J Rheum Dis.
2022 Jul;29(3):171-180. 10.4078/jrd.2022.29.3.171.
Clinical Implications of Shared Epitope and Anti-citrullinated Peptide Antibody in Patients With Rheumatoid Arthritis
- Affiliations
-
- 1Division of Rheumatology, Department of Internal Medicine, St. Vincent’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
- KMID: 2530681
- DOI: http://doi.org/10.4078/jrd.2022.29.3.171
Abstract
Objective
The shared epitope (SE) and anti-citrullinated peptide antibody (ACPA) are involved in the pathogenesis of rheumatoid arthritis (RA). This study evaluated the clinical implications of SE and ACPA in terms of disease manifestation and response to biologic disease modifying anti-rheumatic drugs (DMARDs).
Methods
Patients with identified human leukocyte antigen (HLA)-DRB1 alleles were included to compare the clinical characteristics and drug survival rate of tumor necrosis factor (TNF) inhibitors or abatacept based on the presence of SE and ACPA.
Results
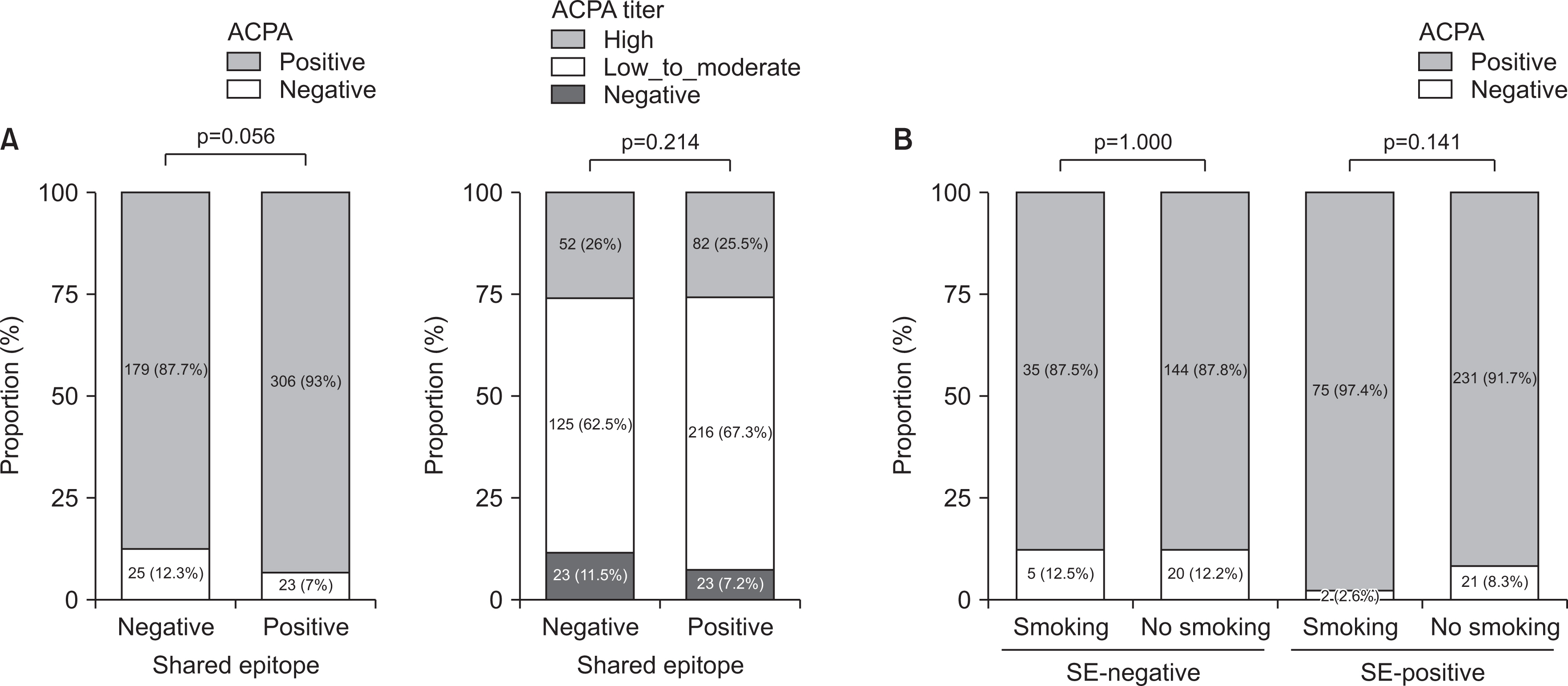
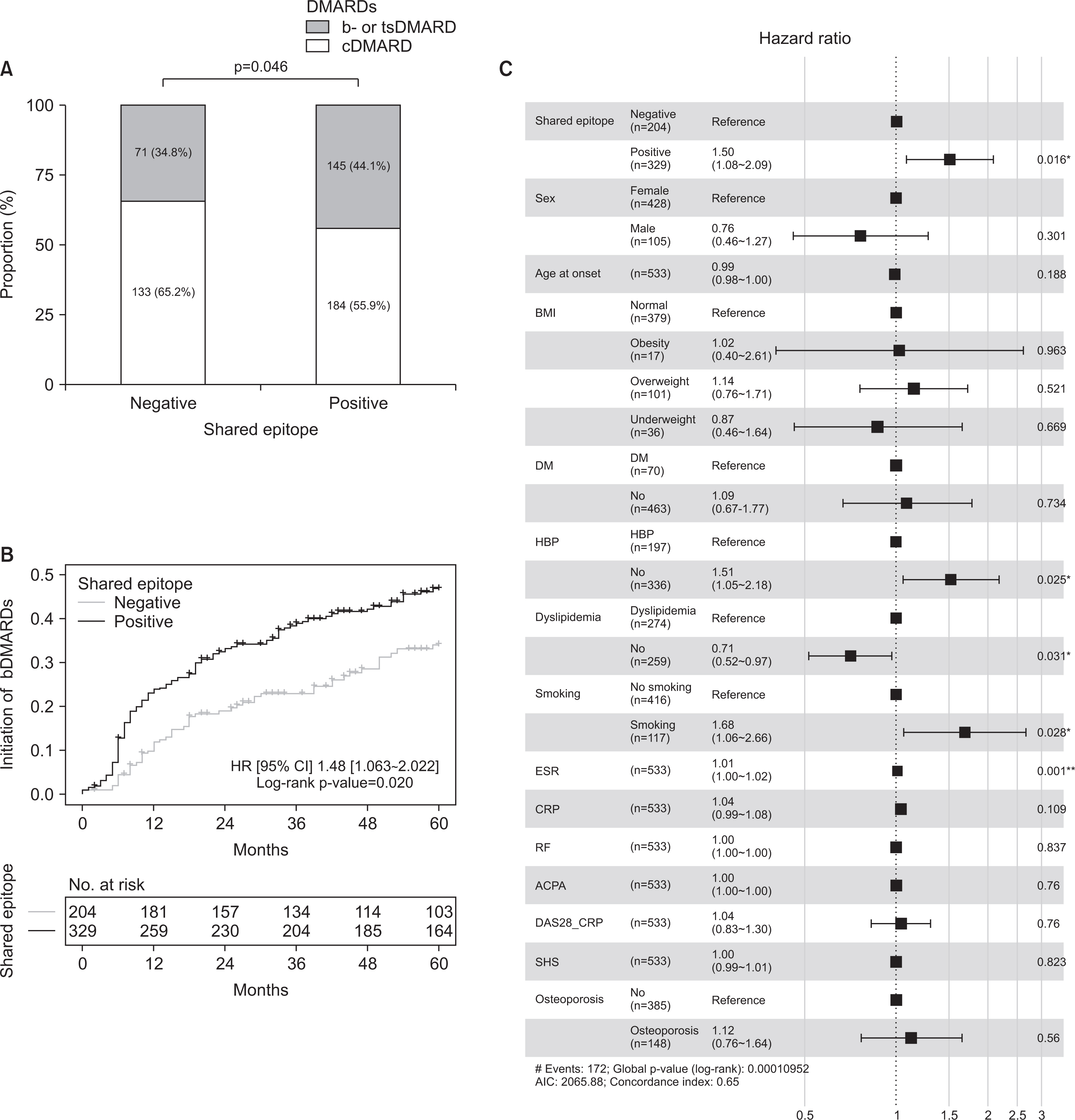
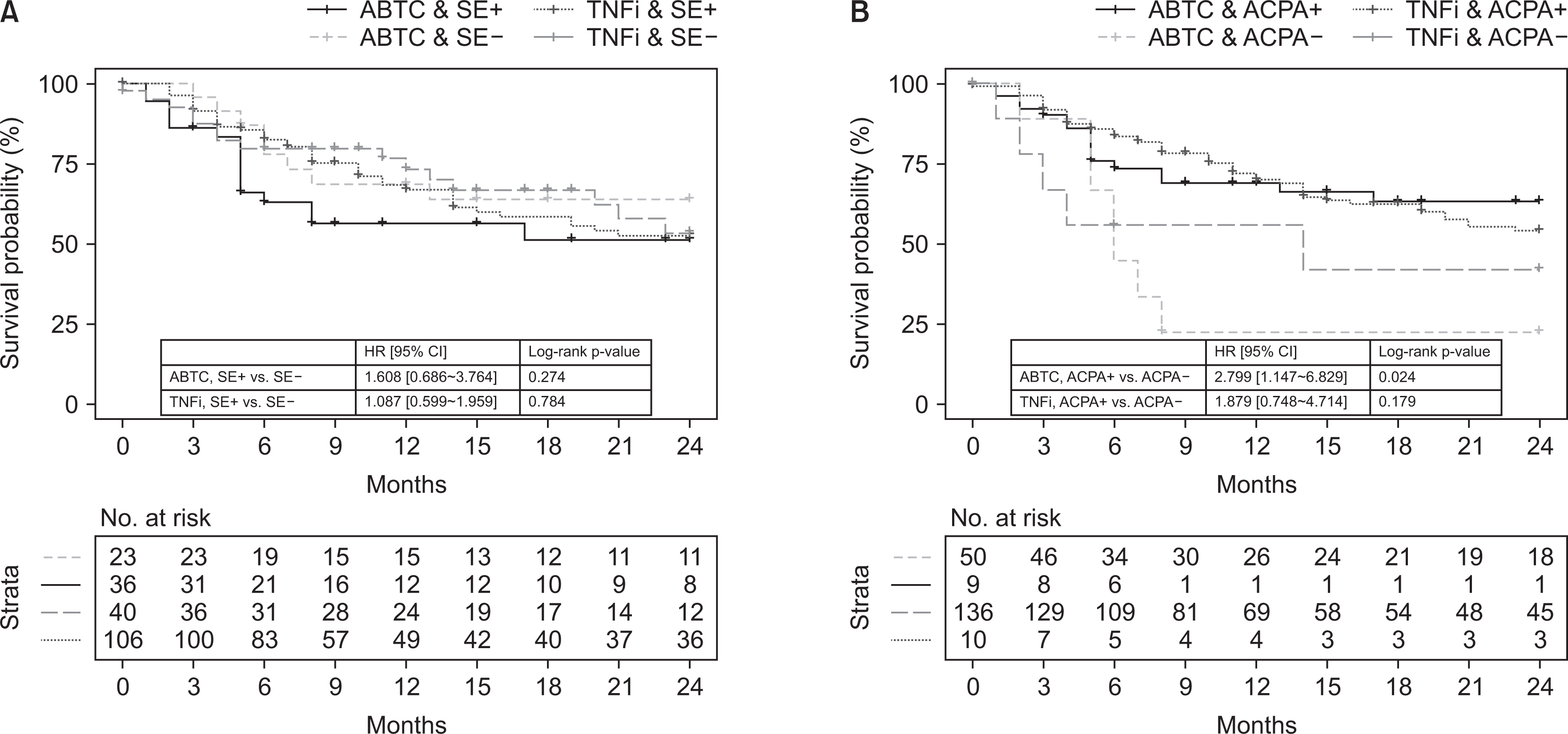
Of the 533 patients with identified HLA-DRB1 alleles, 329 patients (61.7%) with SE alleles showed higher disease activity and erosive changes compared to patients without SE alleles. SE-positive patients were more likely to start biologic (b-) or targeted synthetic DMARDs (tsDMARDs) within the first 5 years (p=0.020). The presence of SE, smoking, dyslipidemia, and higher erythrocyte sedimentation rate were independently associated with the initiation of b- or tsDMARDs (p=0.016, 0.028, 0.031, and 0.001, respectively). The presence of SE and ACPA did not affect the drug survival rate of TNF inhibitors, whereas the abatacept retention rate was higher in ACPA-positive patients (p=0.024).
Conclusion
The presence of SE affected disease characteristics and prognosis in Korean patients with RA without a significant impact on drug survival rate of TNF inhibitors and abatacept. ACPA positivity was associated with abatacept drug retention, suggesting that abatacept may be helpful in ACPA-positive patients than in ACPA-negative patients.
Keyword
Figure
Reference
-
1. Smolen JS, Aletaha D, Barton A, Burmester GR, Emery P, Firestein GS, et al. 2018; Rheumatoid arthritis. Nat Rev Dis Primers. 4:18001. DOI: 10.1038/nrdp.2018.1. PMID: 29417936.
Article2. Bang SY, Lee KH, Cho SK, Lee HS, Lee KW, Bae SC. 2010; Smoking increases rheumatoid arthritis susceptibility in individuals carrying the HLA-DRB1 shared epitope, regardless of rheumatoid factor or anti-cyclic citrullinated peptide antibody status. Arthritis Rheum. 62:369–77. DOI: 10.1002/art.27272. PMID: 20112396.3. Gonzalez-Gay MA, Garcia-Porrua C, Hajeer AH. 2002; Influence of human leukocyte antigen-DRB1 on the susceptibility and severity of rheumatoid arthritis. Semin Arthritis Rheum. 31:355–60. DOI: 10.1053/sarh.2002.32552. PMID: 12077707.
Article4. Moreno I, Valenzuela A, García A, Yélamos J, Sánchez B, Hernánz W. 1996; Association of the shared epitope with radiological severity of rheumatoid arthritis. J Rheumatol. 23:6–9.5. Too CL, Yahya A, Murad S, Dhaliwal JS, Larsson PT, Muhamad NA, et al. 2012; Smoking interacts with HLA-DRB1 shared epitope in the development of anti-citrullinated protein antibody-positive rheumatoid arthritis: results from the Malaysian Epidemiological Investigation of Rheumatoid Arthritis (MyEIRA). Arthritis Res Ther. 14:R89. DOI: 10.1186/ar3813. PMID: 22537824. PMCID: PMC3446463.
Article6. Gregersen PK, Silver J, Winchester RJ. 1987; The shared epitope hypothesis. An approach to understanding the molecular genetics of susceptibility to rheumatoid arthritis. Arthritis Rheum. 30:1205–13. DOI: 10.1002/art.1780301102. PMID: 2446635.
Article7. Fu J, Nogueira SV, Drongelen VV, Coit P, Ling S, Rosloniec EF, et al. 2018; Shared epitope-aryl hydrocarbon receptor crosstalk underlies the mechanism of gene-environment interaction in autoimmune arthritis. Proc Natl Acad Sci U S A. 115:4755–60. DOI: 10.1073/pnas.1722124115. PMID: 29666259. PMCID: PMC5939100.
Article8. Malmström V, Catrina AI, Klareskog L. 2017; The immunopathogenesis of seropositive rheumatoid arthritis: from triggering to targeting. Nat Rev Immunol. 17:60–75. DOI: 10.1038/nri.2016.124. PMID: 27916980.
Article9. Neogi T, Aletaha D, Silman AJ, Naden RL, Felson DT, Aggarwal R, et al. 2010; The 2010 American College of Rheumatology/European League Against Rheumatism classification criteria for rheumatoid arthritis: Phase 2 methodological report. Arthritis Rheum. 62:2582–91. DOI: 10.1002/art.27580. PMID: 20872596. PMCID: PMC3077961.
Article10. Berglin E, Johansson T, Sundin U, Jidell E, Wadell G, Hallmans G, et al. 2006; Radiological outcome in rheumatoid arthritis is predicted by presence of antibodies against cyclic citrullinated peptide before and at disease onset, and by IgA-RF at disease onset. Ann Rheum Dis. 65:453–8. DOI: 10.1136/ard.2005.041376. PMID: 16176994. PMCID: PMC1798112.
Article11. Syversen SW, Gaarder PI, Goll GL, Ødegård S, Haavardsholm EA, Mowinckel P, et al. 2008; High anti-cyclic citrullinated peptide levels and an algorithm of four variables predict radiographic progression in patients with rheumatoid arthritis: results from a 10-year longitudinal study. Ann Rheum Dis. 67:212–7. DOI: 10.1136/ard.2006.068247. PMID: 17526555.
Article12. Joo YB, Park YJ, Park KS, Kim KJ. 2019; Association of cumulative anti-cyclic citrullinated protein antibodies with radiographic progression in patients with rheumatoid arthritis. Clin Rheumatol. 38:2423–32. DOI: 10.1007/s10067-019-04554-w. PMID: 31049760.
Article13. Klareskog L, Rönnelid J, Lundberg K, Padyukov L, Alfredsson L. 2008; Immunity to citrullinated proteins in rheumatoid arthritis. Annu Rev Immunol. 26:651–75. DOI: 10.1146/annurev.immunol.26.021607.090244. PMID: 18173373.
Article14. Huizinga TW, Amos CI, van der Helm-van Mil AH, Chen W, van Gaalen FA, Jawaheer D, et al. 2005; Refining the complex rheumatoid arthritis phenotype based on specificity of the HLA-DRB1 shared epitope for antibodies to citrullinated proteins. Arthritis Rheum. 52:3433–8. DOI: 10.1002/art.21385. PMID: 16255021.
Article15. Jiang X, Frisell T, Askling J, Karlson EW, Klareskog L, Alfredsson L, et al. 2015; To what extent is the familial risk of rheumatoid arthritis explained by established rheumatoid arthritis risk factors? Arthritis Rheumatol. 67:352–62. DOI: 10.1002/art.38927. PMID: 25418518.
Article16. Klareskog L, Stolt P, Lundberg K, Källberg H, Bengtsson C, Grunewald J, et al. 2006; A new model for an etiology of rheumatoid arthritis: smoking may trigger HLA-DR (shared epitope)-restricted immune reactions to autoantigens modified by citrullination. Arthritis Rheum. 54:38–46. DOI: 10.1002/art.21575. PMID: 16385494.
Article17. Terao C, Yano K, Ikari K, Furu M, Yamakawa N, Yoshida S, et al. 2015; Brief report: main contribution of DRB1*04:05 among the shared epitope alleles and involvement of DRB1 amino acid position 57 in association with joint destruction in anti-citrullinated protein antibody-positive rheumatoid arthritis. Arthritis Rheumatol. 67:1744–50. DOI: 10.1002/art.39105. PMID: 25777156.
Article18. Smolen JS, Aletaha D. 2015; Rheumatoid arthritis therapy reappraisal: strategies, opportunities and challenges. Nat Rev Rheumatol. 11:276–89. DOI: 10.1038/nrrheum.2015.8. PMID: 25687177.
Article19. Gottenberg JE, Ravaud P, Cantagrel A, Combe B, Flipo RM, Schaeverbeke T, et al. 2012; Positivity for anti-cyclic citrullinated peptide is associated with a better response to abatacept: data from the 'Orencia and Rheumatoid Arthritis' registry. Ann Rheum Dis. 71:1815–9. DOI: 10.1136/annrheumdis-2011-201109. PMID: 22615458.
Article20. Sokolove J, Schiff M, Fleischmann R, Weinblatt ME, Connolly SE, Johnsen A, et al. 2016; Impact of baseline anti-cyclic citrullinated peptide-2 antibody concentration on efficacy outcomes following treatment with subcutaneous abatacept or adalimumab: 2-year results from the AMPLE trial. Ann Rheum Dis. 75:709–14. DOI: 10.1136/annrheumdis-2015-207942. PMID: 26359449. PMCID: PMC4819608.
Article21. Alten R, Nüßlein HG, Mariette X, Galeazzi M, Lorenz HM, Cantagrel A, et al. 2017; Baseline autoantibodies preferentially impact abatacept efficacy in patients with rheumatoid arthritis who are biologic naïve: 6-month results from a real-world, international, prospective study. RMD Open. 3:e000345. DOI: 10.1136/rmdopen-2016-000345. PMID: 28243468. PMCID: PMC5307376.
Article22. Harrold LR, Litman HJ, Connolly SE, Kelly S, Hua W, Alemao E, et al. 2018; Effect of anticitrullinated protein antibody status on response to abatacept or antitumor necrosis factor-α therapy in patients with rheumatoid arthritis: a US national observational study. J Rheumatol. 45:32–9. DOI: 10.3899/jrheum.170007. PMID: 29093151.
Article23. Alemao E, Postema R, Elbez Y, Mamane C, Finckh A. 2020; Presence of anti-cyclic citrullinated peptide antibodies is associated with better treatment response to abatacept but not to TNF inhibitors in patients with rheumatoid arthritis: a meta-analysis. Clin Exp Rheumatol. 38:455–66.24. Griffiths B, Situnayake RD, Clark B, Tennant A, Salmon M, Emery P. 2000; Racial origin and its effect on disease expression and HLA-DRB1 types in patients with rheumatoid arthritis: a matched cross-sectional study. Rheumatology (Oxford). 39:857–64. DOI: 10.1093/rheumatology/39.8.857. PMID: 10952739.
Article25. Helliwell PS, Ibrahim G. 2003; Ethnic differences in responses to disease modifying drugs. Rheumatology (Oxford). 42:1197–201. DOI: 10.1093/rheumatology/keg354. PMID: 12810940.
Article26. Smolen JS, Landewé RBM, Bijlsma JWJ, Burmester GR, Dougados M, Kerschbaumer A, et al. 2020; EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2019 update. Ann Rheum Dis. 79:685–99. DOI: 10.1136/annrheumdis-2019-216655. PMID: 31969328.27. van Riel PL, Fransen J. 2005; DAS28: a useful instrument to monitor infliximab treatment in patients with rheumatoid arthritis. Arthritis Res Ther. 7:189–90. DOI: 10.1186/ar1820. PMID: 16207346. PMCID: PMC1257454.28. Seo DH, Lee JM, Park MO, Lee HJ, Moon SY, Oh M, et al. 2018; Development of HLA-A, -B and -DR typing method using next-generation sequencing. Korean J Blood Transfus. 29:310–9. DOI: 10.17945/kjbt.2018.29.3.310.
Article29. Fisher BA, Bang SY, Chowdhury M, Lee HS, Kim JH, Charles P, et al. 2014; Smoking, the HLA-DRB1 shared epitope and ACPA fine-specificity in Koreans with rheumatoid arthritis: evidence for more than one pathogenic pathway linking smoking to disease. Ann Rheum Dis. 73:741–7. DOI: 10.1136/annrheumdis-2012-202535. PMID: 23505239.
Article30. Hecht C, Englbrecht M, Rech J, Schmidt S, Araujo E, Engelke K, et al. 2015; Additive effect of anti-citrullinated protein antibodies and rheumatoid factor on bone erosions in patients with RA. Ann Rheum Dis. 74:2151–6. DOI: 10.1136/annrheumdis-2014-205428. PMID: 25115448.
Article31. van der Heijde D. 2000; How to read radiographs according to the Sharp/van der Heijde method. J Rheumatol. 27:261–3. DOI: 10.1097/00002281-200007000-00005. PMID: 10910177.32. Lundström E, Källberg H, Alfredsson L, Klareskog L, Padyukov L. 2009; Gene-environment interaction between the DRB1 shared epitope and smoking in the risk of anti-citrullinated protein antibody-positive rheumatoid arthritis: all alleles are important. Arthritis Rheum. 60:1597–603. DOI: 10.1002/art.24572. PMID: 19479873. PMCID: PMC2732897.
Article33. Willemze A, van der Woude D, Ghidey W, Levarht EW, Stoeken-Rijsbergen G, Verduyn W, et al. 2011; The interaction between HLA shared epitope alleles and smoking and its contribution to autoimmunity against several citrullinated antigens. Arthritis Rheum. 63:1823–32. DOI: 10.1002/art.30409. PMID: 21506097.
Article34. Teitsma XM, Jacobs JWG, Welsing PMJ, de Jong PHP, Hazes JMW, Weel AEAM, et al. 2018; Inadequate response to treat-to-target methotrexate therapy in patients with new-onset rheumatoid arthritis: development and validation of clinical predictors. Ann Rheum Dis. 77:1261–7. DOI: 10.1136/annrheumdis-2018-213035. PMID: 29760159.
Article35. Mattey DL, Hassell AB, Dawes PT, Cheung NT, Poulton KV, Thomson W, et al. 2001; Independent association of rheumatoid factor and the HLA-DRB1 shared epitope with radiographic outcome in rheumatoid arthritis. Arthritis Rheum. 44:1529–33. DOI: 10.1002/1529-0131(200107)44:7<1529::AID-ART275>3.0.CO;2-P.
Article36. Wysocki T, Olesińska M, Paradowska-Gorycka A. 2020; Current understanding of an emerging role of HLA-DRB1 gene in rheumatoid arthritis-from research to clinical practice. Cells. 9:1127. DOI: 10.3390/cells9051127. PMID: 32370106. PMCID: PMC7291248.
Article37. Criswell LA, Lum RF, Turner KN, Woehl B, Zhu Y, Wang J, et al. 2004; The influence of genetic variation in the HLA-DRB1 and LTA-TNF regions on the response to treatment of early rheumatoid arthritis with methotrexate or etanercept. Arthritis Rheum. 50:2750–6. DOI: 10.1002/art.20469. PMID: 15457442.38. Oryoji K, Yoshida K, Kashiwado Y, Tanaka K, Mizuki SI, Tsukamoto H, et al. 2018; Shared epitope positivity is related to efficacy of abatacept in rheumatoid arthritis. Ann Rheum Dis. 77:1234–6. DOI: 10.1136/annrheumdis-2017-eular.5586.
Article39. Neovius M, Arkema EV, Olsson H, Eriksson JK, Kristensen LE, Simard JF, et al. 2015; Drug survival on TNF inhibitors in patients with rheumatoid arthritis comparison of adalimumab, etanercept and infliximab. Ann Rheum Dis. 74:354–60. DOI: 10.1136/annrheumdis-2013-204128. PMID: 24285495. PMCID: PMC4316855.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Periodontitis is associated with rheumatoid arthritis: a study with longstanding rheumatoid arthritis patients in Korea
- Association of Anti-cyclic Citrullinated Peptide (CCP) Antibodies and Functional Status in Rheumatoid Arthritis
- Diagnostic Role of the Anti-Cyclic Citrullinated Peptide Antibodies in Rheumatoid Arthritis
- The Clinical Significance of Shared Epitope in Rheumatoid Arthritis
- A Case of Anti-Cyclic Citrullinated Peptide Antibody-Positive Rheumatoid Arthritis Following Peginterferon Alpha-2A and Ribavirin Therapy for Chronic Hepatitis C