea
- Neurocognitive Function Tests for Elderly
- Neurocognitive Functions in Posttraumatic Stress Disorder
- Proton therapy in pediatric brain tumors