Nutr Res Pract.
2023 Oct;17(5):844-854. 10.4162/nrp.2023.17.5.844.
Antimetastatic effect of fucoidan against non-small cell lung cancer by suppressing non-receptor tyrosine kinase and extracellular signalrelated kinase pathway
- Affiliations
-
- 1Division of Health and Applied Sciences, Faculty of Science, Prince of Songkla University, Hat Yai, Songkhla 90110, Thailand
- KMID: 2546943
- DOI: http://doi.org/10.4162/nrp.2023.17.5.844
Abstract
- BACKGROUND/OBJECTIVES
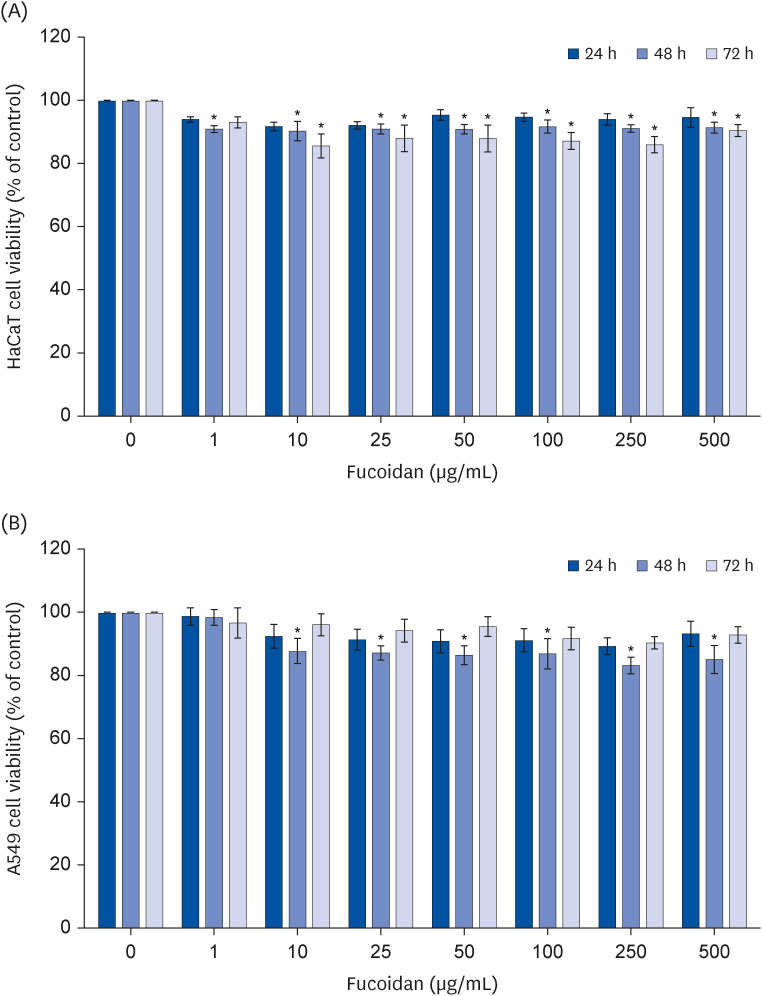
Fucoidan, a polysaccharide content in brown algae, has been reported to inhibit the growth of cancer cells. The present study aimed to investigate the suppression effects of fucoidan on A549 non-small cell lung cancer cells migration.
MATERIALS/METHODS
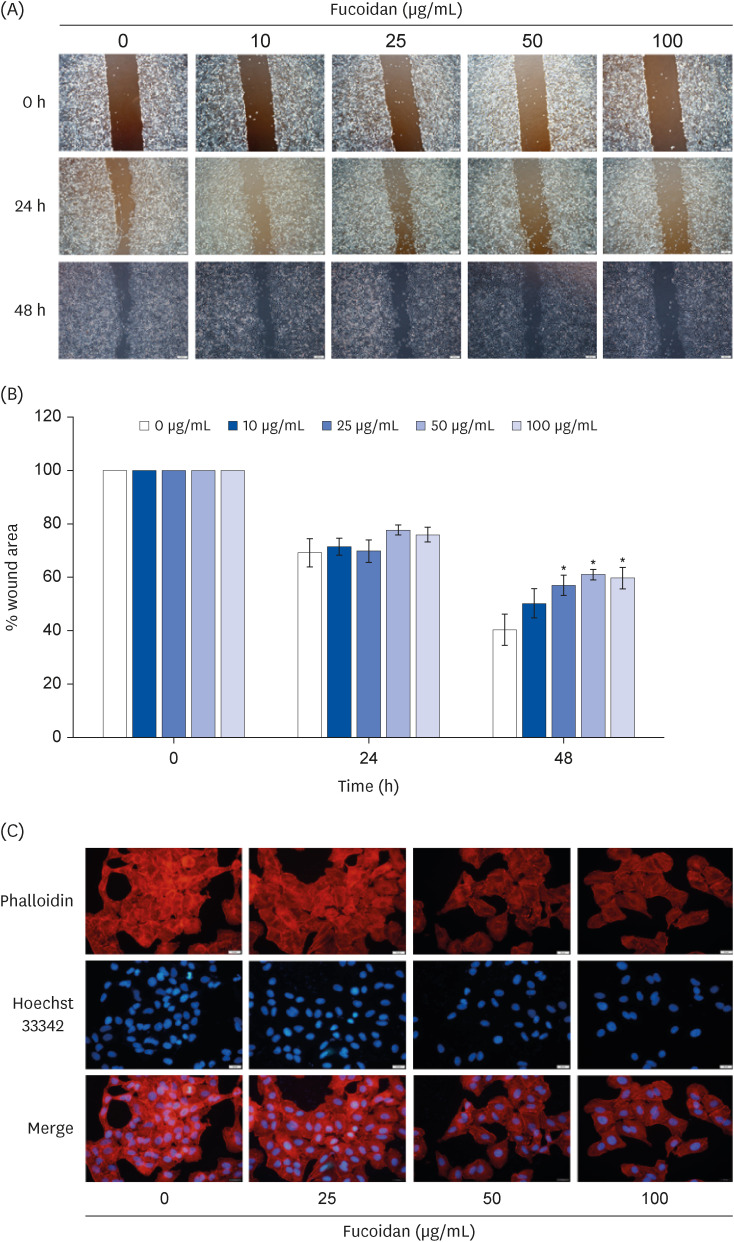
The anti-migratory activity of fucoidan in A549 cells was examined by wound healing assay and phalloidin-rhodamine staining in response to fucoidan (0–100 µg/mL) treatment for 48 h. Western blot analysis was performed to clarify the protein expressions relevant to migratory activity.
RESULTS
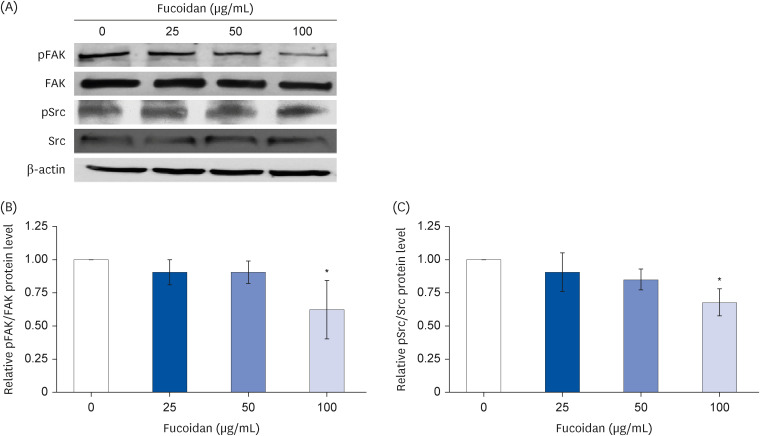
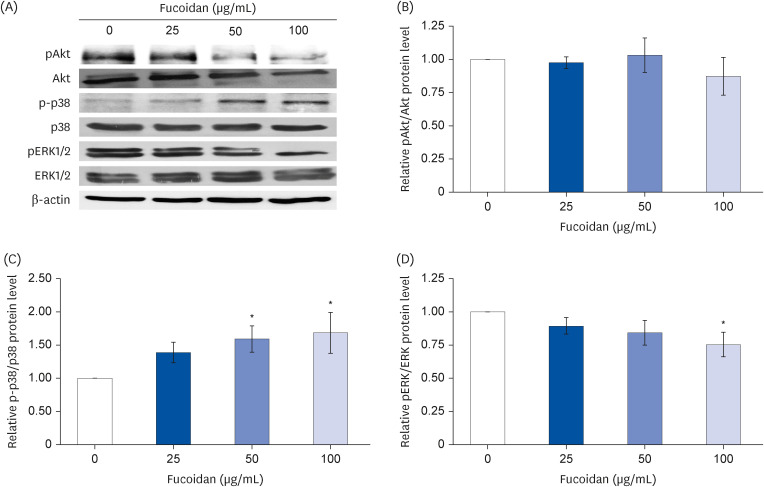
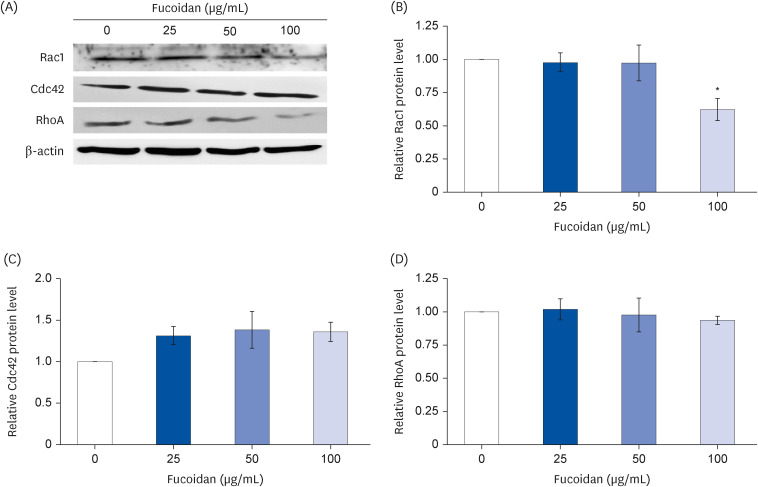
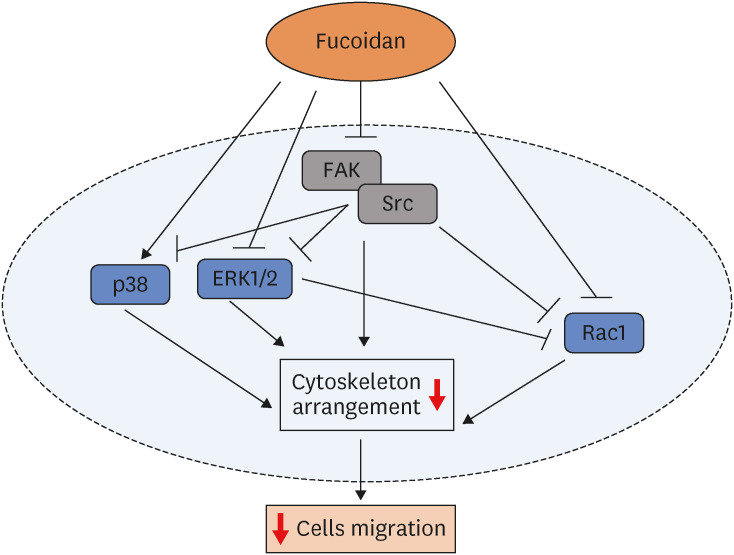
Fucoidan (25–100 µg/mL) significantly suppressed A549 cells migration together with reduced the intensity of phalloidin-rhodamine which detect filopodia and lamellipodia protrusions at 48 h of treatment. The protein expression indicated that fucoidan significantly suppressed the phosphorylation of focal adhesion kinase (FAK), Src, and extracellular signalrelated kinase (ERK). In addition, the phosphorylation of p38 in A549 cells was found to be increased.
CONCLUSIONS
Our data conclude that fucoidan exhibits anti-migratory activities against lung cancer A549 cells mediated by inhibiting ERK1/2 and FAK-Src pathway.
Keyword
Figure
Reference
-
1. Barta JA, Powell CA, Wisnivesky JP. Global epidemiology of lung cancer. Ann Glob Health. 2019; 85:8. PMID: 30741509.2. Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018; 553:446–454. PMID: 29364287.3. Olson MF, Sahai E. The actin cytoskeleton in cancer cell motility. Clin Exp Metastasis. 2009; 26:273–287. PMID: 18498004.4. Svitkina T. The actin cytoskeleton and actin-based motility. Cold Spring Harb Perspect Biol. 2018; 10:a018267. PMID: 29295889.5. Carelli S, Zadra G, Vaira V, Falleni M, Bottiglieri L, Nosotti M, Di Giulio AM, Gorio A, Bosari S. Up-regulation of focal adhesion kinase in non-small cell lung cancer. Lung Cancer. 2006; 53:263–271. PMID: 16842883.6. Chikara S, Lindsey K, Borowicz P, Christofidou-Solomidou M, Reindl KM. Enterolactone alters FAK-Src signaling and suppresses migration and invasion of lung cancer cell lines. BMC Complement Altern Med. 2017; 17:30. PMID: 28068967.7. Masraksa W, Tanasawet S, Hutamekalin P, Wongtawatchai T, Sukketsiri W. Luteolin attenuates migration and invasion of lung cancer cells via suppressing focal adhesion kinase and non-receptor tyrosine kinase signaling pathway. Nutr Res Pract. 2020; 14:127–133. PMID: 32256987.8. Mitra SK, Schlaepfer DD. Integrin-regulated FAK-Src signaling in normal and cancer cells. Curr Opin Cell Biol. 2006; 18:516–523. PMID: 16919435.9. van Weelden G, Bobiński M, Okła K, van Weelden WJ, Romano A, Pijnenborg JM. Fucoidan structure and activity in relation to anti-cancer mechanisms. Mar Drugs. 2019; 17:32. PMID: 30621045.10. Kim H, Ahn JH, Song M, Kim DW, Lee TK, Lee JC, Kim YM, Kim JD, Cho JH, Hwang IK, et al. Pretreated fucoidan confers neuroprotection against transient global cerebral ischemic injury in the gerbil hippocampal CA1 area via reducing of glial cell activation and oxidative stress. Biomed Pharmacother. 2019; 109:1718–1727. PMID: 30551426.11. Wang Y, Wang Q, Han X, Ma Y, Zhang Z, Zhao L, Guan F, Ma S. Fucoidan: a promising agent for brain injury and neurodegenerative disease intervention. Food Funct. 2021; 12:3820–3830. PMID: 33861265.12. Narayani SS, Saravanan S, Ravindran J, Ramasamy MS, Chitra J. In vitro anticancer activity of fucoidan extracted from Sargassum cinereum against Caco-2 cells. Int J Biol Macromol. 2019; 138:618–628. PMID: 31344415.13. Atashrazm F, Lowenthal RM, Woods GM, Holloway AF, Dickinson JL. Fucoidan and cancer: a multifunctional molecule with anti-tumor potential. Mar Drugs. 2015; 13:2327–2346. PMID: 25874926.14. Woonnoi W, Chotphruethipong L, Tanasawet S, Benjakul S, Sutthiwong N, Sukketsiri W. Hydrolyzed collagen from salmon skin increases the migration and filopodia formation of skin keratinocytes by activation of FAK/Src pathway. Pol J Food Nutr Sci. 2021; 71:323–332.15. Sohretoglu D, Huang S. Ganoderma lucidum polysaccharides as an anti-cancer agent. Anticancer Agents Med Chem. 2018; 18:667–674. PMID: 29141563.16. Yang JJ, Wang YH, Yin J, Leng H, Shen SD. Polysaccharides from Ulva prolifera O.F. Müller inhibit cell proliferation via activating MAPK signaling in A549 and H1650 cells. Food Funct. 2021; 12:6915–6924. PMID: 34132294.17. Lee H, Kim JS, Kim E. Fucoidan from seaweed Fucus vesiculosus inhibits migration and invasion of human lung cancer cell via PI3K-Akt-mTOR pathways. PLoS One. 2012; 7:e50624. PMID: 23226337.18. Ryu MJ, Chung HS. Anti-inflammatory activity of fucoidan with blocking NF-kappa B and STAT1 in human keratinocytes cells. Nat Prod Sci. 2015; 21:205–209.19. Sung CJ, Wang HH, Sun KH, Hsieh CC, Huang R, Sun GH, Tang SJ. Fucoidan from Sargassum hemiphyllum inhibits the stemness of cancer stem cells and epithelial-mesenchymal transitions in bladder cancer cells. Int J Biol Macromol. 2022; 221:623–633. PMID: 36099992.20. Ho CH, Chen ML, Huang HL, Lai CJ, Liu CH, Chuu CP, Lin YH. Active targeting of P-selectin by fucoidan modulates the molecular profiling of metastasis in docetaxel-resistant prostate cancer. Mar Drugs. 2022; 20:542. PMID: 36135731.21. Luo J, Li L, Zhu Z, Chang B, Deng F, Wang D, Lu X, Zuo D, Chen Q, Zhou J. Fucoidan inhibits EGFR redistribution and potentiates sorafenib to overcome sorafenib-resistant hepatocellular carcinoma. Biomed Pharmacother. 2022; 154:113602. PMID: 36029544.22. Kciuk M, Gielecińska A, Budzinska A, Mojzych M, Kontek R. Metastasis and MAPK pathways. Int J Mol Sci. 2022; 23:3847. PMID: 35409206.23. Sulzmaier FJ, Jean C, Schlaepfer DD. FAK in cancer: mechanistic findings and clinical applications. Nat Rev Cancer. 2014; 14:598–610. PMID: 25098269.24. Jia XB, Zhang Q, Xu L, Yao WJ, Wei L. Lotus leaf flavonoids induce apoptosis of human lung cancer A549 cells through the ROS/p38 MAPK pathway. Biol Res. 2021; 54:7. PMID: 33653412.25. Zou T, Mao X, Yin J, Li X, Chen J, Zhu T, Li Q, Zhou H, Liu Z. Emerging roles of RAC1 in treating lung cancer patients. Clin Genet. 2017; 91:520–528. PMID: 27790713.26. Kyykallio H, Oikari S, Bueno Álvez M, Gallardo Dodd CJ, Capra J, Rilla K. The density and length of filopodia associate with the activity of hyaluronan synthesis in tumor cells. Cancers (Basel). 2020; 12:1908. PMID: 32679746.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Overview of ALK and ROS1 Rearranged Lung Cancer
- Molecular Basis of Drug Resistance: Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitors and Anaplastic Lymphoma Kinase Inhibitors
- Luteolin attenuates migration and invasion of lung cancer cells via suppressing focal adhesion kinase and non-receptor tyrosine kinase signaling pathway
- Systemic Nocardiosis Mimicking Disease Flare-up after Discontinuation of Gefitinib in a Patient with EGFR-Mutant Lung Cancer
- Inhibition of Ubiquitin-specific Peptidase 8 Suppresses Growth of Gefitinib-resistant Non-small Cell Lung Cancer Cells by Inducing Apoptosis