J Korean Med Sci.
2022 Jan;37(4):e26. 10.3346/jkms.2022.37.e26.
Korean Clinical Practice Guidelines: Current Status of Adherence to the RIGHT Checklist
- Affiliations
-
- 1Division of Health Technology Assessment Research, National Evidence-based Healthcare Collaborating Agency, Seoul, Korea
- 2Department of Laboratory Medicine and Genetics, Soonchunhyang University College of Medicine, Bucheon, Korea
- 3Department of Family Medicine, Kangdong Sacred Heart Hospital, Hallym University College of Medicine, Seoul, Korea
- KMID: 2525007
- DOI: http://doi.org/10.3346/jkms.2022.37.e26
Abstract
- Background
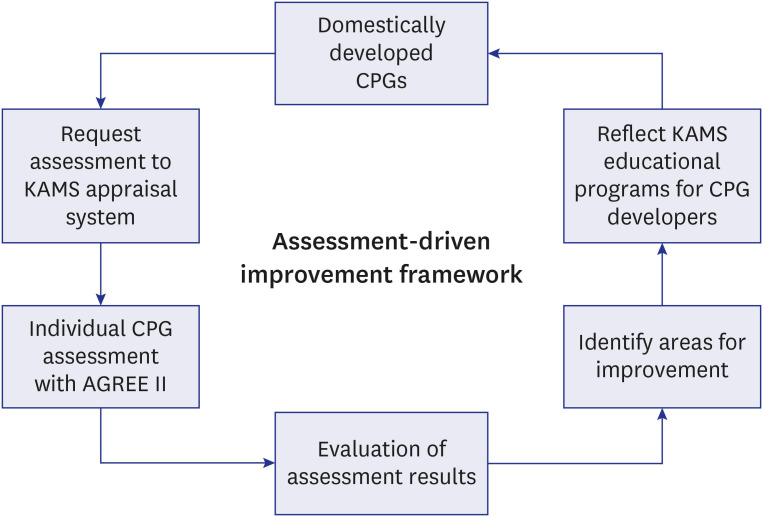
The Korean Academy of Medical Sciences (KAMS) has been utilizing AGREE II to audit the quality of clinical practice guidelines (CPGs) developed in Korea. Monitoring the RIGHT Checklist adherence could help monitor the quality status and discover areas for improvement of CPG development.
Methods
We included 129 CPGs from the past 5 years and assessed each item of the RIGHT Checklist. STATA version 15.0 was used for statistical analysis.
Results
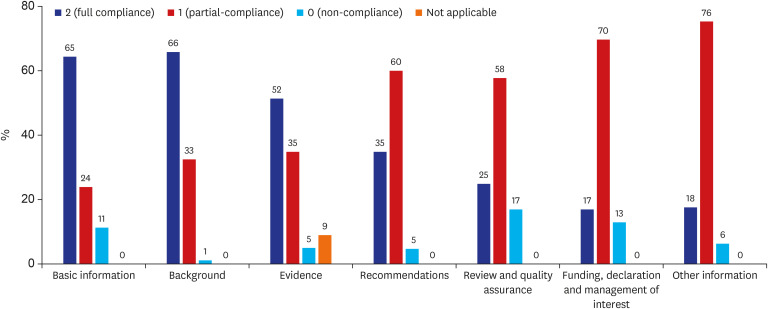
Among the seven sections of the RIGHT checklist, sections with a full compliance rate over 60% were ‘basic information’ (65%) and ‘background’ (66%). The other sections’ mean full compliance rates were ‘Evidence’ 52%, ‘Recommendation’ 35%, ‘Review and quality assurance’ 25% and ‘Funding, declaration and management of interest’ 17%. Sections with a partial compliance rate over 60% were ‘Recommendation’ (60%) and ‘Funding, declaration and management of interest’ (70%). Non-compliance was highest in the ‘Review and quality assurance’ (17%) domain. In comparison between groups 1 (under median group) and 2 (over median group), group 2 showed a tendency to have multi-stakeholder involvement and present sufficient information on financial resources and conflict of interest declarations. For the CPGs developmental methodology aspect, group 2 provided more pertinent information than group 1 about supporting evidence-making and the process from evidence to recommendation.
Conclusion
This study evaluated adherence to the RIGHT Checklist of CPGs developed in Korea. It can provide helpful information to develop strategic plans for enhancing the capabilities of developing CPGs in Korea.
Keyword
Figure
Reference
-
1. Korean Medical Guideline Information Center (KOMGI). List of developed Korean Clinical Practice Guidelines. Updated 2021. Accessed April 7, 2020. https://www.guideline.or.kr/guide/?sub_depth=3&c=1 .2. Alonso-Coello P, Irfan A, Solà I, Gich I, Delgado-Noguera M, Rigau D, et al. The quality of clinical practice guidelines over the last two decades: a systematic review of guideline appraisal studies. Qual Saf Health Care. 2010; 19(6):e58. PMID: 21127089.
Article3. Lee YK, Shin ES, Shim JY, Min KJ, Kim JM, Lee SH, et al. Developing a scoring guide for the Appraisal of Guidelines for Research and Evaluation II instrument in Korea: a modified Delphi consensus process. J Korean Med Sci. 2013; 28(2):190–194. PMID: 23400114.
Article4. Oh MK, Jo H, Lee YK. Improving the reliability of clinical practice guideline appraisals: effects of the Korean AGREE II scoring guide. J Korean Med Sci. 2014; 29(6):771–775. PMID: 24932076.
Article5. Chang SG, Kim DI, Shin ES, Jang JE, Yeon JY, Lee YS. Methodological quality appraisal of 27 Korean guidelines using a scoring guide based on the AGREE II instrument and a web-based evaluation. J Korean Med Sci. 2016; 31(5):682–687. PMID: 27134487.
Article6. Choi M, Kim N, Sheen S, Ji S, Lyu D, You J. Current status of clinical practice guideline development and dissemination in Korea for identifying collaborative research demands. Evid Value. 2015; 1(1):16–23.7. EQUATOR. Updated 2021. Accessed April 7, 2020. https://www.equator-network.org .8. Brouwers MC, Kerkvliet K, Spithoff K. AGREE Next Steps Consortium. The AGREE Reporting Checklist: a tool to improve reporting of clinical practice guidelines. BMJ. 2016; 352:i1152. PMID: 26957104.
Article9. Chen Y, Yang K, Marušic A, Qaseem A, Meerpohl JJ, Flottorp S, et al. A reporting tool for practice guidelines in health care: the RIGHT statement. Ann Intern Med. 2017; 166(2):128–132. PMID: 27893062.
Article10. Yao X, Ma J, Wang Q, Kanters D, Ali MU, Florez ID. A comparison of AGREE and RIGHT: which clinical practice guideline reporting checklist should be followed by guideline developers? J Gen Intern Med. 2020; 35(3):894–898. PMID: 31713037.
Article11. Choi M, Kim SY, Lee YK. Executive Committee for Clinical Practice Guidelines, The Korean Academy of Medical Sciences. Current status of clinical practice guidelines in Korea. J Korean Med Sci. 2021; 36(6):e35. PMID: 33559406.
Article12. Ji SM, Kim SY, Sheen SS, Heo DS, Kim NS. Consensus on definition and quality standard of clinical practice guideline using RAND method. Health Policy Manag. 2010; 20(2):1–16.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Development and Implementation of Clinical Practice Guidelines: Current Status in Korea
- A survey of attitudes to clinical practice guidelines in general and adherence of the Korea practical guidelines for management of gallbladder polyp: a survey among private clinicians in Korea
- Association between Smoking Status, Adherence to Physical Activity Guidelines, and the Atherogenic Index of Plasma in Korean Adults: A Cross-Sectional Study
- Standardized Patients' Accuracy in Recording Checklist Items during Clinical Performance Examinations
- The current capacity and quality of colonoscopy in Korea