J Korean Med Sci.
2022 Aug;37(34):e265. 10.3346/jkms.2022.37.e265.
A Case Report for Acute Myopericarditis After NVX-CoV2373 (Novavax®) COVID-19 Vaccination
- Affiliations
-
- 1Department of Cardiovascular Medicine, Chonnam National University Medical School/Hospital, Gwangju, Korea
- 2Department of Pathology, Chonnam National University Medical School, Gwangju, Korea
- KMID: 2532758
- DOI: http://doi.org/10.3346/jkms.2022.37.e265
Abstract
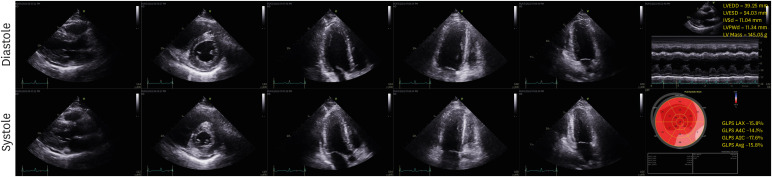
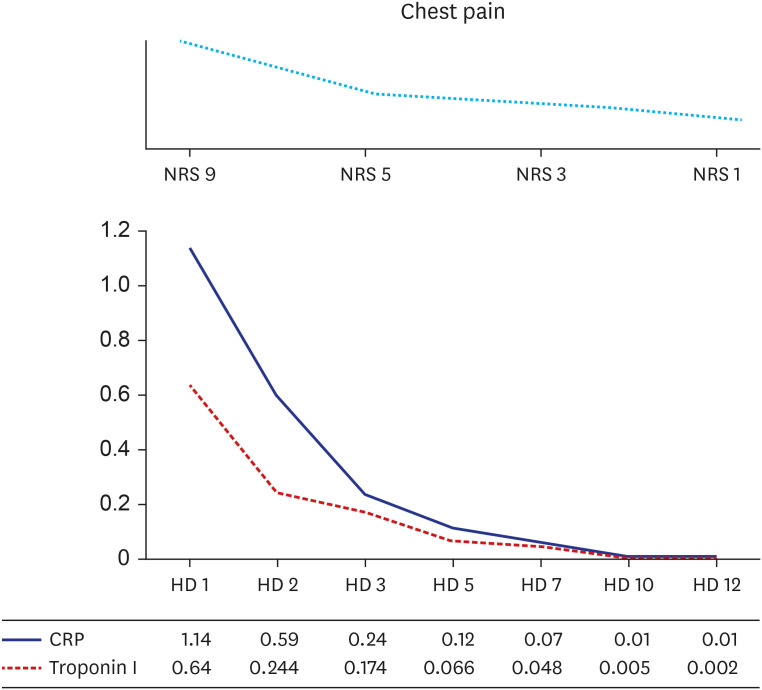
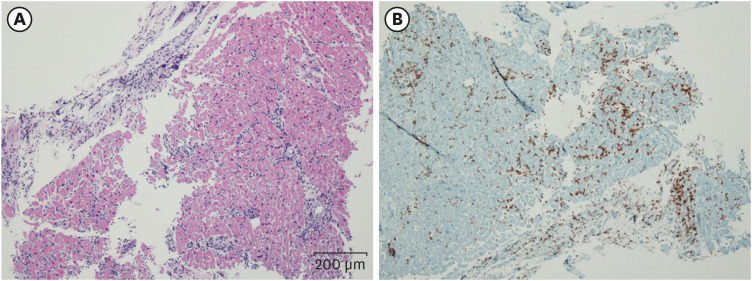
- Post-vaccination myocarditis after administration of the NVX-CoV2373 coronavirus disease 2019 (COVID-19) vaccine has been reported in a limited population. We report the first biopsy-proven case of myopericarditis after administration of second dose of NVX-CoV2373 COVID-19 vaccine (Novavax®) in Korea. A 30-year-old man was referred to emergency department with complaints of chest pain and mild febrile sense for two days. He received the second dose vaccine 17 days ago. Acute myopericarditis by the vaccination was diagnosed by cardiac endomyocardial biopsy. He was treated with corticosteroid 1 mg/kg/day for 5 days and tapered for one week. He successfully recovered and was discharged on the 12th day of hospitalization. The present case suggests acute myopericarditis as a vaccination complication by Novavax® in Korea.
Keyword
Figure
Reference
-
1. Sadoff J, Gray G, Vandebosch A, Cárdenas V, Shukarev G, Grinsztejn B, et al. Safety and efficacy of single-dose Ad26.COV2.S vaccine against Covid-19. N Engl J Med. 2021; 384(23):2187–2201. PMID: 33882225.
Article2. Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020; 383(27):2603–2615. PMID: 33301246.
Article3. Barda N, Dagan N, Ben-Shlomo Y, Kepten E, Waxman J, Ohana R, et al. Safety of the BNT162b2 mRNA Covid-19 vaccine in a nationwide setting. N Engl J Med. 2021; 385(12):1078–1090. PMID: 34432976.
Article4. Gargano JW, Wallace M, Hadler SC, Langley G, Su JR, Oster ME, et al. Use of mRNA COVID-19 vaccine after reports of myocarditis among vaccine recipients: update from the advisory committee on immunization practices - United States, June 2021. MMWR Morb Mortal Wkly Rep. 2021; 70(27):977–982. PMID: 34237049.
Article5. Abu Mouch S, Roguin A, Hellou E, Ishai A, Shoshan U, Mahamid L, et al. Myocarditis following COVID-19 mRNA vaccination. Vaccine. 2021; 39(29):3790–3793. PMID: 34092429.
Article6. Choi S, Lee S, Seo JW, Kim MJ, Jeon YH, Park JH, et al. Myocarditis-induced Sudden Death after BNT162b2 mRNA COVID-19 Vaccination in Korea: Case Report Focusing on Histopathological Findings. J Korean Med Sci. 2021; 36(40):e286. PMID: 34664804.
Article7. Kim D, Choi JH, Jang JY, So O, Cho E, Choi H, et al. A case report for myopericarditis after BNT162b2 COVID-19 mRNA vaccination in a Korean young male. J Korean Med Sci. 2021; 36(39):e277. PMID: 34636504.
Article8. Bautista García J, Peña Ortega P, Bonilla Fernández JA, Cárdenes León A, Ramírez Burgos L, Caballero Dorta E. Miocarditis aguda tras administración de vacuna BNT162b2 contra la COVID-19. Rev Esp Cardiol. 2021; 74(9):812–814.
Article9. Kim HW, Jenista ER, Wendell DC, Azevedo CF, Campbell MJ, Darty SN, et al. Patients with acute myocarditis following mRNA COVID-19 vaccination. JAMA Cardiol. 2021; 6(10):1196–1201. PMID: 34185046.
Article10. Montgomery J, Ryan M, Engler R, Hoffman D, McClenathan B, Collins L, et al. Myocarditis following immunization with mRNA COVID-19 Vaccines in Members of the US Military. JAMA Cardiol. 2021; 6(10):1202–1206. PMID: 34185045.
Article11. U.S. Food and Drug Administration. Novavax COVID-19 vaccine (NVX-CoV2373) VRBPAC briefing document. Updated 2022. Accessed June 7, 2022. https://www.fda.gov/media/158912/download .12. Heath PT, Galiza EP, Baxter DN, Boffito M, Browne D, Burns F, et al. Safety and efficacy of NVX-CoV2373 Covid-19 vaccine. N Engl J Med. 2021; 385(13):1172–1183. PMID: 34192426.
Article13. Hadi YB, Thakkar S, Shah-Khan SM, Hutson W, Sarwari A, Singh S. COVID-19 vaccination is safe and effective in patients with inflammatory bowel disease: analysis of a large multi-institutional research network in the United States. Gastroenterology. 2021; 161(4):1336–1339.e3. PMID: 34144044.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case Report for Myopericarditis after BNT162b2 COVID-19 mRNA Vaccination in a Korean Young Male
- A Case of Aphthous Stomatitis in a Healthy Adult Following COVID-19 Vaccination: Clinical Reasoning
- COVID-19 Vaccination in Korea
- Vestibular Neuritis after COVID-19 Vaccination
- A Generalized Lichen Planus Following COVID-19 Vaccination: A Case Report