J Korean Med Sci.
2022 Jul;37(28):e220. 10.3346/jkms.2022.37.e220.
Establishment of Patient-Derived Gastric Cancer Organoid Model From Tissue Obtained by Endoscopic Biopsies
- Affiliations
-
- 1Laboratory of Gastrointestinal Mucosal Immunology, Chung-Ang University College of Medicine, Seoul, Korea
- 2Department of Internal Medicine, Chung-Ang University College of Medicine, Seoul, Korea
- 3Institute of MD Healthcare Inc., Seoul, Korea
- 4Department of Pathology, Chung-Ang University College of Medicine, Seoul, Korea
- KMID: 2531802
- DOI: http://doi.org/10.3346/jkms.2022.37.e220
Abstract
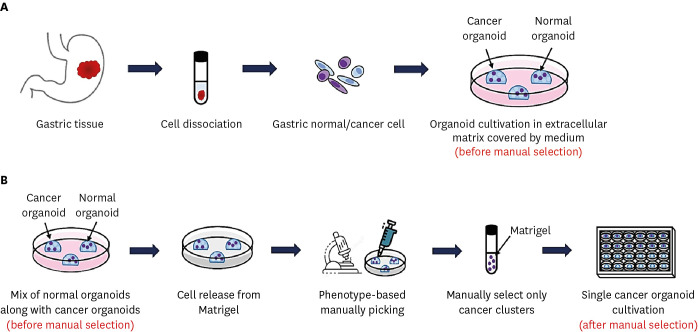
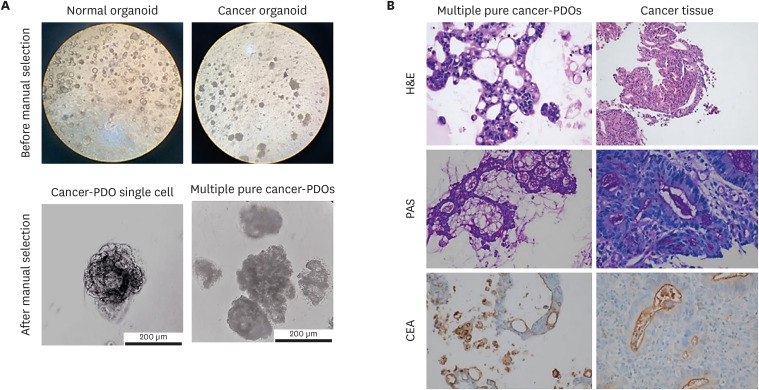
- Cancer organoids are three-dimensional mini-organ analogues derived from cancer tissues and have been proposed as models capable of simulating the structure and function of human organs and tissues in vitro. We sought to establish gastric cancer patient-derived organoids (PDOs) from tissues obtained by endoscopic biopsies. Gastric cancer-PDOs were successfully established and cultured from cancer tissues with gastric adenocarcinoma by endoscopic biopsies. To confirm that gastric cancer-PDOs were derived from cancer tissue, the consistency of the original cancer tissue was assessed by histopathological examination. As a result, it was confirmed that the shape and internal structure of gastric cancer-PDO were derived from the original gastric cancer cells, and the tumor specificity of gastric cancerPDO was confirmed through Periodic acid-Schiff (PAS) and polyclonal carcinoembryonic antigen antibody staining. These results demonstrate that gastric cancer-PDO models show the characteristics of primary tumors and have potential for drug screening and providing a personalized medicine platform.
Keyword
Figure
Reference
-
1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancer in 185 countries. CA Cancer J Clin. 2021; 71(3):209–249. PMID: 33538338.
Article2. Guideline Committee of the Korean Gastric Cancer Association (KGCA), Development Working Group & Review Panel. Korean practice guideline for gastric cancer 2018: an evidence-based, multi-disciplinary approach. J Gastric Cancer. 2019; 19(1):1–48. PMID: 30944757.3. Jeon J, Cheong JH. Clinical implementation of precision medicine in gastric cancer. J Gastric Cancer. 2019; 19(3):235–253. PMID: 31598369.
Article4. Aboulkheyr Es H, Montazeri L, Aref AR, Vosough M, Baharvand H. Personalized cancer medicine: an organoid approach. Trends Biotechnol. 2018; 36(4):358–371. PMID: 29366522.
Article5. Seidlitz T, Merker SR, Rothe A, Zakrzewski F, von Neubeck C, Grützmann K, et al. Human gastric cancer modelling using organoids. Gut. 2019; 68(2):207–217. PMID: 29703791.
Article6. Yan HH, Siu HC, Law S, Ho SL, Yue SS, Tsui WY, et al. A comprehensive human gastric cancer organoid biobank captures tumor subtype heterogeneity and enables therapeutic screening. Cell Stem Cell. 2018; 23(6):882–897.e11. PMID: 30344100.
Article7. Steele NG, Chakrabarti J, Wang J, Biesiada J, Holokai L, Chang J, et al. An organoid-based preclinical model of human gastric cancer. Cell Mol Gastroenterol Hepatol. 2019; 7(1):161–184. PMID: 30522949.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Establishment of Patient-Derived Pancreatic Cancer Organoids from Endoscopic Ultrasound-Guided Fine-Needle Aspiration Biopsies
- Clinical risk factors and pattern of initial fungal contamination in endoscopic biopsy-derived gastrointestinal cancer organoid culture
- Treatment Strategy of Gastric Adenoma
- Patient-Specific Tumor Microenvironment Models
- An Insufficient Preoperative Diagnosis of Borrmann Type 4 Gastric Cancer in Spite of EMR