Cancer Res Treat.
2022 Jan;54(1):174-181. 10.4143/crt.2020.749.
Evaluation of a Direct Reverse Transcription Loop-Mediated Isothermal Amplification Method without RNA Extraction (Direct RT-LAMP) for the Detection of Lymph Node Metastasis in Early Breast Cancer
- Affiliations
-
- 1Division of Hematology/Oncology, Department of Internal Medicine, Daegu Catholic University School of Medicine, Daegu, Korea
- 2Department of Surgery, Kyungpook National University Chilgok Hospital, School of Medicine, Kyungpook National University, Daegu, Korea
- 3Department of Oncology/Hematology, Kyungpook National University Chilgok Hospital, School of Medicine, Kyungpook National University, Daegu, Korea
- 4Department of Pathology, Kyungpook National University Chilgok Hospital, School of Medicine, Kyungpook National University, Daegu, Korea
- 5M Monitor, Inc., Daegu, Korea
- 6DNP Biotech, Inc., Daegu, Korea
- KMID: 2524598
- DOI: http://doi.org/10.4143/crt.2020.749
Abstract
- Purpose
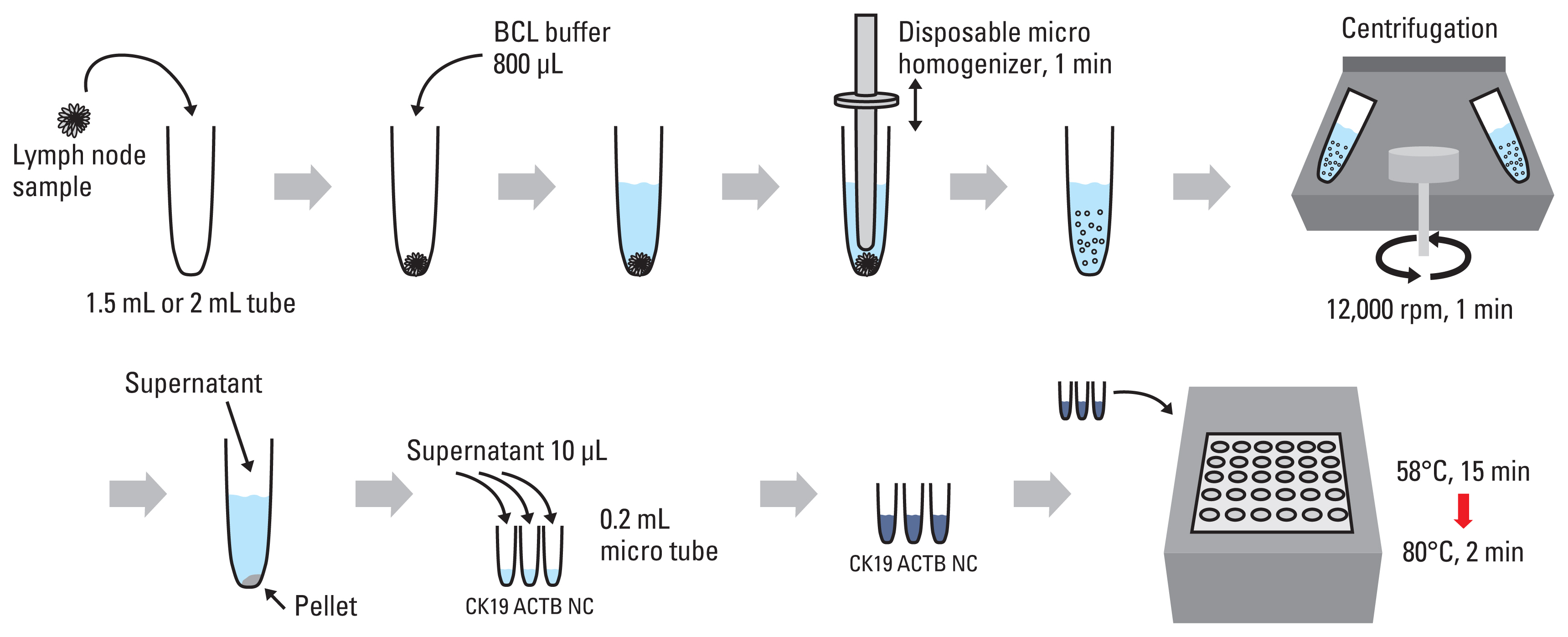
Assessing lymph node metastasis, tumor-derived DNA, or tumor-derived RNA has previously been studied in place of immunohistochemical assay. Because a direct reverse transcription loop-mediated isothermal amplification method (direct RT-LAMP) has been previously developed in order to rapidly identify viruses in place of RNA extraction, our team hypothesized that a direct RT-LAMP assay can be employed as a substitute in order to detect tumor involvement of lymph nodes within breast cancer patients.
Materials and Methods
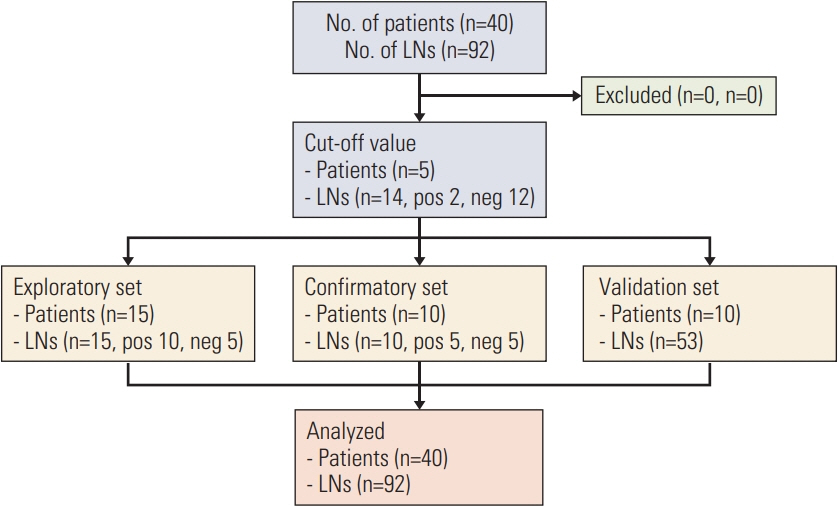
A total amount of 92 lymph nodes removed across 40 patients possessing breast cancer were collected at Kyungpook National University Chilgok Hospital between the months of November 2015 and February 2016. All samples were then evaluated and contrasted via both a direct RT-LAMP assay and routine histopathologic examination.
Results
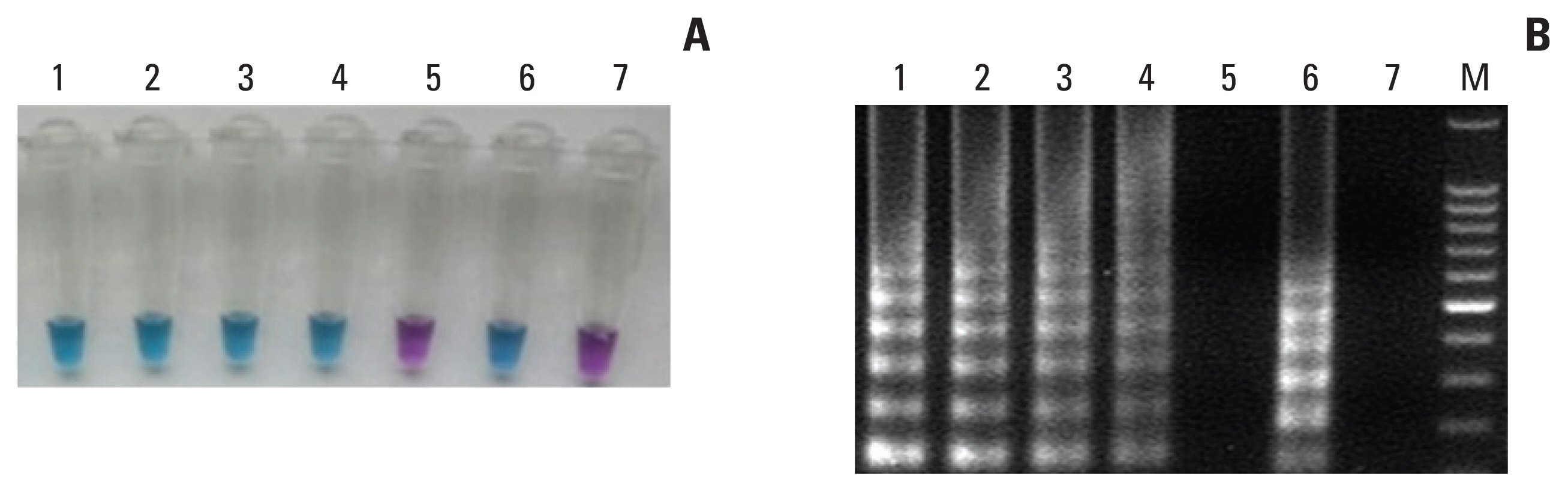
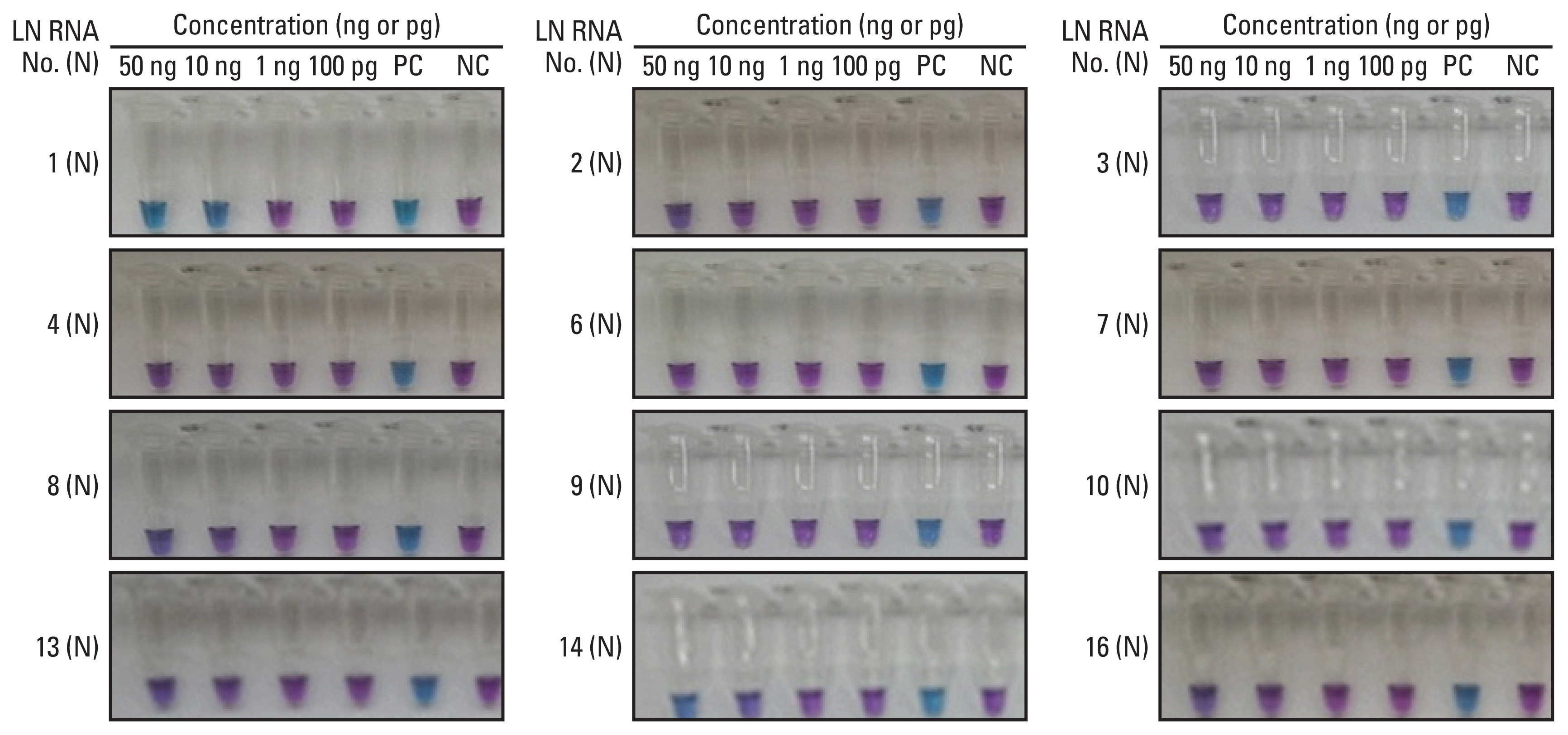
The sensitivity and specificity of the direct RT-LAMP assay were 85.7% and 100%, respectively. The positive predictive value and negative predictive value were 100% and 94.4%, respectively.
Conclusion
Direct RT-LAMP assay is capable of facilitating the detection of sentinel lymph node metastasis within breast cancer patients intraoperatively possessing an excellent sensitivity via a cost-effective and time-saving manner.
Keyword
Figure
Reference
-
References
1. Krag DN, Anderson SJ, Julian TB, Brown AM, Harlow SP, Costantino JP, et al. Sentinel-lymph-node resection compared with conventional axillary-lymph-node dissection in clinically node-negative patients with breast cancer: overall survival findings from the NSABP B-32 randomised phase 3 trial. Lancet Oncol. 2010; 11:927–33.
Article2. Fleissig A, Fallowfield LJ, Langridge CI, Johnson L, Newcombe RG, Dixon JM, et al. Post-operative arm morbidity and quality of life: results of the ALMANAC randomised trial comparing sentinel node biopsy with standard axillary treatment in the management of patients with early breast cancer. Breast Cancer Res Treat. 2006; 95:279–93.
Article3. Tew K, Irwig L, Matthews A, Crowe P, Macaskill P. Meta-analysis of sentinel node imprint cytology in breast cancer. Br J Surg. 2005; 92:1068–80.
Article4. Liu LC, Lang JE, Lu Y, Roe D, Hwang SE, Ewing CA, et al. Intraoperative frozen section analysis of sentinel lymph nodes in breast cancer patients: a meta-analysis and single-institution experience. Cancer. 2011; 117:250–8.5. Mamounas EP, Brown A, Anderson S, Smith R, Julian T, Miller B, et al. Sentinel node biopsy after neoadjuvant chemotherapy in breast cancer: results from National Surgical Adjuvant Breast and Bowel Project Protocol B-27. J Clin Oncol. 2005; 23:2694–702.
Article6. Hsieh K, Mage PL, Csordas AT, Eisenstein M, Soh HT. Simultaneous elimination of carryover contamination and detection of DNA with uracil-DNA-glycosylase-supplemented loop-mediated isothermal amplification (UDG-LAMP). Chem Commun (Camb). 2014; 50:3747–9.
Article7. Szychta P, Westfal B, Maciejczyk R, Smolarz B, Romanowicz H, Krawczyk T, et al. Intraoperative diagnosis of sentinel lymph node metastases in breast cancer treatment with one-step nucleic acid amplification assay (OSNA). Arch Med Sci. 2016; 12:1239–46.
Article8. Tsujimoto M, Nakabayashi K, Yoshidome K, Kaneko T, Iwase T, Akiyama F, et al. One-step nucleic acid amplification for intraoperative detection of lymph node metastasis in breast cancer patients. Clin Cancer Res. 2007; 13:4807–16.
Article9. Smolarz B, Krawczyk T, Westfal B, Maciejczyk R, Zadrozny M, Samulak D, et al. Comparison of one-step nucleic acid amplification (OSNA) method and routine histological investigation for intraoperative detection of lymph node metastasis in Polish women with breast cancer. Pol J Pathol. 2013; 64:104–8.
Article10. Cserni G. Intraoperative analysis of sentinel lymph nodes in breast cancer by one-step nucleic acid amplification. J Clin Pathol. 2012; 65:193–9.
Article11. Shi F, Liang Z, Zhang Q, Wang C, Liu X. The performance of one-step nucleic acid amplification assay for intraoperative detection of sentinel lymph node macrometastasis in breast cancer: An updated meta-analysis. Breast. 2018; 39:39–45.
Article12. Nie K, Qi SX, Zhang Y, Luo L, Xie Y, Yang MJ, et al. Evaluation of a direct reverse transcription loop-mediated isothermal amplification method without RNA extraction for the detection of human enterovirus 71 subgenotype C4 in nasopharyngeal swab specimens. PLoS One. 2012; 7:e52486.
Article13. Mohon AN, Hundt J, van Marle G, Pabbaraju K, Berenger B, Griener T, et al. Development and validation of direct RT-LAMP for SARS-CoV-2. Preprint at: https://www.medrxiv.org/content/10.1101/2020.04.29.20075747v2 . 2020.14. Nie K, Zhang Y, Luo L, Yang MJ, Hu XM, Wang M, et al. Visual detection of human enterovirus 71 subgenotype C4 and Coxsackievirus A16 by reverse transcription loop-mediated isothermal amplification with the hydroxynaphthol blue dye. J Virol Methods. 2011; 175:283–6.
Article15. Tanis PJ, Boom RP, Koops HS, Faneyte IF, Peterse JL, Nieweg OE, et al. Frozen section investigation of the sentinel node in malignant melanoma and breast cancer. Ann Surg Oncol. 2001; 8:222–6.
Article16. Giuliano AE, Dale PS, Turner RR, Morton DL, Evans SW, Krasne DL. Improved axillary staging of breast cancer with sentinel lymphadenectomy. Ann Surg. 1995; 222:394–9.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A reverse transcription loop-mediated isothermal amplification assay to rapidly diagnose foot-and-mouth disease virus C
- Uracil-DNA glycosylase-treated reverse transcription loop-mediated isothermal amplification for rapid detection of avian influenza virus preventing carry-over contamination
- Development of reverse transcription loop-mediated isothermal amplification assays for point-of-care testing of avian influenza virus subtype H5 and H9
- Rapid detection of Mycobacterium leprae for the diagnosis of leprosy: comparison between loop-mediated isothermal amplification and PCR.
- Loop-mediated isothermal amplification assay for the detection of Salmonella spp. in pig feces