Korean J healthc assoc Infect Control Prev.
2023 Jun;28(1):92-98. 10.14192/kjicp.2023.28.1.92.
The Difference in Anti-nucleocapsid Protein Antibody Responses Between Vaccinated and Unvaccinated Individuals After Asymptomatic, Mild, or Moderate COVID-19 Infection
- Affiliations
-
- 1Department of Infectious Diseases, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- KMID: 2543139
- DOI: http://doi.org/10.14192/kjicp.2023.28.1.92
Abstract
- Background
Anti-nucleocapsid protein IgG antibody (N-IgG) responses are not elicited by the current COVID-19 vaccines; therefore, these responses have been used to determine previous COVID-19 infections. However, data on whether COVID-19 vaccination affects the seroconversion of the N-IgG response are limited. This study aimed to compare the seropositivity of N-IgG responses in vaccinated individuals versus unvaccinated individuals with COVID-19 confirmed by polymerase chain reaction (PCR).
Methods
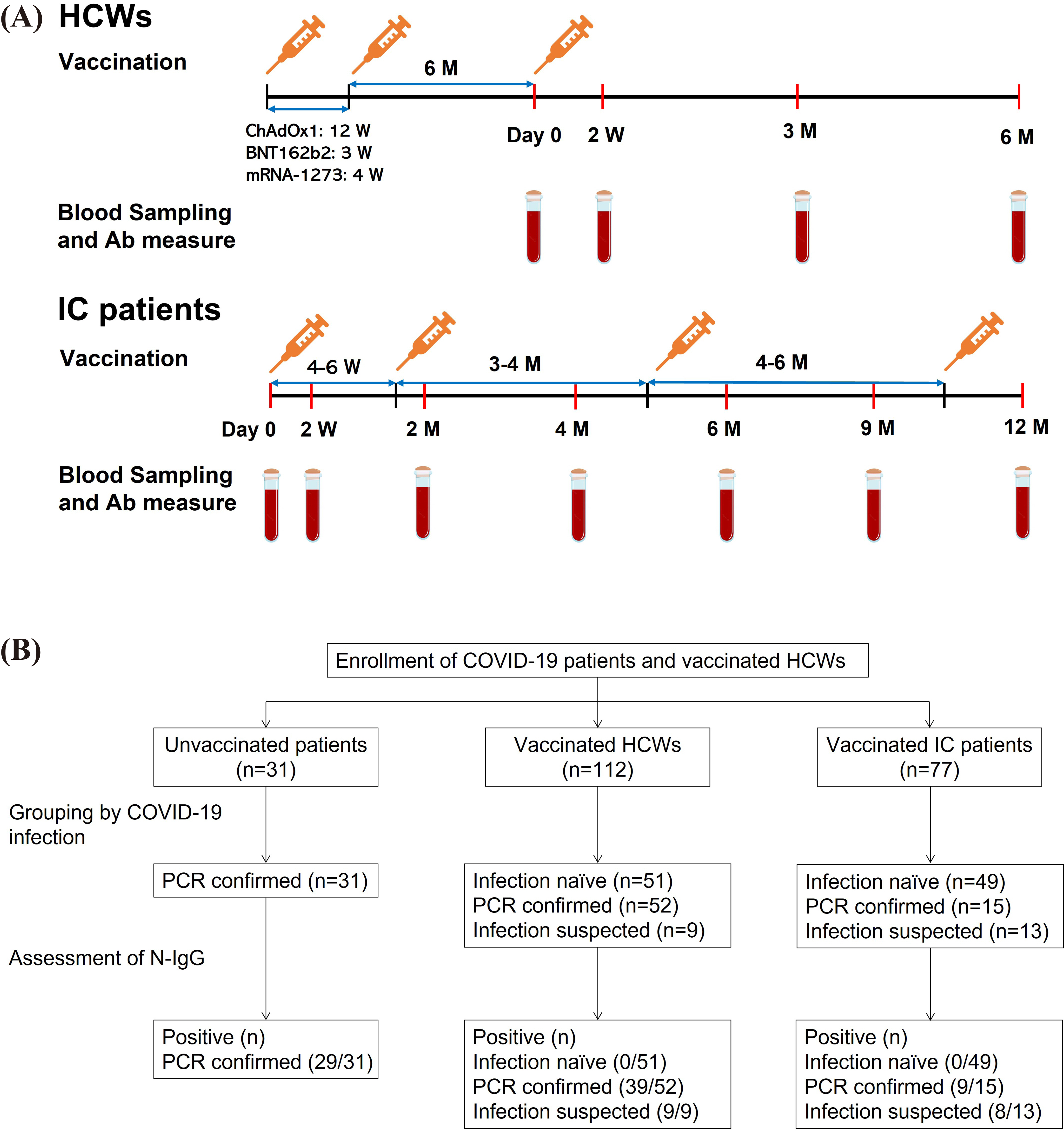
The study included healthcare workers (HCWs) and immunocompromised (IC) patients with liver or kidney transplants, regardless of their COVID-19 infection status, who have received COVID-19 vaccines (ChAdOx1, BNT162b2, or mRNA-1273) between March 2021 and December 2021. We also enrolled unvaccinated patients infected with COVID-19, who were asymptomatic or had mild or moderate symptoms. Anti-spike 1 protein IgG antibody (S1-IgG) and N-IgG responses were measured in plasma obtained from the participants.
Results
None of the 100 individuals (51 HCWs and 49 IC patients) without Severe Acute Respiratory Syndrome-Coronavirus-2 (SARS-CoV-2) infection demonstrated a positive N-IgG response. Among the patients with PCR-confirmed SARS-CoV-2 infection, the rate of N-IgG positivity was 93.5% (29/31) in the unvaccinated patients, which was significantly higher than that in the vaccinated HCWs (75.0% [39/52], P=0.04). The positive rate of N-IgG in vaccinated IC patients was numerically lower (60.0% [9/15]) than that in vaccinated HCWs; however, this difference was not statistically significant (P=0.33).
Conclusion
COVID-19 vaccination lowered the seroconversion rate of N-IgG in patients with COVID-19. Therefore, the estimate of SARS-CoV-2 infection based on the N-IgG response may underestimate the seroprevalence of SARS-CoV-2 infection in highly vaccinated populations.
Figure
Reference
-
1. Smits VAJ, Hernández-Carralero E, Paz-Cabrera MC, Cabrera E, Hernández-Reyes Y, Hernández-Fernaud JR, et al. 2021; The Nucleocapsid protein triggers the main humoral immune response in COVID-19 patients. Biochem Biophys Res Commun. 543:45–9. DOI: 10.1016/j.bbrc.2021.01.073. PMID: 33515911. PMCID: PMC7825866.
Article2. Lin L, Luo S, Qin R, Yang M, Wang X, Yang Q, et al. 2020; Long-term infection of SARS-CoV-2 changed the body's immune status. Clin Immunol. 218:108524. DOI: 10.1016/j.clim.2020.108524. PMID: 32659373. PMCID: PMC7351676.
Article3. Šimánek V, Pecen L, Řezáčková H, Topolčan O, Fajfrlík K, Sedláček D, et al. 2021; Long-term monitoring of the antibody response to a SARS-CoV-2 infection. Diagnostics (Basel). 11:1915. DOI: 10.3390/diagnostics11101915. PMID: 34679613. PMCID: PMC8534661.
Article4. Gallais F, Gantner P, Bruel T, Velay A, Planas D, Wendling MJ, et al. 2021; Evolution of antibody responses up to 13 months after SARS-CoV-2 infection and risk of reinfection. EBioMedicine. 71:103561. DOI: 10.1016/j.ebiom.2021.103561. PMID: 34455390. PMCID: PMC8390300.
Article5. Follmann D, Janes HE, Buhule OD, Zhou H, Girard B, Marks K, et al. 2022; Antinucleocapsid antibodies after SARS-CoV-2 infection in the blinded phase of the randomized, placebo-controlled mRNA-1273 COVID-19 vaccine efficacy clinical trial. Ann Intern Med. 175:1258–65. DOI: 10.7326/M22-1300. PMID: 35785530. PMCID: PMC9258784.
Article6. Our World in Data. Coronavirus (COVID-19) vaccinations. https://ourworldindata.org/covid-vaccinations. (Updated on 19 December 2022).7. Korea Disease Control. COVID-19. https://m.news.naver.com/covid19/index. (Updated on 19 December 2022).8. Kim JY, Kwon JS, Bae S, Cha HH, Lim JS, Kim MC, et al. 2021; SARS-CoV-2-specific antibody and T cell response kinetics according to symptom severity. Am J Trop Med Hyg. 105:395–400. DOI: 10.4269/ajtmh.20-1594. PMID: 34143752. PMCID: PMC8437179.
Article9. Kim JY, Lim SY, Park S, Kwon JS, Bae S, Park JY, et al. 2022; Immune responses to the ChAdOx1 nCoV-19 and BNT162b2 vaccines and to natural coronavirus disease 2019 infections over a 3-month period. J Infect Dis. 225:777–84. DOI: 10.1093/infdis/jiab579. PMID: 34850034. PMCID: PMC8767884.
Article10. Lee SM, Kim IS, Lim S, Lee SJ, Kim WJ, Shin KH, et al. 2021; Comparison of serologic response of hospitalized COVID-19 patients using 8 immunoassays. J Korean Med Sci. 36:e64. DOI: 10.3346/jkms.2021.36.e64. PMID: 33686810. PMCID: PMC7940118.
Article11. Yang L, Xu Q, Yang B, Li J, Dong R, Da J, et al. 2021; IgG antibody titers against SARS-CoV-2 nucleocapsid protein correlate with the severity of COVID-19 patients. BMC Microbiol. 21:351. DOI: 10.1186/s12866-021-02401-0. PMID: 34922455. PMCID: PMC8683808.
Article12. Feikin DR, Higdon MM, Abu-Raddad LJ, Andrews N, Araos R, Goldberg Y, et al. 2022; Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: results of a systematic review and meta-regression. Lancet. 399:924–44. DOI: 10.1016/S0140-6736(22)00152-0. PMID: 35202601. PMCID: PMC8863502.
Article13. Kim JA, Kim YY, Kim RK, Lee SJ, Park YJ, Yeom HS, et al. 2021; COVID -19 vaccine effectiveness on severity and death from May 2021 to July 2021. Public Health Wkly Rep. 14:2612–5.14. Guthmiller JJ, Stovicek O, Wang J, Changrob S, Li L, Halfmann P, et al. 2021; SARS-CoV-2 infection severity is linked to superior humoral immunity against the spike. mBio. 12:e02940–20. DOI: 10.1128/mBio.02940-20. PMID: 33468695. PMCID: PMC7845638.
Article15. Sughayer MA, Souan L, Abu Alhowr MM, Al Rimawi D, Siag M, Albadr S, et al. 2022; Comparison of the effectiveness and duration of anti-RBD SARS-CoV-2 IgG antibody response between different types of vaccines: implications for vaccine strategies. Vaccine. 40:2841–7. DOI: 10.1016/j.vaccine.2022.03.069. PMID: 35397946. PMCID: PMC8971065.
Article16. Allen N, Brady M, Martin AIC, Domegan L, Walsh C, Doherty L, et al. 2021; Serological markers of SARS-CoV-2 infection; anti-nucleocapsid antibody positivity may not be the ideal marker of natural infection in vaccinated individuals. J Infect. 83:e9–10. DOI: 10.1016/j.jinf.2021.08.012. PMID: 34384812. PMCID: PMC8351117.
Article17. Jung J, Kim JY, Kwon JS, Yun SC, Kim SH. 2022; Comparison of waning immunity between booster vaccination and 2-dose vaccination with BNT162b2. Immune Netw. 22:e31. DOI: 10.4110/in.2022.22.e31. PMID: 36081526. PMCID: PMC9433190.
Article18. PCL. POCT platform. PCLOK II. http://www.pclchip.com/en/sub02.php?page=02. (Updated on date Mon 2022).19. Søraas A, Grødeland G, Granerud BK, Ueland T, Lind A, Fevang B, et al. 2022; Breakthrough infections with the omicron and delta variants of SARS-CoV-2 result in similar re-activation of vaccine-induced immunity. Front Immunol. 13:964525. DOI: 10.3389/fimmu.2022.964525. PMID: 36159859. PMCID: PMC9493489.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- COVID-19 infection and severe clinical outcomes in patients with kidney disease by vaccination status: a nationwide cohort study in Korea
- Clinical Utility of Sero-Immunological Responses Against SARS-CoV-2 Nucleocapsid Protein During Subsequent Prevalence of Wild-Type, Delta Variant, and Omicron Variant
- A Study on The Efficacy of Vaccination in Elderly Patients with Breakthrough COVID-19 Infection
- Antibody production after SARS-CoV2 vaccination and COVID-19 incidence in liver transplant recipients
- Predictors of moderate, severe, and critical COVID-19 infection in a largely vaccinated kidney transplant recipient cohort during the Omicron era: the importance of timely booster vaccinations and early presentation to care