Korean Circ J.
2022 Apr;52(4):324-337. 10.4070/kcj.2021.0321.
Ticagrelor Monotherapy After 3-Month Dual Antiplatelet Therapy in Acute Coronary Syndrome by High Bleeding Risk: The Subanalysis From the TICO Trial
- Affiliations
-
- 1Division of Cardiology, Severance Cardiovascular Hospital, Yonsei University College of Medicine, Seoul, Korea
- 2Division of Cardiology, Department of Internal Medicine, Myongji Hospital, Hanyang University College of Medicine, Goyang, Korea
- 3Division of Cardiology, Wonkwang University Hospital, Iksan, Korea
- 4Division of Cardiology, Kangwon National University School of Medicine, Chuncheon, Korea
- 5Division of Cardiology, Dankook University Hospital, Dankook University College of Medicine, Cheonan, Korea
- 6Division of Cardiology, National Health Insurance Service Ilsan Hospital, Goyang, Korea
- 7Division of Cardiology, Gangneung Asan Hospital, University of Ulsan College of Medicine, Gangneung, Korea
- 8Division of Cardiology, Yongin Severance Hospital, Yonsei University College of Medicine, Yongin, Korea
- 9Division of Cardiology, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul, Korea
- 10Department of Preventive Medicine and Biostatistics, Yonsei University College of Medicine, Seoul, Korea
- 11Division of Cardiology, CHA Bundang Medical Center, CHA University College of Medicine, Seongnam, Korea
- KMID: 2528027
- DOI: http://doi.org/10.4070/kcj.2021.0321
Abstract
- Background and Objectives
Identifying patients with high bleeding risk (HBR) is important when making decisions for antiplatelet therapy strategy. This study evaluated the impact of ticagrelor monotherapy after 3-month dual antiplatelet therapy (DAPT) according to HBR in acute coronary syndrome (ACS) patients treated with drug eluting stents (DESs).
Methods
In this post-hoc analysis of the TICO trial, HBR was defined by 2 approaches: meeting Academic Research Consortium for HBR (ARC-HBR) criteria or Predicting Bleeding Complications in Patients Undergoing Stent Implantation and Subsequent DAPT (PRECISEDAPT) score ≥25. The primary outcome was a 3–12 months net adverse clinical event (composite of major bleeding and adverse cardiac and cerebrovascular events).
Results
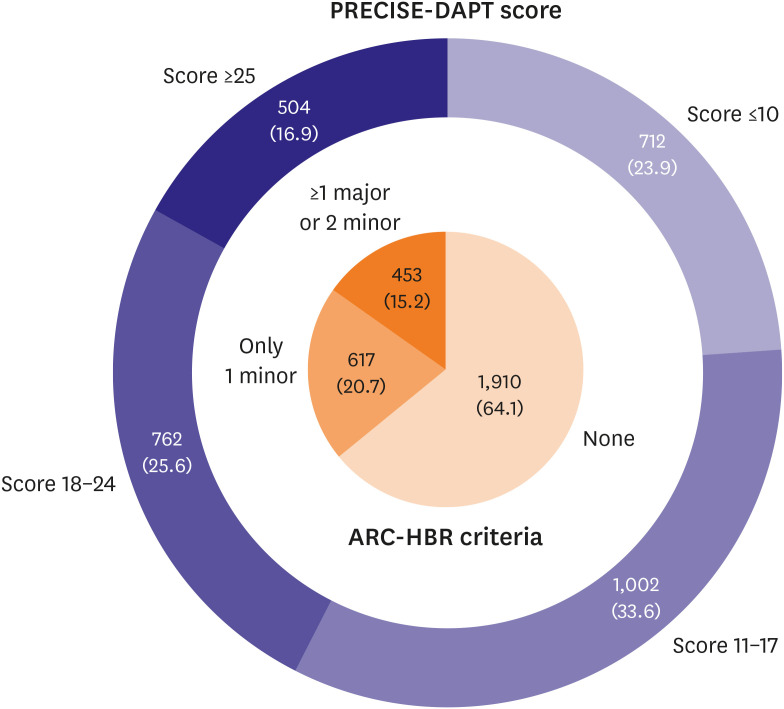
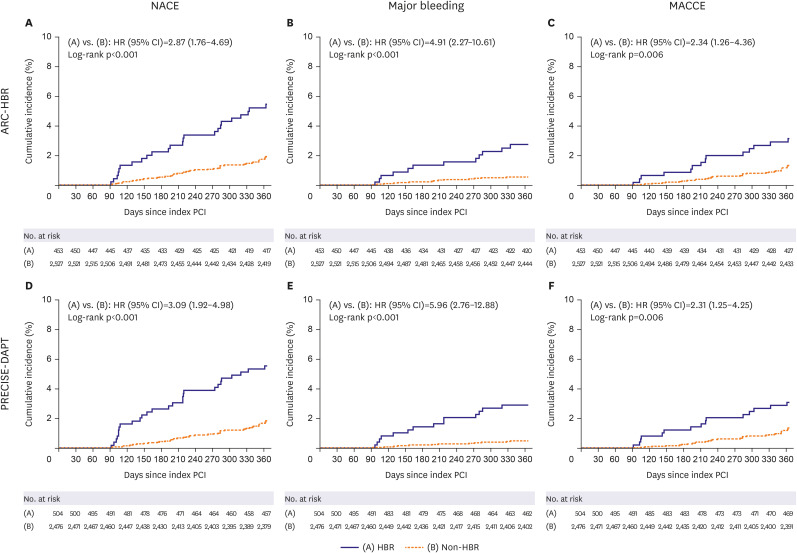
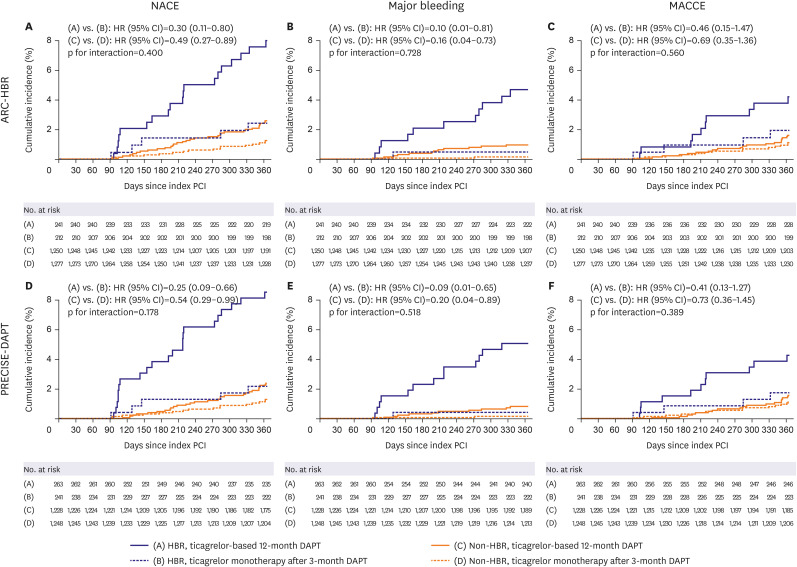
Of the 2,980 patients without adverse events during the first 3 months after DES implantation, 453 (15.2%) were HBR by ARC-HBR criteria and 504 (16.9%) were HBR by PRECISE-DAPT score. The primary outcome rate was higher in HBR versus non-HBR patients (by ARC-HBR criteria: hazard ratio [HR], 2.87; 95% confidence interval [CI], 1.76– 4.69; p<0.001; by PRECISE-DAPT score: HR, 3.09; 95% CI, 1.92–4.98; p<0.001). Ticagrelor monotherapy after 3-month DAPT was associated with lower primary outcome rate than ticagrelor-based 12-month DAPT regardless of HBR by ARC-HBR criteria, with similar magnitudes of therapy effect for HBR and non-HBR patients (p-interaction=0.400). Results were consistent by PRECISE-DAPT score (p-interaction=0.178).
Conclusions
In ACS patients treated with DESs, ticagrelor monotherapy after 3-month DAPT was associated with lower rate of adverse clinical outcomes regardless of HBR, with similar magnitudes of therapy effect between HBR and non-HBR. Trial Registration: ClinicalTrials.gov Identifier: NCT02494895
Keyword
Figure
Cited by 1 articles
-
Balancing Between Ischemic and Bleeding Risk in PCI Patients With ‘Bi-Risk’
Mahn-Won Park
Korean Circ J. 2022;52(4):338-340. doi: 10.4070/kcj.2021.0429.
Reference
-
1. Levine GN, Bates ER, Bittl JA, et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. J Am Coll Cardiol. 2016; 68:1082–1115. PMID: 27036918.2. Valgimigli M, Bueno H, Byrne RA, et al. 2017 ESC focused update on dual antiplatelet therapy in coronary artery disease developed in collaboration with EACTS: the task force for dual antiplatelet therapy in coronary artery disease of the European Society of Cardiology (ESC) and of the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2018; 39:213–260. PMID: 28886622.3. Urban P, Mehran R, Colleran R, et al. Defining high bleeding risk in patients undergoing percutaneous coronary intervention. Circulation. 2019; 140:240–261. PMID: 31116032.
Article4. Costa F, van Klaveren D, James S, et al. Derivation and validation of the predicting bleeding complications in patients undergoing stent implantation and subsequent dual antiplatelet therapy (PRECISE-DAPT) score: a pooled analysis of individual-patient datasets from clinical trials. Lancet. 2017; 389:1025–1034. PMID: 28290994.
Article5. Ueki Y, Karagiannis A, Zanchin C, et al. Validation of high-risk features for stent-related ischemic events as endorsed by the 2017 DAPT guidelines. JACC Cardiovasc Interv. 2019; 12:820–830. PMID: 30981574.
Article6. Ueki Y, Bar S, Losdat S, et al. Validation of the Academic Research Consortium for High Bleeding Risk (ARC-HBR) criteria in patients undergoing percutaneous coronary intervention and comparison with contemporary bleeding risk scores. EuroIntervention. 2020; 16:371–379. PMID: 32065586.
Article7. Natsuaki M, Morimoto T, Shiomi H, et al. Application of the academic research consortium high bleeding risk criteria in an all-comers registry of percutaneous coronary intervention. Circ Cardiovasc Interv. 2019; 12:e008307. PMID: 31707804.
Article8. Wallentin L, Becker RC, Budaj A, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009; 361:1045–1057. PMID: 19717846.
Article9. Wiviott SD, Braunwald E, McCabe CH, et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2007; 357:2001–2015. PMID: 17982182.
Article10. Mehran R, Baber U, Sharma SK, et al. Ticagrelor with or without aspirin in high-risk patients after PCI. N Engl J Med. 2019; 381:2032–2042. PMID: 31556978.
Article11. Kim BK, Hong SJ, Cho YH, et al. Effect of ticagrelor monotherapy vs ticagrelor with aspirin on major bleeding and cardiovascular events in patients with acute coronary syndrome: the TICO randomized clinical trial. JAMA. 2020; 323:2407–2416. PMID: 32543684.
Article12. Kim C, Hong SJ, Shin DH, et al. Randomized evaluation of ticagrelor monotherapy after 3-month dual-antiplatelet therapy in patients with acute coronary syndrome treated with new-generation sirolimus-eluting stents: TICO trial rationale and design. Am Heart J. 2019; 212:45–52. PMID: 30933857.
Article13. Mehran R, Rao SV, Bhatt DL, et al. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation. 2011; 123:2736–2747. PMID: 21670242.
Article14. Thygesen K, Alpert JS, Jaffe AS, et al. Third universal definition of myocardial infarction. Circulation. 2012; 126:2020–2035. PMID: 22923432.
Article15. Cutlip DE, Windecker S, Mehran R, et al. Clinical end points in coronary stent trials: a case for standardized definitions. Circulation. 2007; 115:2344–2351. PMID: 17470709.16. Sacco RL, Kasner SE, Broderick JP, et al. An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013; 44:2064–2089. PMID: 23652265.
Article17. Kim BK, Hong MK, Shin DH, et al. A new strategy for discontinuation of dual antiplatelet therapy: the RESET Trial (REal Safety and Efficacy of 3-month dual antiplatelet Therapy following Endeavor zotarolimus-eluting stent implantation). J Am Coll Cardiol. 2012; 60:1340–1348. PMID: 22999717.18. Capodanno D, Alfonso F, Levine GN, Valgimigli M, Angiolillo DJ. ACC/AHA versus ESC guidelines on dual antiplatelet therapy: JACC guideline comparison. J Am Coll Cardiol. 2018; 72:2915–2931. PMID: 30522654.19. Costa F, Van Klaveren D, Feres F, et al. Dual antiplatelet therapy duration based on ischemic and bleeding risks after coronary stenting. J Am Coll Cardiol. 2019; 73:741–754. PMID: 30784667.
Article20. Kandzari DE, Mauri L, Koolen JJ, et al. Ultrathin, bioresorbable polymer sirolimus-eluting stents versus thin, durable polymer everolimus-eluting stents in patients undergoing coronary revascularisation (BIOFLOW V): a randomised trial. Lancet. 2017; 390:1843–1852. PMID: 28851504.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Dual Antiplatelet Therapy after Noncardioembolic Ischemic Stroke or Transient Ischemic Attack: Pros and Cons
- Recent Evidence of Potent P2Y12 Inhibitor Monotherapy After Short-term Dual Antiplatelet Therapy for Patients With Acute Coronary Syndrome
- De-escalation strategies of dual antiplatelet therapy in patients undergoing percutaneous coronary intervention for acute coronary syndrome
- De-Escalation of P2Yâ‚â‚‚ Receptor Inhibitor Therapy after Acute Coronary Syndromes in Patients Undergoing Percutaneous Coronary Intervention
- Hypersensitivity to ticagrelor and low response to clopidogrel: a case report