J Korean Med Sci.
2023 Jun;38(22):e170. 10.3346/jkms.2023.38.e170.
A Nationwide Survey of mRNA COVID-19 Vaccinee’s Experiences on Adverse Events and Its Associated Factors
- Affiliations
-
- 1School of Pharmacy, Sungkyunkwan University, Suwon, Koreav
- 2School of Pharmacy, Jeonbuk National University, Jeonju, Korea
- 3Department of Pediatrics, Korea University Anam Hospital, Seoul, Korea
- 4Department of Preventive Medicine, Korea University College of Medicine, Seoul, Korea
- 5Division of Life Sciences, Korea University, Seoul, Korea
- 6Department of Preventive Medicine, Gachon University College of Medicine, Incheon, Korea
- 7Department of Clinical Research Design & Evaluation, Samsung Advanced Institute for Health Sciences & Technology (SAIHST), Sungkyunkwan University, Seoul, Korea
- 8Department of Biohealth Regulatory Science, School of Pharmacy, Sungkyunkwan University, Suwon, Korea
- KMID: 2542802
- DOI: http://doi.org/10.3346/jkms.2023.38.e170
Abstract
- Background
Although coronavirus disease 2019 (COVID-19) vaccines have been distributed worldwide under emergency use authorization, the real-world safety profiles of mRNA vaccines still need to be clearly defined. We aimed to identify the overall incidence and factors associated with adverse events (AEs) following mRNA COVID-19 vaccination.
Methods
We conducted web-based survey from December 2 to 10 in 2021 with a 2,849 nationwide sampled panel. Study participants were individuals who had elapsed at least twoweeks after completing two dosing schedules of COVID-19 vaccination aged between 18–49 years. We weighted the participants to represent the Korean population. The outcome was the overall incidence of AEs following mRNA COVID-19 vaccination and associated factors. We estimated the weighted odds ratios (ORs) using multivariable logistic regression models to identify the factors associated with AEs.
Results
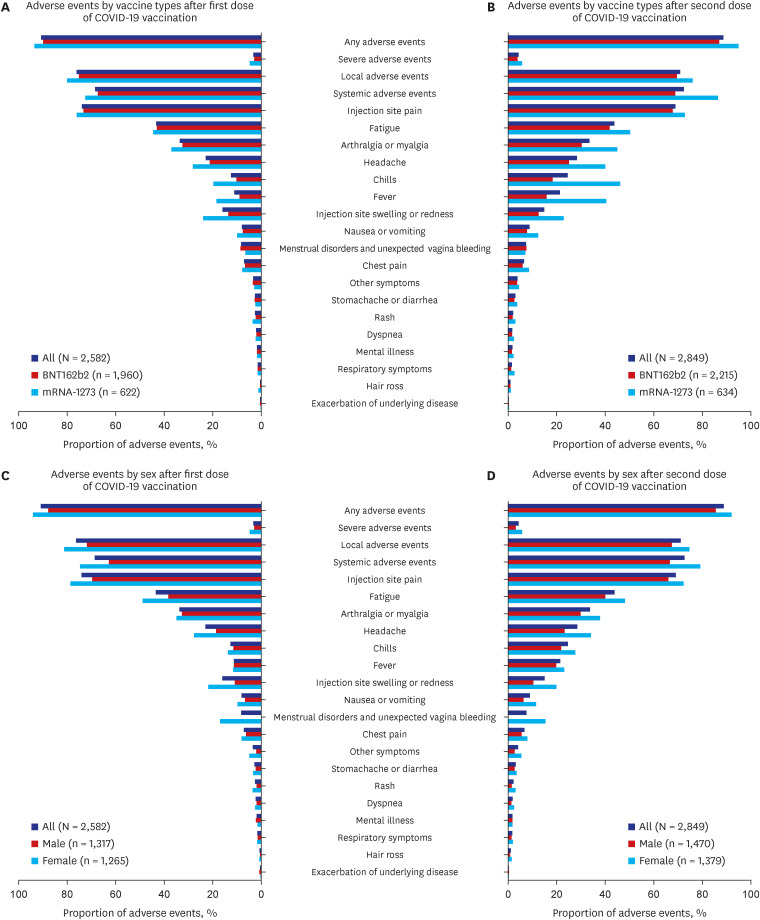
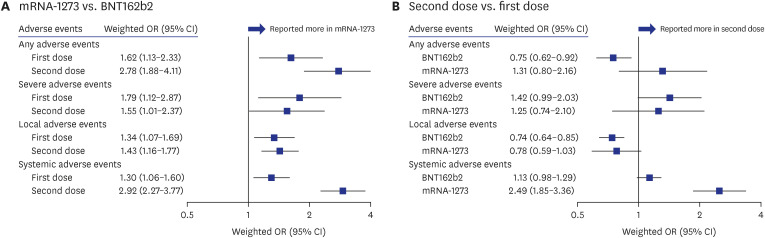
Of the 2,849 participants (median [interquartile range] age, 35 [27–42] years; 51.6% male), 90.8% (n = 2,582) for the first dose and 88.7% (n = 2,849) for the second dose reported AEs, and 3.3% and 4.3% reported severe AEs, respectively. Occurrence of AEs was more prevalent in mRNA-1273 (OR, 2.06; 95% confidence interval [CI], 1.59–2.67 vs. BNT162b2), female sex (1.88; 1.52–2.32), and those with dermatologic diseases (2.51; 1.32–4.77). History of serious allergic reactions (1.96; 1.06–3.64) and anticoagulant medication use (4.72; 1.92–11.6) were associated with severe AEs.
Conclusion
Approximately 90% of participants reported AEs following mRNA COVID-19 vaccination. Substantial factors, including vaccine type (mRNA-1273), female sex, and dermatologic diseases were associated with AEs. Our findings could aid policymakers in establishing vaccination strategies tailored to those potentially susceptible to AEs.
Keyword
Figure
Cited by 1 articles
-
Effective Vaccination and Education Strategies for Emerging Infectious Diseases Such as COVID-19
Seong-Heon Wie, Jaehun Jung, Woo Joo Kim
J Korean Med Sci. 2023;38(44):e371. doi: 10.3346/jkms.2023.38.e371.
Reference
-
1. World Health Organization. Coronavirus disease (COVID-19): vaccines. Updated 2022. Accessed February 10, 2022. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/question-and-answers-hub/q-a-detail/coronavirus-disease-(covid-19)-vaccines?adgroupsurvey={adgroupsurvey}&gclid=CjwKCAiA6Y2QBhAtEiwAGHybPbMrq-qL1RpdEBxT17XsyyFYFdttOUBz5va1mZb0vd12K96Ke_9eVxoCYvQQAvD_BwE .2. Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N Engl J Med. 2020; 383(27):2603–2615. PMID: 33301246.
Article3. Baden LR, El Sahly HM, Essink B, Kotloff K, Frey S, Novak R, et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2021; 384(5):403–416. PMID: 33378609.
Article4. Korea Disease Control and Prevention Agency. COVID-19 vaccination: daily updates by vaccine types. Updated 2022. Accessed July 22, 2022. http://www.xn--19-9n4ip0xd1egzrilds0a816b.kr/ .5. Pardi N, Hogan MJ, Porter FW, Weissman D. mRNA vaccines - a new era in vaccinology. Nat Rev Drug Discov. 2018; 17(4):261–279. PMID: 29326426.
Article6. Leo LU. U.S. FDA proposes shift to annual COVID vaccine shots. Updated 2023. Accessed January 23, 2023. https://www.reuters.com/world/us/us-fda-proposes-simplify-covid-vaccine-strategy-2023-01-23/ .7. Barda N, Dagan N, Ben-Shlomo Y, Kepten E, Waxman J, Ohana R, et al. Safety of the BNT162b2 mRNA COVID-19 vaccine in a nationwide setting. N Engl J Med. 2021; 385(12):1078–1090. PMID: 34432976.
Article8. Chen G, Li X, Sun M, Zhou Y, Yin M, Zhao B, et al. COVID-19 mRNA vaccines are generally safe in the short term: a vaccine vigilance real-world study says. Front Immunol. 2021; 12:669010. PMID: 34093567.
Article9. Menni C, Klaser K, May A, Polidori L, Capdevila J, Louca P, et al. Vaccine side-effects and SARS-CoV-2 infection after vaccination in users of the COVID Symptom Study app in the UK: a prospective observational study. Lancet Infect Dis. 2021; 21(7):939–949. PMID: 33930320.
Article10. Beatty AL, Peyser ND, Butcher XE, Cocohoba JM, Lin F, Olgin JE, et al. Analysis of COVID-19 vaccine type and adverse effects following vaccination. JAMA Netw Open. 2021; 4(12):e2140364. PMID: 34935921.
Article11. Choi YY, Kim MK, Kwon HC, Kim GH. Safety monitoring after the BNT162b2 COVID-19 vaccine among adults aged 75 years or older. J Korean Med Sci. 2021; 36(45):e318. PMID: 34811980.
Article12. Kwon Y, Hwang I, Ko M, Kim H, Kim S, Seo SY, et al. Self-reported adverse events following the second dose of COVID-19 Vaccines in the Republic of KOREA: recipient survey, February to December 2021. Epidemiol Health. 2022; e2023006. PMID: 37183320.
Article13. Our World in Data. Coronavirus (COVID-19) vaccinations. Updated 2022. Accessed September 7, 2022. https://ourworldindata.org/covid-vaccinations .14. WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004; 363(9403):157–163. PMID: 14726171.15. Pitt SC, Schwartz TA, Chu D. AAPOR reporting guidelines for survey studies. JAMA Surg. 2021; 156(8):785–786. PMID: 33825811.
Article16. Klein NP, Lewis N, Goddard K, Fireman B, Zerbo O, Hanson KE, et al. Surveillance for adverse events after COVID-19 mRNA vaccination. JAMA. 2021; 326(14):1390–1399. PMID: 34477808.
Article17. Hwang I, Park K, Kim TE, Kwon Y, Lee YK. COVID-19 vaccine safety monitoring in Republic of Korea from February 26, 2021 to October 31, 2021. Osong Public Health Res Perspect. 2021; 12(6):396–402. PMID: 34965689.
Article18. Kitagawa H, Kaiki Y, Sugiyama A, Nagashima S, Kurisu A, Nomura T, et al. Adverse reactions to the BNT162b2 and mRNA-1273 mRNA COVID-19 vaccines in Japan. J Infect Chemother. 2022; 28(4):576–581. PMID: 35058126.
Article19. Valera-Rubio MM, Sierra-Torres MI, Castillejo García RR, Cordero-Ramos JJ, López-Márquez MR, Cruz-Salgado ÓO, et al. Adverse events reported after administration of BNT162b2 and mRNA-1273 COVID-19 vaccines among hospital workers: a cross-sectional survey-based study in a Spanish hospital. Expert Rev Vaccines. 2022; 21(4):533–540. PMID: 34986076.
Article20. Chapin-Bardales J, Gee J, Myers T. Reactogenicity following receipt of mRNA-based COVID-19 vaccines. JAMA. 2021; 325(21):2201–2202. PMID: 33818592.
Article21. Borobia AM, Carcas AJ, Pérez-Olmeda M, Castaño L, Bertran MJ, García-Pérez J, et al. Immunogenicity and reactogenicity of BNT162b2 booster in ChAdOx1-S-primed participants (CombiVacS): a multicentre, open-label, randomised, controlled, phase 2 trial. Lancet. 2021; 398(10295):121–130. PMID: 34181880.22. Hillus D, Schwarz T, Tober-Lau P, Vanshylla K, Hastor H, Thibeault C, et al. Safety, reactogenicity, and immunogenicity of homologous and heterologous prime-boost immunisation with ChAdOx1 nCoV-19 and BNT162b2: a prospective cohort study. Lancet Respir Med. 2021; 9(11):1255–1265. PMID: 34391547.
Article23. Liu X, Shaw RH, Stuart AS, Greenland M, Aley PK, Andrews NJ, et al. Safety and immunogenicity of heterologous versus homologous prime-boost schedules with an adenoviral vectored and mRNA COVID-19 vaccine (Com-COV): a single-blind, randomised, non-inferiority trial. Lancet. 2021; 398(10303):856–869. PMID: 34370971.24. Parés-Badell O, Martínez-Gómez X, Pinós L, Borras-Bermejo B, Uriona S, Otero-Romero S, et al. Local and systemic adverse reactions to mRNA COVID-19 vaccines comparing two vaccine types and occurrence of previous COVID-19 infection. Vaccines (Basel). 2021; 9(12):1463. PMID: 34960209.
Article25. Green MS, Peer V, Magid A, Hagani N, Anis E, Nitzan D. Gender differences in adverse events following the Pfizer-BioNTech COVID-19 vaccine. Vaccines (Basel). 2022; 10(2):233. PMID: 35214694.
Article26. Edelman A, Boniface ER, Benhar E, Han L, Matteson KA, Favaro C, et al. Association between menstrual cycle length and coronavirus disease 2019 (COVID-19) vaccination: A U.S. Cohort. Obstet Gynecol. 2022; 139(4):481–489. PMID: 34991109.
Article27. Tissot N, Brunel AS, Bozon F, Rosolen B, Chirouze C, Bouiller K. Patients with history of COVID-19 had more side effects after the first dose of COVID-19 vaccine. Vaccine. 2021; 39(36):5087–5090. PMID: 34332800.
Article28. Baldolli A, Michon J, Appia F, Galimard C, Verdon R, Parienti JJ. Tolerance of BNT162b2 mRNA COVI-19 vaccine in patients with a medical history of COVID-19 disease: a case control study. Vaccine. 2021; 39(32):4410–4413. PMID: 34210574.
Article29. Jeong JY, Han KH. Common Skin Diseases in Korea. Seoul, Korea: MDworld;2009.30. Imai K, Tanaka F, Kawano S, Esaki K, Arakawa J, Nishiyama T, et al. Incidence and risk factors of immediate hypersensitivity reactions and immunization stress-related responses with COVID-19 mRNA vaccine. J Allergy Clin Immunol Pract. 2022; 10(10):2667–2676.E10. PMID: 35953016.
Article31. Dages KN, Pitlick MM, Joshi AY, Park MA. Risk of allergic reaction in patients with atopic disease and recent coronavirus disease 2019 vaccination. Ann Allergy Asthma Immunol. 2021; 127(2):257–258. PMID: 33962029.
Article32. Heratizadeh A, Wichmann K, Werfel T. Food allergy and atopic dermatitis: how are they connected? Curr Allergy Asthma Rep. 2011; 11(4):284–291. PMID: 21614611.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Adverse events following vaccination against coronavirus disease 2019
- Adverse events and preventive measures related to COVID-19 vaccines
- A Case of Myocarditis Presenting With a Hyperechoic Nodule After the First Dose of COVID-19 mRNA Vaccine
- Comparison of Adverse Events of the First Dose and the Second Dose after Vaccination of the COVID-19 Pfizer Vaccine
- Ipsilateral Radial Neuropathy after COVID-19 mRNA Vaccination in an Immunocompetent Young Man