Korean J Gastroenterol.
2022 Aug;80(2):85-92. 10.4166/kjg.2022.090.
Comparison of Two Types of 1-L Polyethylene Glycol-ascorbic Acid as Colonoscopic Bowel Preparation: A Prospective Randomized Study
- Affiliations
-
- 1Division of Gastroenterology, Department of Internal Medicine, Seoul Paik Hospital, Inje University College of Medicine, Seoul, Korea
- 2Department of Nursing, Seoul Paik Hospital, Inje University College of Medicine, Seoul, Korea
- 3Division of Gastroenterology, Department of Internal Medicine, Chung-Ang University College of Medicine, Seoul, Korea
- KMID: 2532683
- DOI: http://doi.org/10.4166/kjg.2022.090
Abstract
- Background/Aims
Recently, 1-L polyethylene glycol-ascorbic acid (PEG-Asc) has been used to reduce the volume of preparation agents in colonoscopy. This clinical trial aimed to compare the efficacy and safety of two types of 1-L PEG-Asc (CleanViewAL ® [Tae Joon Pharmaceutical Company, Seoul, Korea] and Plenvu ® [Norgine, Harefield, United Kingdom]) in average-aged adults.
Methods
This study was a prospective, randomized, non-inferiority, open-label, phase 4 clinical trial. The primary endpoint was the efficacy evaluated using the Boston bowel preparation scale (BBPS), and the secondary endpoint was clinical safety.
Results
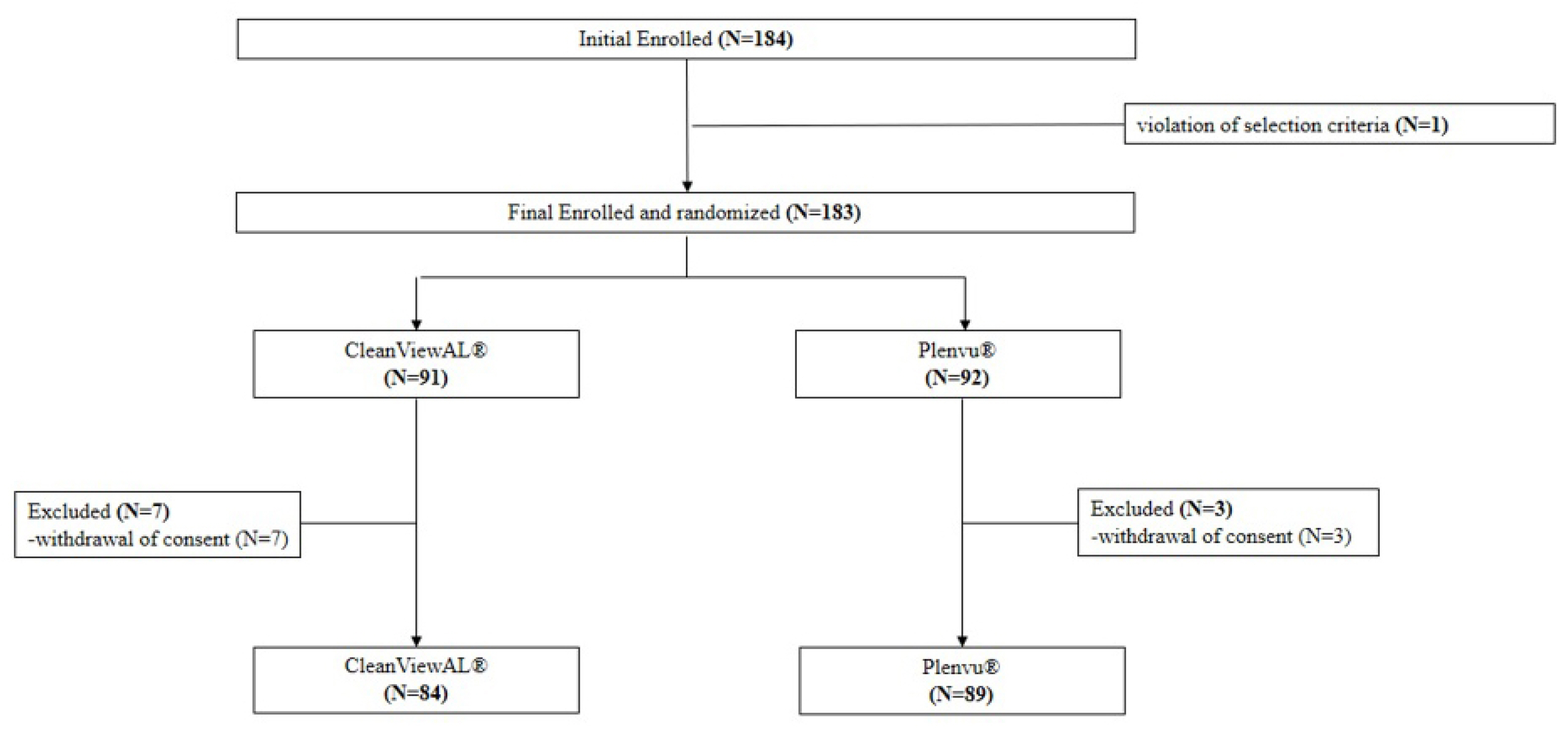
In total, 173 patients were assigned to either the CleanViewAL ® (n=84) or Plenvu ® (n=89) group. Overall cleansing successes of 97.6% (82/84) and 98.8% (88/89) were achieved in the CleanViewAL ® group and in the Plenvu ® group, respectively, showing that CleanViewAL ® has similar bowel cleansing efficacy to Plenvu ® (95% CI, -0.052 to 0.027; p=0.207). The total BBPS score was 8.67±1.00 and 8.70±0.76 in the CleanViewAL ® group and Plenvu ® group, respectively (p=0.869). The most common adverse symptom was nausea, and no adverse symptoms requiring hospitalization were reported in either group. There were no cases of critical hypernatremia and liver dysfunction exceeding the common terminology criteria for adverse events grade I. An overall satisfaction score (scale of 1 to 10) showed no difference between the two groups (p=0.289). However, the CleanViewAL ® group showed a higher taste satisfaction score (scale of 1 to 5) than the Plenvu ® group (CleanViewAL ® : 2.90±0.91, Plenvu ® : 2.60±0.86, p=0.028).
Conclusions
Both types of 1-L PEG-Asc, CleanViewAL ® and Plenvu ® , are effective and safe bowel cleansing agents in average-aged adults. CleanViewAL ® was preferred in terms of taste satisfaction.
Keyword
Figure
Reference
-
1. Mareth K, Gurm H, Madhoun MF. 2022; Endoscopic recognition and classification of colorectal polyps. Gastrointest Endosc Clin N Am. 32:227–240. DOI: 10.1016/j.giec.2021.12.003. PMID: 35361333.2. Sood A, Mahajan R, Singh A, Midha V, Mehta V. Endoscopy for assessment of mucosal healing in ulcerative colitis: time bound or response guided? Intest Res. 2022; Feb. 8. [Epub ahead of print]. DOI: 10.5217/ir.2021.00099. PMID: 35124950. PMCID: PMC9344249.3. Toledo TK, DiPalma JA. 2001; Review article: colon cleansing preparation for gastrointestinal procedures. Aliment Pharmacol Ther. 15:605–611. DOI: 10.1046/j.1365-2036.2001.00966.x. PMID: 11328253.4. Quezada-Gutiérrez C, Álvarez-Bañuelos MT, Morales-Romero J, Sampieri CL, Guzmán-García RE, Montes-Villaseñor E. 2020; Factors associated with the survival of colorectal cancer in Mexico. Intest Res. 18:315–324. DOI: 10.5217/ir.2019.09179. PMID: 32418415. PMCID: PMC7385577.5. Jang JY, Chun HJ. 2014; Bowel preparations as quality indicators for colonoscopy. World J Gastroenterol. 20:2746–2750. DOI: 10.3748/wjg.v20.i11.2746. PMID: 24659866. PMCID: PMC3961991.6. Amitay EL, Niedermaier T, Gies A, Hoffmeister M, Brenner H. 2021; Risk Factors of Inadequate Bowel Preparation for Screening Colonoscopy. J Clin Med. 10:2740. DOI: 10.3390/jcm10122740. PMID: 34205800. PMCID: PMC8233947.7. Froehlich F, Wietlisbach V, Gonvers JJ, Burnand B, Vader JP. 2005; Impact of colonic cleansing on quality and diagnostic yield of colonoscopy: the European Panel of Appropriateness of Gastrointestinal Endoscopy European multicenter study. Gastrointest Endosc. 61:378–384. DOI: 10.1016/S0016-5107(04)02776-2. PMID: 15758907.8. Jeon SC, Kim JH, Kim SJ, et al. 2019; Effect of sending educational video clips via smartphone mobile messenger on bowel preparation before colonoscopy. Clin Endosc. 52:53–58. DOI: 10.5946/ce.2018.072. PMID: 30153724. PMCID: PMC6370934.9. Tan JJ, Tjandra JJ. 2006; Which is the optimal bowel preparation for colonoscopy- a meta-analysis. Colorectal Dis. 8:247–258. DOI: 10.1111/j.1463-1318.2006.00970.x. PMID: 16630226.10. Kim J, Kim HG, Kim KO, et al. 2019; Clinical comparison of low-volume agents (oral sulfate solution and sodium picosulfate with magnesium citrate) for bowel preparation: the EASE study. Intest Res. 17:413–418. DOI: 10.5217/ir.2018.00156. PMID: 30959582. PMCID: PMC6667374.11. Lee YH, Jeong SY, Kim YS, et al. 2015; Randomized controlled trial of sodium phosphate tablets versus 2 L polyethylene glycol solution for bowel cleansing prior to colonoscopy. Korean J Gastroenterol. 65:27–34. DOI: 10.4166/kjg.2015.65.1.27. PMID: 25603851.12. Di Palma JA, Rodriguez R, McGowan J, Cleveland Mv. 2009; A randomized clinical study evaluating the safety and efficacy of a new, reduced-volume, oral sulfate colon-cleansing preparation for colonoscopy. Am J Gastroenterol. 104:2275–2284. DOI: 10.1038/ajg.2009.389. PMID: 19584830.13. Yang HJ, Park DI, Park SK, et al. 2020; Novel sulfate tablet PBK-1701TC versus oral sulfate solution for colon cleansing: a randomized phase 3 trial. J Gastroenterol Hepatol. 35:29–36. DOI: 10.1111/jgh.14826. PMID: 31396995.14. Weir MA, Fleet JL, Vinden C, et al. 2014; Hyponatremia and sodium picosulfate bowel preparations in older adults. Am J Gastroenterol. 109:686–694. DOI: 10.1038/ajg.2014.20. PMID: 24589671.15. Belsey J, Epstein O, Heresbach D. 2009; Systematic review: adverse event reports for oral sodium phosphate and polyethylene glycol. Aliment Pharmacol Ther. 29:15–28. DOI: 10.1111/j.1365-2036.2008.03837.x. PMID: 18729847.16. Lee SW, Bang CS, Park TY, Suk KT, Baik GH, Kim DJ. 2017; Split-dose bowel preparation for colonoscopy: 2 liters polyethylene glycol with ascorbic acid versus sodium picosulfate versus oral sodium phosphate tablets. Korean J Gastroenterol. 70:89–95. DOI: 10.4166/kjg.2017.70.2.89. PMID: 28830134.17. Belsey J, Crosta C, Epstein O, et al. 2012; Meta-analysis: the relative efficacy of oral bowel preparations for colonoscopy 1985-2010. Aliment Pharmacol Ther. 35:222–237. DOI: 10.1111/j.1365-2036.2011.04927.x. PMID: 22112043.18. Bitoun A, Ponchon T, Barthet M, et al. 2006; Results of a prospective randomised multicentre controlled trial comparing a new 2-L ascorbic acid plus polyethylene glycol and electrolyte solution vs. sodium phosphate solution in patients undergoing elective colonoscopy. Aliment Pharmacol Ther. 24:1631–1642. DOI: 10.1111/j.1365-2036.2006.03167.x. PMID: 17094774.19. Hassan C, East J, Radaelli F, et al. 2019; Bowel preparation for colonoscopy: European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Update 2019. Endoscopy. 51:775–794. DOI: 10.1055/a-0959-0505. PMID: 31295746.20. Bechtold ML, Mir F, Puli SR, Nguyen DL. 2016; Optimizing bowel preparation for colonoscopy: a guide to enhance quality of visualization. Ann Gastroenterol. 29:137–146. DOI: 10.20524/aog.2016.0005. PMID: 27065725. PMCID: PMC4805732.21. Bisschops R, Manning J, Clayton LB, Ng Kwet Shing R, Álvarez-González M. 2019; Colon cleansing efficacy and safety with 1 L NER1006 versus 2 L polyethylene glycol + ascorbate: a randomized phase 3 trial. Endoscopy. 51:60–72. DOI: 10.1055/a-0638-8125. PMID: 30025414.22. Koo JS, Byeon JS, Lee BI, Jung SA, Kim TI, Jeen YT. 2022; Efficacy and safety of TJP-008 compared to 2 L PEG with ascorbate in colon cleansing: a randomized phase 3 trial. Gut Liver. 16:259–268. DOI: 10.5009/gnl210092. PMID: 34810296. PMCID: PMC8924813.23. Maida M, Sinagra E, Morreale GC, et al. 2020; Effectiveness of very low-volume preparation for colonoscopy: a prospective, multicenter observational study. World J Gastroenterol. 26:1950–1961. DOI: 10.3748/wjg.v26.i16.1950. PMID: 32390705. PMCID: PMC7201146.24. Kim EJ, Park YI, Kim YS, et al. 2014; A Korean experience of the use of Boston bowel preparation scale: a valid and reliable instrument for colonoscopy-oriented research. Saudi J Gastroenterol. 20:219–224. DOI: 10.4103/1319-3767.136950. PMID: 25038207. PMCID: PMC4131304.25. Calderwood AH, Jacobson BC. 2010; Comprehensive validation of the Boston bowel preparation scale. Gastrointest Endosc. 72:686–692. DOI: 10.1016/j.gie.2010.06.068. PMID: 20883845. PMCID: PMC2951305.26. 2022. Common terminology criteria for adverse events. [Internet]. UpToDate;Waltham (MA): Jun. updated 2022 Jun 27; cited 2021 Aug 15. Available from https://www.uptodate.com/contents/common-terminology-criteria-for-adverse-events.27. Keswani RN, Crockett SD, Calderwood AH. 2021; AGA clinical practice update on strategies to improve quality of screening and surveillance colonoscopy: expert review. Gastroenterology. 161:701–711. DOI: 10.1053/j.gastro.2021.05.041. PMID: 34334168.28. Woo DH, Kim KO, Jeong DE, et al. 2018; Prospective analysis of factors associated with inadequate bowel preparation for colonoscopy in actual clinical practice. Intest Res. 16:293–298. DOI: 10.5217/ir.2018.16.2.293. PMID: 29743843. PMCID: PMC5934603.29. DeMicco MP, Clayton LB, Pilot J, Epstein MS. NOCT Study Group. 2018; Novel 1 L polyethylene glycol-based bowel preparation NER1006 for overall and right-sided colon cleansing: a randomized controlled phase 3 trial versus trisulfate. Gastrointest Endosc. 87:677–687.e3. DOI: 10.1016/j.gie.2017.07.047. PMID: 28803744.30. Schreiber S, Baumgart DC, Drenth JPH, et al. 2019; Colon cleansing efficacy and safety with 1 L NER1006 versus sodium picosulfate with magnesium citrate: a randomized phase 3 trial. Endoscopy. 51:73–84. DOI: 10.1055/a-0639-5070. PMID: 30025415.31. Rex DK. 2019; Hyperosmotic low-volume bowel preparations: is NER1006 safe? Gastrointest Endosc. 89:656–658. DOI: 10.1016/j.gie.2018.11.009. PMID: 30448411.32. Hong SN, Lee CK, Im JP, et al. 2022; Efficacy and safety of split-dose bowel preparation with 1 L polyethylene glycol and ascorbate compared with 2 L polyethylene glycol and ascorbate in a Korean population: a phase IV, multicenter, randomized, endoscopist-blinded study. Gastrointest Endosc. 95:500–511.e2. DOI: 10.1016/j.gie.2021.09.041. PMID: 34653425.33. Maida M, Morreale GC, Sferrazza S, et al. 2021; Effectiveness and safety of 1L PEG-ASC preparation for colonoscopy in patients with inflammatory bowel diseases. Dig Liver Dis. 53:1171–1177. DOI: 10.1016/j.dld.2021.04.006. PMID: 33994129.34. Yoshida N, Naito Y, Murakami T, et al. 2016; Safety and Efficacy of a same-day low-volume 1 L PEG bowel preparation in colonoscopy for the elderly people and people with renal dysfunction. Dig Dis Sci. 61:3229–3235. DOI: 10.1007/s10620-016-4262-7. PMID: 27487795.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Optimal and Safe Bowel Preparation for Colonoscopy
- A Randomized Prospective Trial Comparing a New Polyethylene Glycol Based Lavage Solution with the Standard Polyethylene Glycol Solution in the Preparation of Patients Undergoing Colonoscopy (Clinical trial of new PEG solution in bowel preparation)
- Mechanical Bowel Preparation for Elective Colorectal Surgery: A Prospective Randomized Study Comparing Two Liters and Four Liters of Polyethylene Glycol-Based Oral Lavage Solutions
- Severe hyponatremia and seizures after bowel preparation with low-volume polyethylene glycol plus ascorbic acid solution
- Comparison of Oral Sulfate Solution and Polyethylene Glycol Plus Ascorbic Acid on the Efficacy of Bowel Preparation