Korean J Transplant.
2023 Mar;37(1):49-56. 10.4285/kjt.22.0056.
Different antibody responses between liver and kidney transplant recipients elicited by third doses of COVID-19 mRNA vaccines
- Affiliations
-
- 1Department of Infectious Diseases, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- 2Division of Hepatobiliary Surgery and Liver Transplantation, Department of Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- 3Department of Convergence Medicine, Asan Medical Institute of Convergence Science and Technology (AMIST), Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- 4Division of Kidney and Pancreas Transplantation, Department of Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- KMID: 2541312
- DOI: http://doi.org/10.4285/kjt.22.0056
Abstract
- Background
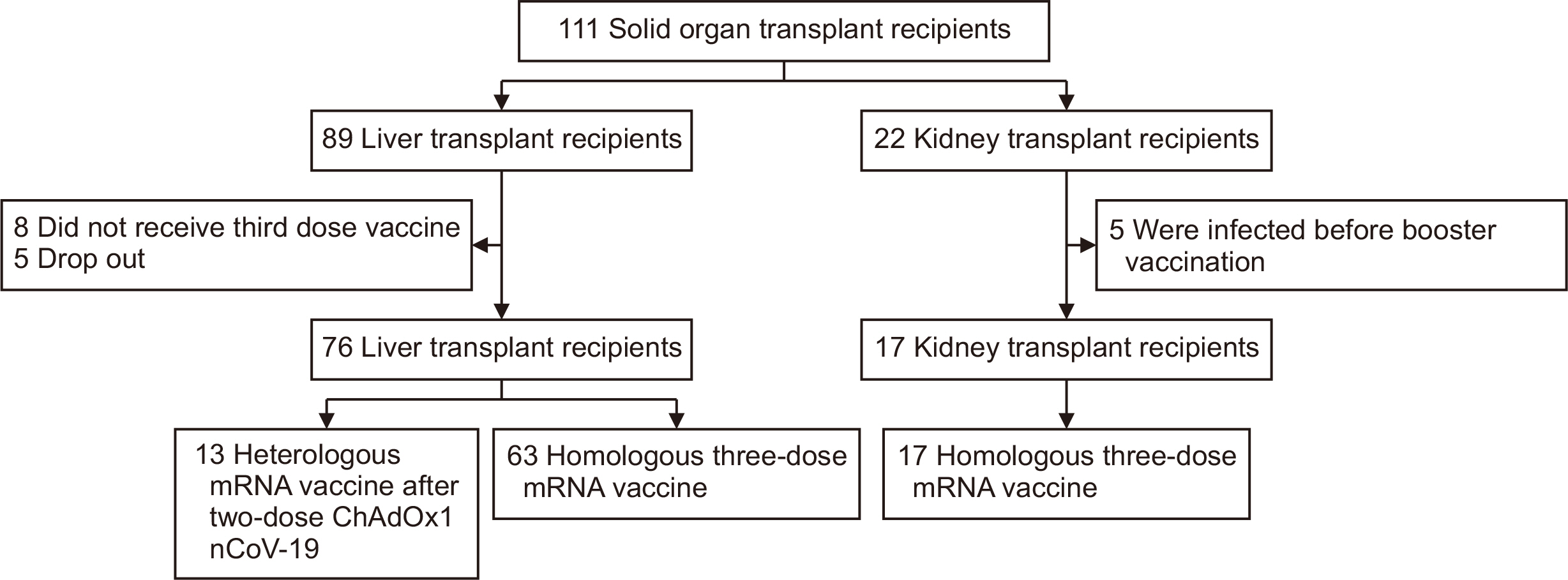
Solid organ transplant recipients exhibit decreased antibody responses, mainly due to their weakened immune systems. However, data are limited on antibody responses after the primary series of coronavirus disease 2019 (COVID-19) vaccines among recipients of various solid organ transplant types. Thus, we compared the antibody responses after three COVID-19 vaccine doses between liver transplant (LT) and kidney transplant (KT) recipients.
Methods
We prospectively enrolled solid organ transplant recipients who received three COVID-19 vaccine doses from June 2021 to February 2022 and measured S1-specific immunoglobulin G antibodies using an enzyme-linked immunosorbent assay.
Results
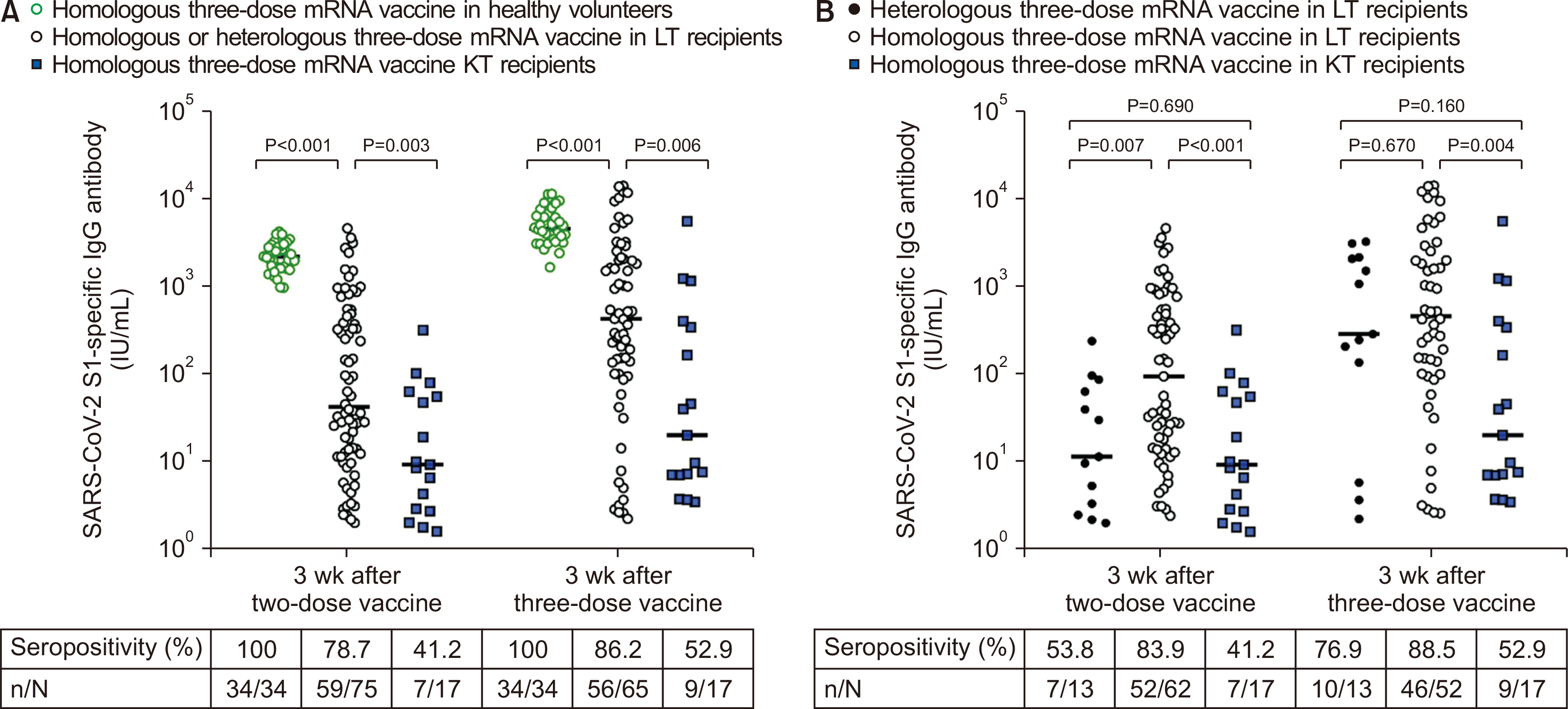
Seventy-six LT and 17 KT recipients were included in the final analysis. KT recipients showed consistently lower antibody responses even after the third vaccine dose (86.2% vs. 52.9%, P=0.008) and lower antibody titers (median, 423.0 IU/mL [interquartile range, 99.6–2,057 IU/mL] vs. 19.7 IU/mL [interquartile range, 6.9–339.4 IU/mL]; P=0.006) than were observed in LT recipients. Mycophenolic acid was a significant risk factor for a seropositive antibody response after the third vaccine dose in the multivariable analysis (odds ratio, 0.06; 95% confidence interval, 0.00–0.39; P=0.02).
Conclusions
We found a weaker antibody response despite the completion of the primary series of COVID-19 vaccines in KT recipients than in LT recipients. Mycophenolic acid use in KT recipients might be the main contributor to this observation.
Keyword
Figure
Reference
-
1. Boyarsky BJ, Werbel WA, Avery RK, Tobian AA, Massie AB, Segev DL, et al. 2021; Antibody response to 2-dose SARS-CoV-2 mRNA vaccine series in solid organ transplant recipients. JAMA. 325:2204–6. DOI: 10.1001/jama.2021.7489. PMID: 33950155. PMCID: PMC8100911.2. Naylor KL, Kim SJ, Smith G, McArthur E, Kwong JC, Dixon SN, et al. 2022; Effectiveness of first, second, and third COVID-19 vaccine doses in solid organ transplant recipients: a population-based cohort study from Canada. Am J Transplant. 22:2228–36. DOI: 10.1111/ajt.17095. PMID: 35578576. PMCID: PMC9347443.3. Barda N, Dagan N, Cohen C, Hernán MA, Lipsitch M, Kohane IS, et al. 2021; Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: an observational study. Lancet. 398:2093–100. DOI: 10.1016/S0140-6736(21)02249-2. PMID: 34756184.4. Tartof SY, Slezak JM, Fischer H, Hong V, Ackerson BK, Ranasinghe ON, et al. 2021; Effectiveness of mRNA BNT162b2 COVID-19 vaccine up to 6 months in a large integrated health system in the USA: a retrospective cohort study. Lancet. 398:1407–16. DOI: 10.1016/S0140-6736(21)02183-8. PMID: 34619098.5. Centers for Disease Control and Prevention (CDC). COVID-19 vaccines for people who are moderately or severely immunocompromised [Internet]. CDC;2022. cited 2022 Aug 17. Available from: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/recommendations/immuno.html.6. Caillard S, Thaunat O. 2021; COVID-19 vaccination in kidney transplant recipients. Nat Rev Nephrol. 17:785–7. DOI: 10.1038/s41581-021-00491-7. PMID: 34580488. PMCID: PMC8475856.7. Benotmane I, Gautier-Vargas G, Cognard N, Olagne J, Heibel F, Braun-Parvez L, et al. 2021; Low immunization rates among kidney transplant recipients who received 2 doses of the mRNA-1273 SARS-CoV-2 vaccine. Kidney Int. 99:1498–500. DOI: 10.1016/j.kint.2021.04.005. PMID: 33887315. PMCID: PMC8055921.8. Rozen-Zvi B, Yahav D, Agur T, Zingerman B, Ben-Zvi H, Atamna A, et al. 2021; Antibody response to SARS-CoV-2 mRNA vaccine among kidney transplant recipients: a prospective cohort study. Clin Microbiol Infect. 27:1173.e1–4. DOI: 10.1016/j.cmi.2021.04.028. PMID: 33957273. PMCID: PMC8091803.9. Cucchiari D, Egri N, Bodro M, Herrera S, Del Risco-Zevallos J, Casals-Urquiza J, et al. 2021; Cellular and humoral response after MRNA-1273 SARS-CoV-2 vaccine in kidney transplant recipients. Am J Transplant. 21:2727–39. DOI: 10.1111/ajt.16701. PMID: 34036720. PMCID: PMC8222867.10. Grupper A, Rabinowich L, Schwartz D, Schwartz IF, Ben-Yehoyada M, Shashar M, et al. 2021; Reduced humoral response to mRNA SARS-CoV-2 BNT162b2 vaccine in kidney transplant recipients without prior exposure to the virus. Am J Transplant. 21:2719–26. DOI: 10.1111/ajt.16615. PMID: 33866672. PMCID: PMC8250589.11. Nazaruk P, Monticolo M, Jędrzejczak AM, Krata N, Moszczuk B, Sańko-Resmer J, et al. 2021; Unexpectedly high efficacy of SARS-CoV-2 BNT162b2 vaccine in liver versus kidney transplant recipients-is it related to immunosuppression only? Vaccines (Basel). 9:1454. DOI: 10.3390/vaccines9121454. PMID: 34960200. PMCID: PMC8703477.12. Lim SY, Yoon YI, Kim JY, Tak E, Song GW, Kim SH, et al. 2022; Antibody response induced by two doses of ChAdOx1 nCoV-19, mRNA-1273, or BNT162b2 in liver transplant recipients. Immune Netw. 22:e24. DOI: 10.4110/in.2022.22.e24. PMID: 35799703. PMCID: PMC9250869.13. Karnell JL, Karnell FG 3rd, Stephens GL, Rajan B, Morehouse C, Li Y, et al. 2011; Mycophenolic acid differentially impacts B cell function depending on the stage of differentiation. J Immunol. 187:3603–12. DOI: 10.4049/jimmunol.1003319. PMID: 21873529. PMCID: PMC4180087.14. Rabinowich L, Grupper A, Baruch R, Ben-Yehoyada M, Halperin T, Turner D, et al. 2021; Low immunogenicity to SARS-CoV-2 vaccination among liver transplant recipients. J Hepatol. 75:435–8. DOI: 10.1016/j.jhep.2021.04.020. PMID: 33892006. PMCID: PMC8058047.15. Meunier L, Sanavio M, Dumortier J, Meszaros M, Faure S, Ursic Bedoya J, et al. 2022; Mycophenolate mofetil decreases humoral responses to three doses of SARS-CoV-2 vaccine in liver transplant recipients. Liver Int. 42:1872–8. DOI: 10.1111/liv.15258. PMID: 35338550. PMCID: PMC9115190.16. Sanders JF, Bemelman FJ, Messchendorp AL, Baan CC, van Baarle D, van Binnendijk R, et al. 2022; The RECOVAC immune-response study: the immunogenicity, tolerability, and safety of COVID-19 vaccination in patients with chronic kidney disease, on dialysis, or living with a kidney transplant. Transplantation. 106:821–34. DOI: 10.1097/TP.0000000000003983. PMID: 34753894. PMCID: PMC8942603.17. Macedo C, Hadi K, Walters J, Elinoff B, Marrari M, Zeevi A, et al. 2018; Impact of induction therapy on circulating T follicular helper cells and subsequent donor-specific antibody formation after kidney transplant. Kidney Int Rep. 4:455–69. DOI: 10.1016/j.ekir.2018.11.020. PMID: 30899873. PMCID: PMC6409398.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Antibody Response Induced by Two Doses of ChAdOx1 nCoV-19, mRNA-1273, or BNT162b2 in Liver Transplant Recipients
- Immunogenicity of SARS-CoV-2 vaccine in kidney transplant recipients: a cross-sectional study in Korea
- Letter regarding “COVID-19 vaccine immunogenicity among chronic liver disease patients and liver transplant recipients: A meta-analysis”
- Comparison of the rapidity of SARS-CoV-2 immune responses between primary and booster vaccination for COVID-19
- Antibody production after SARS-CoV2 vaccination and COVID-19 incidence in liver transplant recipients