J Liver Cancer.
2022 Sep;22(2):115-124. 10.17998/jlc.2022.04.20.
The dual role of transforming growth factor-beta signatures in human B viral multistep hepatocarcinogenesis: early and late responsive genes
- Affiliations
-
- 1Department of Pathology, Yonsei University College of Medicine, Seoul, Korea
- 2Natural Products Research Center, Korea Institute of Science and Technology (KIST), Gangneung, Korea
- KMID: 2534238
- DOI: http://doi.org/10.17998/jlc.2022.04.20
Abstract
- Background
/Aim: Transforming growth factor-beta (TGF-β) has a dichotomous role, functioning as a tumor suppressor and tumor promoter. TGF-β signatures, explored in mouse hepatocytes, have been reported to predict the clinical outcomes of hepatocellular carcinoma (HCC) patients; HCCs exhibiting early TGF-β signatures showed a better prognosis than those with late TGF-β signatures. The expression status of early and late TGF-β signatures remains unclear in defined lesions of human B-viral multistep hepatocarcinogenesis.
Methods
The expression of TGF-β signatures, early and late responsive signatures of TGF-β were investigated and analyzed for their correlation in cirrhosis, low-grade dysplastic nodules (DNs), high-grade DNs, early HCCs and progressed HCCs (pHCCs) by real-time PCR and immunohistochemistry.
Results
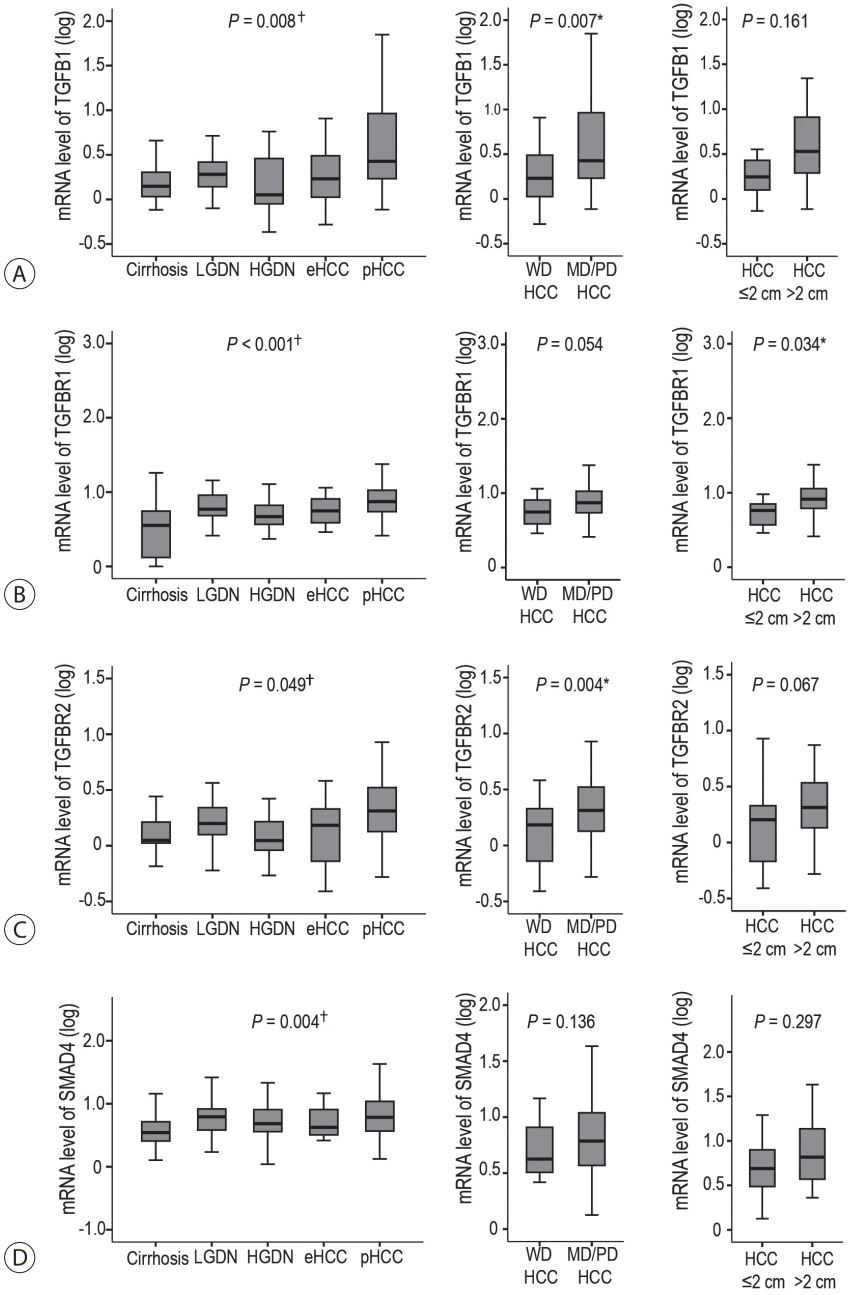
The expression levels of TGF-β signaling genes (TGFB1, TGFBR1, TGFBR2 and SMAD4) gradually increased with the progression of hepatocarcinogenesis, peaking in pHCCs. The expression of early responsive genes of TGF-β (GADD45B, FBP1, CYP1A2 and CYP3A4) gradually decreased, and that of the late TGF-β signatures (TWIST and SNAI1) significantly increased according to the progression of multistep hepatocarcinogenesis. Furthermore, mRNA levels of TWIST and SNAI1 were well correlated with those of stemness markers, with upregulation of TGF-β signaling, whereas FBP1 expression was inversely correlated with that of stemness markers.
Conclusions
The enrichment of the late responsive signatures of TGF-β with induction of stemness is considered to be involved in the progression of the late stage of multistep hepatocarcinogenesis, whereas the early responsive signatures of TGF-β are suggested to have tumor-suppressive roles in precancerous lesions of the early stage of multistep hepatocarcinogenesis.
Keyword
Figure
Reference
-
1. Massagué J. How cells read TGF-beta signals. Nat Rev Mol Cell Biol. 2000; 1:169–178.2. Coulouarn C, Factor VM, Thorgeirsson SS. Transforming growth factor-beta gene expression signature in mouse hepatocytes predicts clinical outcome in human cancer. Hepatology. 2008; 47:2059–2067.3. Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell. 2008; 133:704–715.4. Wellner U, Schubert J, Burk UC, Schmalhofer O, Zhu F, Sonntag A, et al. The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs. Nat Cell Biol. 2009; 11:1487–1495.5. Kim H, Choi GH, Na DC, Ahn EY, Kim GI, Lee JE, et al. Human hepatocellular carcinomas with "Stemness"-related marker expression: keratin 19 expression and a poor prognosis. Hepatology. 2011; 54:1707–1717.6. Rhee H, Nahm JH, Kim H, Choi GH, Yoo JE, Lee HS, et al. Poor outcome of hepatocellular carcinoma with stemness marker under hypoxia: resistance to transarterial chemoembolization. Mod Pathol. 2016; 29:1038–1049.7. Yoo JE, Kim YJ, Rhee H, Kim H, Ahn EY, Choi JS, et al. Progressive enrichment of stemness features and tumor stromal alterations in multistep hepatocarcinogenesis. PLoS One. 2017; 12:e0170465.8. Wan S, Zhao E, Kryczek I, Vatan L, Sadovskaya A, Ludema G, et al. Tumor-associated macrophages produce interleukin 6 and signal via STAT3 to promote expansion of human hepatocellular carcinoma stem cells. Gastroenterology. 2014; 147:1393–1404.9. The International Consensus Group for Hepatocellular Neoplasia. Pathologic diagnosis of early hepatocellular carcinoma: a report of the international consensus group for hepatocellular neoplasia. Hepatology. 2009; 49:658–664.10. Derynck R, Akhurst RJ, Balmain A. TGF-beta signaling in tumor suppression and cancer progression. Nat Genet. 2001; 29:117–129.11. Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006; 6:392–401.12. Yoo J, Ghiassi M, Jirmanova L, Balliet AG, Hoffman B, Fornace AJ Jr, et al. Transforming growth factor-beta-induced apoptosis is mediated by Smad-dependent expression of GADD45b through p38 activation. J Biol Chem. 2003; 278:43001–43007.13. Yang J, Wang C, Zhao F, Luo X, Qin M, Arunachalam E, et al. Loss of FBP1 facilitates aggressive features of hepatocellular carcinoma cells through the Warburg effect. Carcinogenesis. 2017; 38:134–143.14. Chen H, Shen ZY, Xu W, Fan TY, Li J, Lu YF, et al. Expression of P450 and nuclear receptors in normal and end-stage Chinese livers. World J Gastroenterol. 2014; 20:8681–8690.15. Ren J, Chen GG, Liu Y, Su X, Hu B, Leung BC, et al. Cytochrome P450 1A2 metabolizes 17β-estradiol to suppress hepatocellular carcinoma. PLoS One. 2016; 11:e0153863.16. Kar S, Wang M, Carr BI. 2-Methoxyestradiol inhibits hepatocellular carcinoma cell growth by inhibiting Cdc25 and inducing cell cycle arrest and apoptosis. Cancer Chemother Pharmacol. 2008; 62:831–840.17. Abdel-Razzak Z, Corcos L, Fautrel A, Campion JP, Guillouzo A. Transforming growth factor-beta 1 down-regulates basal and polycyclic aromatic hydrocarbon-induced cytochromes P-450 1A1 and 1A2 in adult human hepatocytes in primary culture. Mol Pharmacol. 1994; 46:1100–1110.18. Müller GF, Döhr O, El-Bahay C, Kahl R, Abel J. Effect of transforming growth factor-beta1 on cytochrome P450 expression: inhibition of CYP1 mRNA and protein expression in primary rat hepatocytes. Arch Toxicol. 2000; 74:145–152.19. Kobayashi M, Ikeda K, Hosaka T, Sezaki H, Someya T, Akuta N, et al. Dysplastic nodules frequently develop into hepatocellular carcinoma in patients with chronic viral hepatitis and cirrhosis. Cancer. 2006; 106:636–647.20. Borzio M, Fargion S, Borzio F, Fracanzani AL, Croce AM, Stroffolini T, et al. Impact of large regenerative, low grade and high grade dysplastic nodules in hepatocellular carcinoma development. J Hepatol. 2003; 39:208–214.21. Lee TK, Poon RT, Yuen AP, Ling MT, Kwok WK, Wang XH, et al. Twist overexpression correlates with hepatocellular carcinoma metastasis through induction of epithelial-mesenchymal transition. Clin Cancer Res. 2006; 12:5369–5376.22. Olmeda D, Jordá M, Peinado H, Fabra A, Cano A. Snail silencing effectively suppresses tumour growth and invasiveness. Oncogene. 2007; 26:1862–1874.23. Shan YF, Huang YL, Xie YK, Tan YH, Chen BC, Zhou MT, et al. Angiogenesis and clinicopathologic characteristics in different hepatocellular carcinoma subtypes defined by EpCAM and α-fetoprotein expression status. Med Oncol. 2011; 28:1012–1016.24. Yamashita T, Honda M, Nakamoto Y, Baba M, Nio K, Hara Y, et al. Discrete nature of EpCAM+ and CD90+ cancer stem cells in human hepatocellular carcinoma. Hepatology. 2013; 57:1484–1497.25. Dong C, Yuan T, Wu Y, Wang Y, Fan TW, Miriyala S, et al. Loss of FBP1 by Snail-mediated repression provides metabolic advantages in basal-like breast cancer. Cancer Cell. 2013; 23:316–331.26. Zhang J, Wang J, Xing H, Li Q, Zhao Q, Li J. Down-regulation of FBP1 by ZEB1-mediated repression confers to growth and invasion in lung cancer cells. Mol Cell Biochem. 2016; 411:331–340.27. Rhee H, Kim H, Park YN. Clinico-radio-pathological and molecular features of hepatocellular carcinomas with keratin 19 expression. Liver Cancer. 2020; 9:663–681.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Role of Transforming Growth Factor Alpha in Hepatocarcinogenesis
- Immunohistochemical Analysis of Transforming Growth Factor (TGF)-beta1 and TGF-beta Receptor II and Quantitative Analysis of TGF-beta1 mRNA during Multistep Hepatocarcinogenesis Induced by Diethylnitrosamine in Sprague-Dawley Rats
- Expressions of transforming growth factor beta in patients with rheumatioid arthritis and osteoarthritis
- Expression of TGF-beta1 and TGF-betatype II Receptor in Chemically Induced Hepatocarcinogenesis of the Rat
- Targeting the Transforming Growth Factor-beta Signaling in Cancer Therapy